Figures & data
Scheme 1. Reagents and conditions: Synthetic route to target compounds 4a–f and 7a–f. (i) KBrO3, 80°C; (ii) H2SO4 add over a period of 3 h; (iii) heat, 90°C−100°C, 2 h; (iv) CH3OH, H2SO4, reflux for 4 h; (v) NH2NH2.H2O, absolute C2H5OH, reflux for 6 h; (vi) POCl3, Ar-COOH, reflux for 8–12 h; (vii) KOH, CS2, absolute C2H5OH, reflux for 14 h, (viii) NH2NH2.H2O, reflux for 2 h; (ix) POCl3, Ar-COOH, reflux for 3–5 h. Where a, Ar= C6H5; b, Ar= 4-Cl-C6H4; c, Ar= 4-F-C6H4; d, Ar= 4-CH3-C6H4; e, Ar= 4-OCH3-C6H4; f, Ar= C11H9;
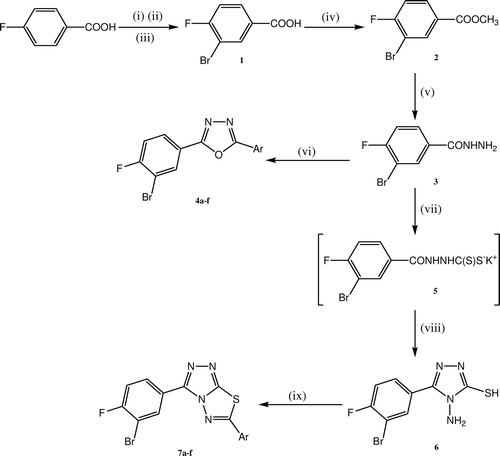
Table 1. Anti-inflammatory activity of the synthesized compounds after oral administration (20 mg/kg).
Table 2. Analgesic, ulcerogenic and lipid peroxidation activities of the synthesized compounds.