Figures & data
Table 1. Effect of the testosterone concentration on 5α-reductase type 1 activity at different pHs. Solubilized microsomes from rat liver were used as source of 5α-reductase type 1.
Figure 1. Steroidal structures and effect of different dehydroepiandrosterone derivatives on the activity of 5α-R enzyme type 1 and 2 (5α-R): 1–12. The IC50 values indicate the required concentration of the novel steroids for the inhibition of 50% of the activity of 5α-R1 and 5α-R2. This figure shows also the relative binding affinity (RBA) of these derivatives to the androgen receptor (AR). The following abbreviations were used: NA, non active compound.
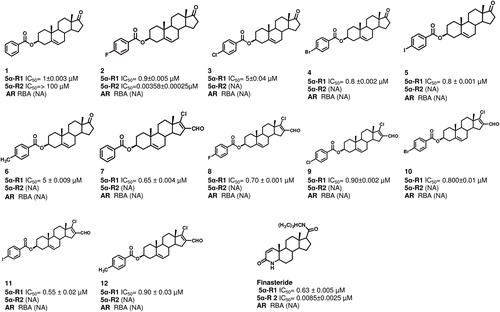
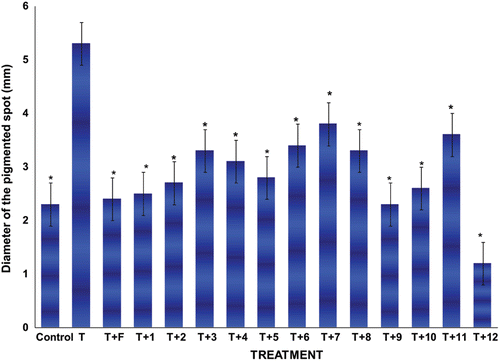
Figure 2. Diameter size ± standard deviations of the pigmented spot from gonadectomized hamster’s flank organs receiving different treatments with T plus the novel steroids for 6 days. Control gonadectomized hamsters were treated with vehicle only. The following abbreviations were used: Testosterone (T), Finasteride (F). *Significant differences with the T-treatment were observed.