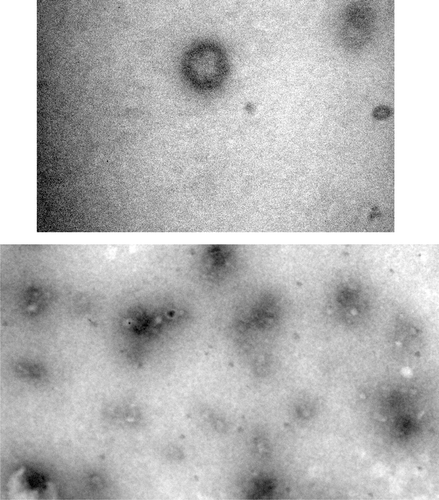
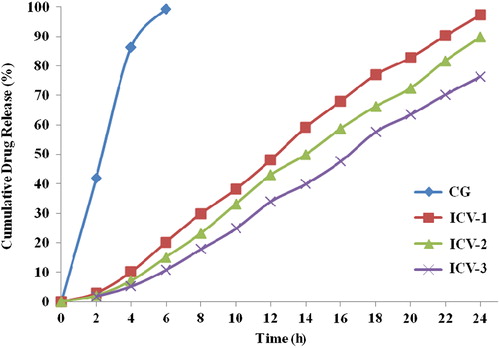
Figures & data
Table I. Composition for ICV formulations (drug lipid ratio = 1:3).
Table II. Physicochemical characterization.
Table III. Physicochemical characterization and drug release of ICV-1 after 90 days at accelerated conditions.
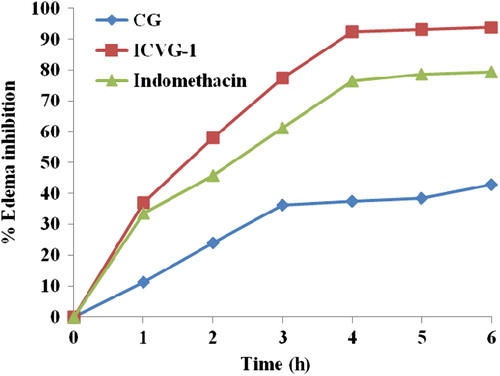
Figure 4. Drug content accumulated in different skin strata. SC, stratum corneum; VED, viable epidermis; D, Dermis; RF, receptor fluid.
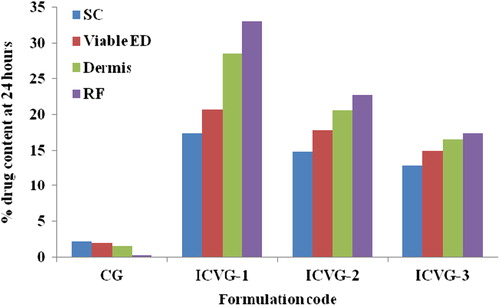
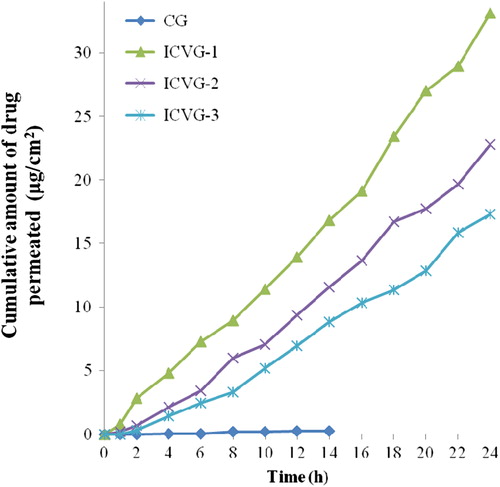
Table IV. Permeation parameters for CG and ICVG formulations.
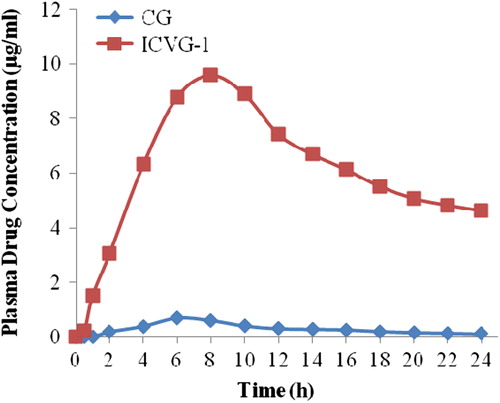
Table V. Pharmacokinetic data of the CG and ICVG-1.