Figures & data
Figure 1. Detection of autoantibodies associated with the anti-Hu syndrome. (A) Patient sera containing Hu-specific autoantibodies specifically recognize neurons in the central nervous system (CNS) (magnification 200 ×). Briefly, hippocampal (left panels) and cerebellar (right panels) rat slices (EuroImmun) were incubated with serum from either an individual not affected by paraneoplastic neurological disorders (PNDs) (negative control), either a patient with a confirmed anti-Hu syndrome (positive control), or a subject suspected to suffer from the anti-Hu syndrome, followed by the detection of bound IgG using biotinylated goat anti-human IgG, ABC and VIP kits (Eurobio). The test serum turned out to contain anti-neuron antibodies exhibiting a staining pattern compatible with that of anti-Hu antibodies. (B) The molecular identification of the specificity of such neuron-specific IgGs is made by immunoblotting-based diagnostic tests. Patient-derived serum (1/100 dilution) and cerebrospinal fluid (when available) are incubated on a membrane stripped (right panel) with paraneoplastic neurological syndrome-associated antigens, including amphiphysin, CV2, PNMA2 (Ma2/Ta), Ri, Yo, and Hu proteins (EuroImmun). Upon washing, bound IgGs are detected using alkaline phosphatase-conjugated anti-human IgG antibodies and their position is confronted to a reference scheme.
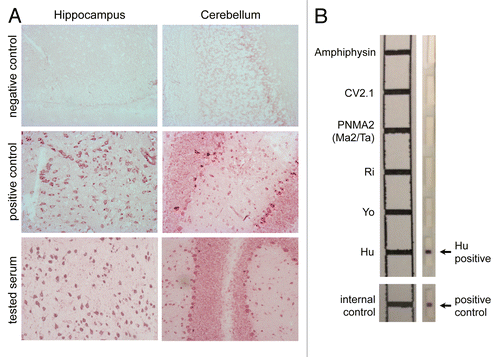
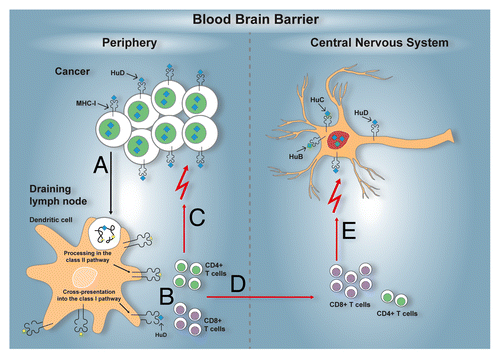
Figure 2. Immunopathogenesis of the anti-Hu syndrome. An anti-Hu syndrome occurs when a vigorous immune response develops against the normally neuron-restricted antigen HuD ectopically expressed by an underlying tumor, most often small-cell lung carcinoma (SCLC). Tissue-resident dendritic cells capture antigens derived from malignant cells, including HuD (A). After the homing of these dendritic cells to tumor-draining lymph nodes, tumor-associated antigens, including HuD, are processed and presented to activate antigen-specific CD4+ and CD8+ T cells (B). The activation of tumor-specific T cells may also occur within neoplastic lesions. In either case, tumor-specific T cells can reach neoplastic lesions. As cancer cells sometimes express MHC class I molecules and likely present HuD-derived peptides, they become attractive targets for HuD-specific CD8+ T cell killing. Partial (or in rare cases complete) control of the tumor growth is therefore afforded (C). In parallel, the HuD-specific T cells activated in tumor-draining lymph nodes and/or within neoplastic lesions, circulate and acquire the capacity to cross the blood-brain-barrier (D). In the central nervous system (CNS), neurons can also express MHC class I molecules, in particular under inflammatory conditions, and can therefore present peptides derived from the highly homologous HuD, HuB, HuC proteins. Thus, neurons become additional targets for HuD-specific CD8+ T cells, resulting in neuronal tissue damage and the related paraneoplastic neurological manifestations (E).