Figures & data
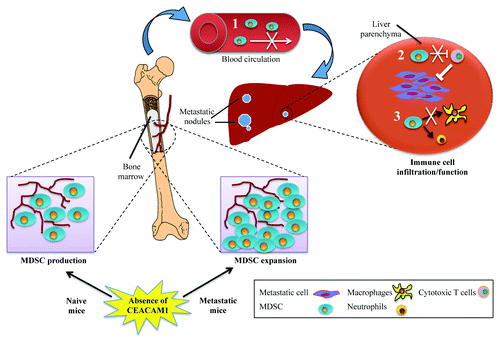
Figure 1. Dysregulation of bone marrow myeloid-derived suppressor cells in CEACAM1-deficient mice under metastatic challenge. Upon formation of metastatic nodules in the liver, Ceacam1−/− (but not wild type) bone marrow (BM) myeloid-derived suppressor cells (MDSCs) undergo expansion. However, this is not followed by increased MDSC infiltration into metastatic livers. Given that the adoptive transfer of wild type MDSCs into Ceacam1−/− mice results in increased metastatic burden, several possible mechanisms can be proposed to explain the role of CEACAM1 in tumor metastasis. (1) migratory defect of BM MDSCs; (2) reduced cytotoxic T-cell inhibition by MDSCs; or (3) blockage of monocytic and/or granulocytic differentiation. As a consequence, tumor infiltration/effector functions of MDSCs are diminished in Ceacam1−/− livers, resulting in enhanced immune responses that efficiently eliminate/control metastatic tumor cells.