Figures & data
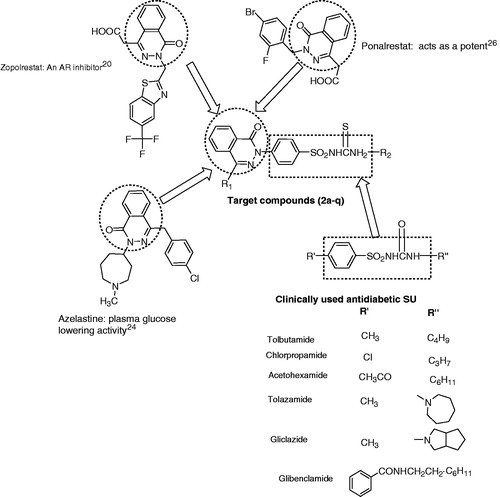
Figure 1. Structure of biologically active agent of benzenesulfonylthiourea’s as blood glucose lowering and rationally designed template for targeted compound.
Scheme 1. Reagents and conditions: (a) anhydrous AlCl3, room temperature; (b) absolute alcohol, reflux; (c) isothiocyanate, K2CO3, dry acetone, reflux 24–72 h. 1a, 2a, 2f, 2j, 2o: Ar = Phenyl; 1b, 2b, 2g, 2k, 2p: Ar = 4-Methylphenyl 2a–2e:R1 = –CH2C6H5; 1c, 2c, 2h, 2l: Ar = 4-Chlorophenyl; 1d, 2d, 2i, 2m, 2q: Ar = 4-Chloro-3-methylphenyl; 1e, 2e, 2n, 2k: Ar = 2-Chloro-5-methyl; 2f–2i: R1 = –C3H7; 2j–2n: R1 = –C4H9; 2o–2q: R1 = –C6H11.
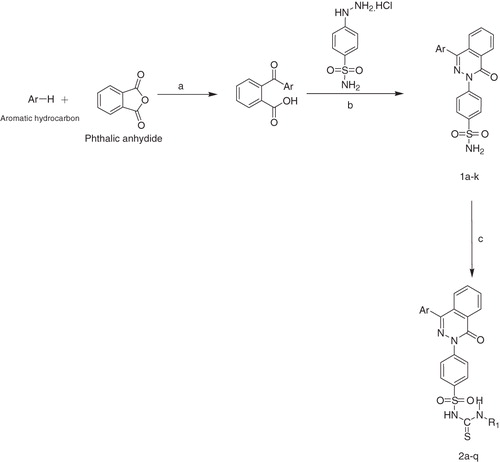
Table 1. Antihyperglycemic activity of SU derivatives (2a–q) and the standard drug gliclazide in glucose-fed (3g/kg) hyperglycemic normal rats.
