Figures & data
Figure 1. Diagram depicting anatomy of the stomach and histological representation of the oxyntic glands of the body of the stomach. It is these glands, which include parietal cells, that are lost in gastric atrophy. Reproduced with permission from Fox and Wang 2007.Citation16
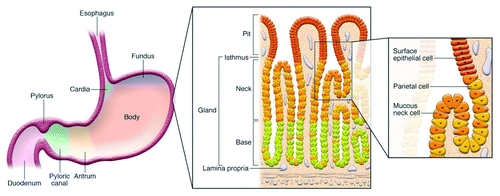
Figure 2. Taxonomic classification of bacteria. Descriptions of the gastric microbiota focus on the levels of phylum and genus.
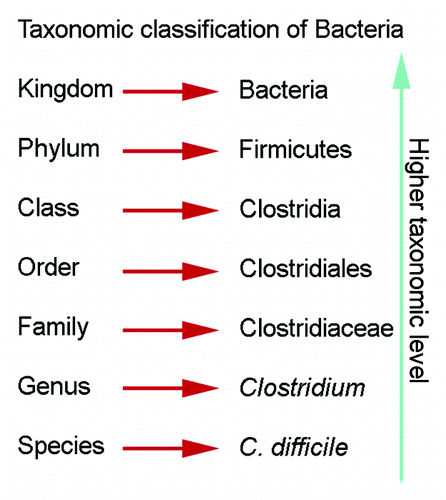
Table 1. Studies analyzing the human gastric microbiota using culture-based methods
Table 2. Studies analyzing the human gastric microbiota using 16S rRNA identification methods
Figure 3. Human microbiota composition in multiple sites of the GI tract, including mouth, stomach, duodenum, colon and stool. Note the high variability between individuals and between the antrum and corpus in the stomach. The stomach microbiota also differs significantly from other sites in the GI tract. Reproduced with permission from Stearns et al. 2011.Citation96
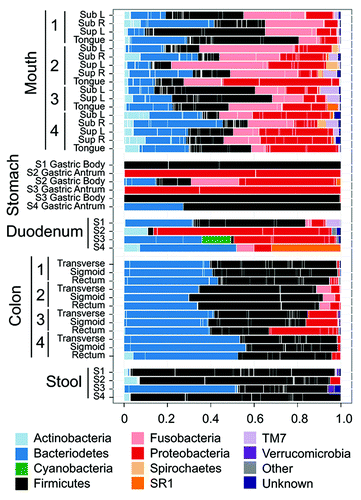
Figure 4. Illustration depicting anatomy of the mouse stomach. The anatomy of the gerbil stomach is similar. The nonglandular forestomach is the site of dense colonization by lactobacilli, which substantially contribute to the differences in the gastric microbiota of humans and rodents.
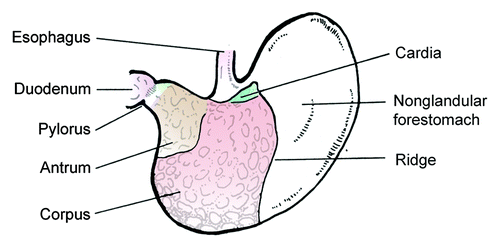
Table 3. Gastric microbiota studies in mice
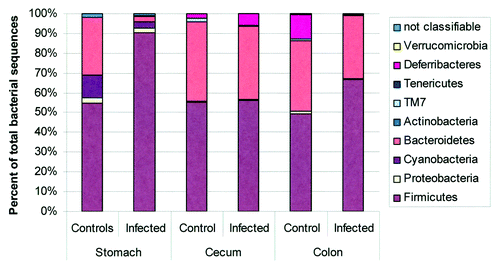
Figure 5. Microbiota composition in stomach, cecum, and colon of H pylori-infected male INS-GAS mice (n = 3, 15 weeks postinfection) vs. uninfected controls (n = 2). Note the significant increase in the relative abundance of Firmicutes and decrease of Bacteroidetes in the stomachs of H pylori–infected INS-GAS mice (p < 0.05), whereas no significant changes were observed in the colon and ceca of H. pylori-infected mice. Reproduced with permission from Lofgren et al. 2011.Citation77