Figures & data
Scheme 1. Reagents and conditions: (i) CH3I, NaH, DMF, rt, 3 h; (ii) DMF, POCl3, 80°C, 3 h; (iii) Substituted acetophenones, NaOH, Ethanol, rt, 24 h; (iv) Guanidine.HCl, DMF, NaH, 110°C, 10 h.
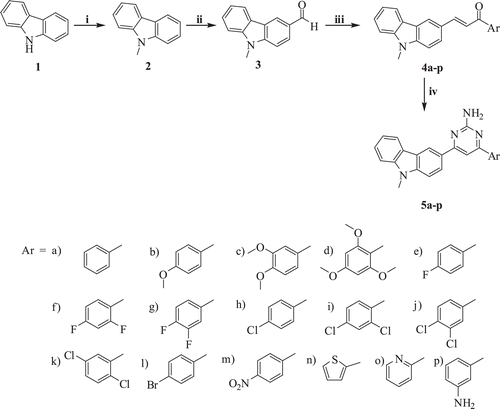
Table 1. Urease inhibition activity.
Table 2. Antimicrobial activity of pyrimidine derivatives.
Table 3. In silico pharmacological parameters for bioavailability.