Figures & data
Figure 1. Single-channel currents recorded in the outside-out configuration from HEK293 cells that were stably expressing TRPC4β. (A) Representative single-channel current traces were recorded at voltages ranging from +80 to -140 mV. The holding potentials were 80, 30, -60 and -120 mV in A. GTPγS (200 μM) was intracellularly applied to activate the TRPC4 channel under the condition of 125 mM [Cs+]i and 125 mM [Cs+]o and nominally [Ca2+]o-free. (B) The corresponding I-V relationships from each potential are shown in the upper panel and the red line represents the fit to obtain a unitary conductance. The plot of NPo was estimated from each potential (bottom panel).
![Figure 1. Single-channel currents recorded in the outside-out configuration from HEK293 cells that were stably expressing TRPC4β. (A) Representative single-channel current traces were recorded at voltages ranging from +80 to -140 mV. The holding potentials were 80, 30, -60 and -120 mV in A. GTPγS (200 μM) was intracellularly applied to activate the TRPC4 channel under the condition of 125 mM [Cs+]i and 125 mM [Cs+]o and nominally [Ca2+]o-free. (B) The corresponding I-V relationships from each potential are shown in the upper panel and the red line represents the fit to obtain a unitary conductance. The plot of NPo was estimated from each potential (bottom panel).](/cms/asset/ce396622-597b-4463-baeb-e1a0f72e480f/kchl_a_10921198_f0001.gif)
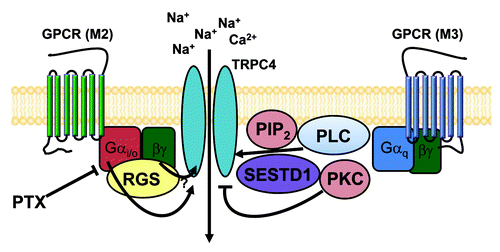
Figure 2. Positive regulatory mechanisms of TRPC4 and TRPC5 channels. TRPC4 and TRPC5 channels are activated by many signals. Extracellular chemical signals, such as the GPCR agonist, lipids, metal ions, EGF and NO, activate TRPC4 and TRPC5 channels directly via Gi/o proteins, calcium, CaM, nitrosylation and PIP2 or indirectly via store depletion and exocytosis.
Figure 3. Negative regulatory mechanisms of TRPC4 and TRPC5 channels. Phosphorylation of TRPC4 and TRPC5 channels by PKA and PKC works as an inactivation pathway of TRPC4 and TRPC5 channels. On the other hand, PtdIns(4,5)P2 hydrolysis negatively regulates TRPC4 and TRPC5.
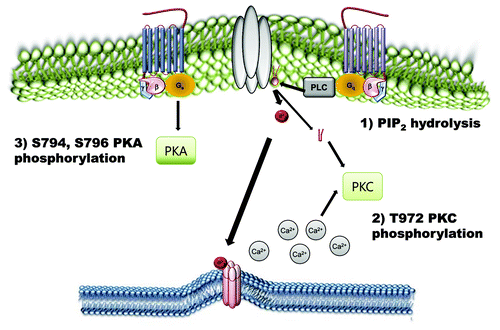
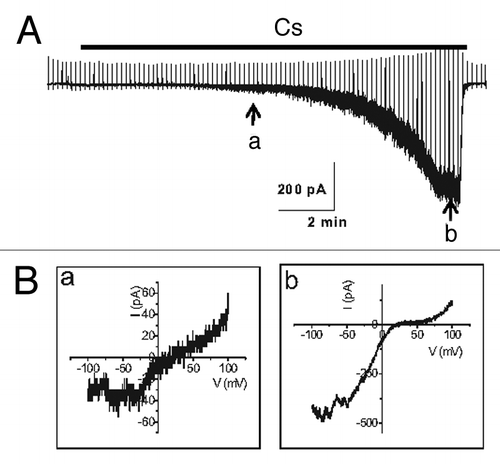
Figure 4. Current trace in the activation of TRPC4β by Gαi2. (A) Representative current traces recorded at -60 mV. The time course of activation of TRPC4β by Gαi2 was delayed in the presence of 10 mM EGTA in the patch pipette, in contrast to the current recorded with the low-calcium buffered internal solution (0.5 mM EGTA). (B) The current-voltage (I-V) relationship of TRPC4 showed a typical doubly rectifying shape. The I-V relationship shown in a and b was obtained at point a and b of (A).