ABSTRACT
Background: Ibogaine is a monoterpene indole alkaloid used in medical and nonmedical settings for the treatment of opioid use disorder. Its mechanism of action is apparently novel. There are no published prospective studies of drug use outcomes with ibogaine. Objectives: To study outcomes following opioid detoxification with ibogaine. Methods: In this observational study, 30 subjects with DSM-IV Opioid Dependence (25 males, 5 females) received a mean total dose of 1,540 ± 920 mg ibogaine HCl. Subjects used oxycodone (n = 21; 70%) and/or heroin (n = 18; 60%) in respective amounts of 250 ± 180 mg/day and 1.3 ± 0.94 g/day, and averaged 3.1 ± 2.6 previous episodes of treatment for opioid dependence. Detoxification and follow-up outcomes at 1, 3, 6, 9, and 12 months were evaluated utilizing the Subjective Opioid Withdrawal Scale (SOWS) and Addiction Severity Index Composite (ASIC) scores, respectively. Results: SOWS scores decreased from 31.0 ± 11.6 pretreatment to 14.0 ± 9.8 at 76.5 ± 30 hours posttreatment (t = 7.07, df = 26, p < 0.001). At 1-month posttreatment follow-up, 15 subjects (50%) reported no opioid use during the previous 30 days. ASIC Drug Use and Legal and Family/Social Status scores were improved relative to pretreatment baseline at all posttreatment time points (p < .001). Improvement in Drug Use scores was maximal at 1 month, and subsequently sustained from 3 to 12 months at levels that did not reach equivalence to the effect at 1 month. Conclusion: Ibogaine was associated with substantive effects on opioid withdrawal symptoms and drug use in subjects for whom other treatments had been unsuccessful, and may provide a useful prototype for discovery and development of innovative pharmacotherapy of addiction.
Introduction
Ibogaine, a monoterpene indole alkaloid that occurs in the root bark of Tabernanthe iboga Baill., is used for the treatment of substance use disorders (SUDs) (Citation1,Citation2). It has been associated with controversy, and the medical and nonmedical settings of ibogaine use have been collectively designated as a “vast uncontrolled experiment” (Citation3), or “medical subculture” (Citation1). Ibogaine has been classified as a hallucinogen and illegal in the US since 1967, and is similarly scheduled in 9 of the 28 countries presently in the European Union. It is unregulated, i.e., neither officially approved nor illegal in much of the rest of the world. New Zealand, Brazil, and South Africa have classified ibogaine as a pharmaceutical substance and restrict its use to licensed medical practitioners.
Ibogaine is used most frequently for detoxification from opioids. A previous study on the known settings of ibogaine use as of 2006 found that approximately 3,400 individuals had taken ibogaine, 68% of whom did so for the treatment of a substance-related disorder, and 53% specifically for opioid detoxification (Citation1). A substantive effect of ibogaine in opioid detoxification is reported in studies of 33 individuals treated in nonmedical settings with single mean dosages of 19.3 ± 6.9 mg/kg (Citation4), and 32 individuals treated in a medical setting with fixed dosages of 800 mg (Citation5). Unpublished data include a series of 53 treatment episodes that was presented to the National Institute on Drug Abuse (NIDA) and influenced NIDA’s decision to undertake an ibogaine project from 1991 to 1995 (Citation6), and an academic thesis on a Web-based survey of 21 individuals who had used ibogaine (Citation7). The subjects in these unpublished case series were predominantly opioid users, and ibogaine appeared to be effective in opioid detoxification, and about one-third of subjects reported abstinence from opioids for periods of 6 months or longer following treatment. A retrospective study on 75 subjects, a subset available from a group of 195 individuals who have been treated or cocaine dependence, almost none of whom used opioids, reported a median relapse-free interval of 5.5 months following single doses of ibogaine of 7.5 to 20 mg/kg (Citation8). Published follow-up regarding drug use outcomes beyond detoxification in opioid use disorder (OUD) has been limited to a small number of case reports (Citation9–Citation11).
Consistent with its apparent effect in opioid detoxification in humans, ibogaine administered intraperitoneally or intracerebrally to animals reduces naloxone- or naltrexone-precipitated opioid withdrawal signs, in rats (Citation12–Citation15), mice (Citation16–Citation19), and primates (Citation20,Citation21). Single dosages of ibogaine administered to rodents diminish self-administration of multiple abused substances including morphine (Citation22–Citation25), heroin (Citation26), cocaine (Citation24,Citation25,Citation27–Citation29), amphetamine (Citation30), and alcohol (Citation31), with normal responding for water. A notable aspect of these self-administration studies has been the observation of a treatment effect of a duration of 48 to 72 hours averaged for the entire sample, with sustained effects for longer time intervals in individual animals. Both ibogaine and its synthetic structural analog 18-methoxycoronaridine (18-MC) diminish an experimental correlate of drug salience, the sensitized response of dopamine efflux in the nucleus accumbens in response to morphine (Citation32,Citation33) and nicotine (Citation34,Citation35).
The mechanism of action of ibogaine is apparently novel, and unexplained by the receptor interactions of medications known to have clinical effects in opioid tolerance or withdrawal (Citation6,Citation36,Citation37). As a small molecule that modifies opioid withdrawal and drug self-administration, ibogaine may offer an interesting prototype for drug discovery and neurobiological investigation. Ibogaine, its major metabolite noribogaine, and 18-MC do not act as orthosteric μ opioid receptor (MOR) agonists. Although ibogaine, noribogaine, and 18-MC bind with low micromolar affinity to the MOR, these compounds do not activate G proteins assessed by the binding of [35S]GTPγS in cells expressing the MOR (Citation36), and the dosages of ibogaine used for detoxification in the setting of severe physical dependence do not produce signs of overdose in opioid-naïve individuals (Citation1).
Ibogaine potentiates morphine analgesia without producing analgesia when administered alone (Citation16,Citation38–Citation43), as might be expected of an allosteric MOR agonist. However ibogaine, noribogaine, and 18-MC do not potentiate the activation of G proteins by morphine or DAMGO (Citation36), indicating that these compounds do not act as allosteric MOR agonists. The reversal by ibogaine of analgesic tolerance to chronic morphine suggests that ibogaine may modify neuroadaptations associated with chronic exposure to opioids (Citation39,Citation43,Citation44).
Ibogaine is an NMDA receptor antagonist (Citation19,Citation45), and NMDA antagonists such as memantine diminish signs of opioid withdrawal in preclinical models and humans (Citation46,Citation47), however 18-MC lacks significant affinity for the NMDA receptor and is equally effective as ibogaine in animal models of opioid withdrawal (Citation12–Citation15,Citation37,Citation48,Citation49). Ibogaine has no significant affinity for the α2 receptor or imidazoline I2 site (Citation50,Citation51), indicating that it does not act as an imidazoline α2 adrenergic receptor agonist such as clonidine (Citation52).
The enhanced expression of glial-derived neurotrophic factor (GDNF) has been proposed to account for ibogaine’s effect on drug self-administration (Citation53). Ibogaine increases the GDNF expression in vivo and in cultured cells, and 18-MC reportedly does not (Citation54), but both compounds are equally effective in animal models of drug self-administration (Citation37). Ibogaine’s action as an allosteric antagonist of the α3β4 nicotinic acetylcholine receptor (nAChR) is suggested to mediate its effect on drug self-administration (Citation55), but does not appear to readily explain the prolonged effects that appear to persist beyond pharmacokinetic elimination (Citation56). Ibogaine’s major metabolite, noribogaine (Citation57,Citation58) has a longer half-life than the parent compound, and has been suggested to account for persistence of effects on drug self-administration and withdrawal (Citation59), although in the animal model the effect of ibogaine in reducing drug self-administration appears to persist beyond the elimination of ibogaine and noribogaine from serum or brain tissue (Citation56).
There are no published prospective studies of ibogaine in the treatment of OUD reporting on drug use outcomes subsequent to detoxification. This study reports on outcomes up to one year following opioid detoxification with ibogaine.
Methods
Treatment setting, inclusion and exclusion criteria, and subject enrollment
The Human Research Review Committee at the California Institute of Integral Studies approved this observational study. By definition, an observational study assesses dependent variables, in this case detoxification and drug use outcomes, without modifying the independent variable, in this case the parameters of treatment, and the investigators had no role in administering ibogaine or clinical management. This study is naturalistic with respect to its focus on treatment as usual in a representative setting of two private clinics that provided treatment with ibogaine on a fee-for-service basis, located respectively in Ensenada (25 subjects), and Playas de Tijuana (5 subjects) in Baja California, Mexico. Treatment consisted of administration of ibogaine followed by a clinic stay of 3 to 6 days.
The study sample consisted of 30 individuals who sought treatment with ibogaine for detoxification from opioids. All of the subjects met criteria for DSM-IV Opioid Dependence with Physiological Dependence on opioids (Citation60). Individuals were excluded from the study if they took ibogaine for any purpose other than the treatment of OUD, if they had a history of prior treatment with ibogaine, or if they appeared to have personal, health, situational, social or other problems which in the view of the investigator would prevent them from being able to fully comply with the requirements of this study. As admission criteria, subjects were required to have a reliable method of communication by which the researchers could contact them during the study follow-up period and to provide the name and contact information of at least one other individual such as a therapist, counselor, parent, spouse or close friend, to communicate with the research team and provide corroboration regarding the subject’s drug use.
During the enrollment period, 30 subjects signed the Information and Consent Form and were enrolled into the study. An additional nine individuals were not enrolled because they had received ibogaine before they could be asked to participate in the study, and 4 declined to participate. Participants were given $10 in the form of a gift certificate for each follow-up phone interview they completed after leaving the clinic.
Study sample
summarizes demographic, drug use, and treatment history characteristics of the study sample. At the time of enrollment in the study, the drugs most commonly cited as the most problematic were heroin (n = 14), and prescription analgesic opioids (n = 10), mostly oxycodone. Subjects were heavy, and relatively selective users of opioids, averaging 29.0 ± 3.2 days of opioid use in the previous 30 days. Eighteen subjects (60%) reported no stimulant use and 18 (60%) reported no alcohol use in the previous 30 days, and only 3 subjects reported five or more drinking occasions of at least five drinks in the previous 30 days. At pretreatment baseline (N = 30), Drug Use was the highest and Alcohol Use was the lowest of all of the ASIC scores (see ).
Table 1. Demographic and drug use characteristics and prior treatment history of the study sample.
Table 2. ASIC scores at pretreatment baseline and 1, 3, 6, 9, and 12 months, and opioid free days: Paired t-tests were used to compare ASIC scores at 1, 3, 6, 9, and 12 months posttreatment to their baseline pretreatment values (N = 30; significance level of p-values indicated by †). Noninferiority tests were used to compare ASIC scores at 3, 6, 9, and 12 months posttreatment to their 1-month posttreatment values (n = 20; significance level of p-values indicated by *). The means and standard deviations are unadjusted and computed on the subjects (n) available at the respective time point. The p-values are adjusted for missing follow-up data by performing the respective statistical tests with missing values set equal to their baseline pretreatment value as described in Methods. Opioid-free days in the previous 30 days are shown in the lower part of the Table.
Subjects had been regularly using at least one opioid substance for an average of 5.2 ± 3.0 consecutive years before presenting for treatment. On days the subjects used heroin, they averaged 1.3 ± 0.94 g/day, the majority by the intravenous route. Among users of oxycodone, the mean daily amount of oxycodone used was 250 ± 180 mg/day. Twenty-nine of the 30 subjects (97%) had previously received treatment for SUD, and the study sample averaged 3.1 ± 2.6 prior treatment episodes, most commonly methadone or buprenorphine maintenance or residential treatment.
Treatment
Prior to the administration of ibogaine and subsequent to arrival at the clinic, subjects were stabilized on a short acting opioid, most often oxycodone, for two to three days in a range of 90 mg to 270 mg per day divided into three doses. Subjects on long-acting opioids had been instructed to change over to short acting opioids for at least two weeks prior to their treatment. An average total dose of 1,540 ± 920 mg ibogaine HCl, 94% purity by certificate of analysis was administered to each subject as described below. Five of those subjects additionally received a crude extract of T. iboga root bark (mean dose 1610 ± 1650 mg). Crude “total alkaloid” extracts have an estimated total alkaloid content between 15% and 50%, about 25% to 50% of which might be expected to be ibogaine (Citation1,Citation61). No opioids were administered subsequent to the initiation of treatment with ibogaine, and subjects did not receive any additional ancillary medications.
The treatment was initiated with a “test” dose of ibogaine of 3 mg/kg, typically administered in the morning after subjects had abstained from opioid use overnight. The test dose was given when subjects had begun to exhibit three or more initial signs of withdrawal, such as restlessness, sweating, yawning or watery eyes. In the experience of ibogaine treatment providers, the test dose typically has some effect of reducing withdrawal signs, and they view the response to the test dose as providing some indication of the degree of physical dependence on opioids. A larger, “flood” ibogaine dose, typically four times the test dose is given approximately 2 to 12 hours after the test dose. The flood dose was sometimes followed by an additional “booster” dose of ibogaine of 3 to 5 mg/kg, at an interval following the flood dose commonly in the range of 1 to 16 hr. Providers administered booster dosages to alleviate residual or re-emergent withdrawal symptoms, or at the election of the patient to increase the intensity of the psychoactive experience.
A set of clinical guidelines for detoxification from opioids with ibogaine has been published online by a group of physician and lay ibogaine treatment providers (Citation62) and is representative of the approach by the providers of treatment in this study. Briefly, pre-treatment evaluation included medical history, EKG, and electrolyte and liver function tests. Monitoring throughout the treatment included continuous pulse oximetry and three-lead EKG, and monitoring of blood pressure. Intravenous access was maintained during the treatment, with a medical professional (MD, nurse, or paramedic) certified in Advanced Cardiac Life Support (ACLS) present for at least the first 24 hours of the treatment.
Assessment and rating
The baseline pretreatment interview was conducted after the participant’s arrival at the clinic, one to three days prior to the administration of ibogaine. The first author (T.B.) conducted the follow-up interviews with subjects and their significant others, either in-person at the clinic or by telephone. Subjects were followed up at intervals of 1, 3, 6, 9, and 12 months following treatment with ibogaine. A member of the research team also attempted to contact by phone a corroborating person identified by the study participant to independently verify information regarding the participant’s substance use from the time of baseline assessment throughout the 12-month follow-up period.
Subjects were evaluated using the Addiction Severity Index (ASI), Lite version (Citation63) at pretreatment baseline and at follow-up intervals of 1, 3, 6, 9, and 12 months after treatment with ibogaine. The ASI has seven composite measures with distinct item content entitled Drug Use, Alcohol Use, and Family/Social, Employment, Legal, Medical, and Psychiatric Status (Citation64). Each ASI Composite (ASIC) score is a sum of values of ASI items, scaled variously as the number of days in the last 30 days, log transformation, binary values, or as continuous ratings of severity. ASIC item values are normalized relative to their maximal possible values, weighted equally and summed. The item sum totals divided by the number of items yields the ASIC score with a value in the range of 0 to 1.0. Higher ASIC scores indicate greater problem severity.
Detoxification outcomes were assessed with the 16-item Subjective Opioid Withdrawal Scale (SOWS) (Citation65), which consists of 16 questions regarding withdrawal symptoms on a zero through the four-point scale. The baseline SOWS was administered within one hour prior to the test dose. The time intervals between test, flood and possible booster dosages were at the election of the provider and non-uniform across subjects. The follow-up SOWS was administered when the provider viewed the treatment as completed, with no need for further dosing, with the subject exhibiting no withdrawal signs, ambulating and communicating normally, and eating.
Statistical tests
The effect of ibogaine in opioid detoxification was evaluated with paired t-tests comparing pre- versus posttreatment SOWS scores. Posttreatment follow-up ASIC scores were compared using paired t-tests and noninferiority tests. Statistical analysis was performed using SAS® software Version 9.4 of the SAS System for Windows 10.
Paired t-tests were used to compare ASIC scores at 1, 3, 6, 9 and 12 months to their pretreatment baseline. An additional question posed to the ASI data was whether a treatment effect observed at 1 month was sustained at subsequent 3-, 6-, 9-, and 12-month time points. One month was selected on the basis of prior observation as a posttreatment time interval at which an ibogaine treatment effect is particularly evident (Citation1). This question was addressed with noninferiority tests (Citation66,Citation67) on the 20 subjects available for follow-up at 1 month. The null hypothesis of the noninferiority test is that the posttreatment 3-, 6-, 9-, and 12-month ASIC scores are greater (worse) than their 1-month posttreatment values by a margin of ≥0.1 ASIC point. Rejection of the null hypothesis on the basis of a low p-value indicates that relative to its 1-month value, the ASIC score at the later time point had not increased by a margin of ≥0.1 point, and was noninferior, i.e., not worse, suggesting a sustained treatment effect that is at least equivalent, or better relative to that seen at 1 month. Conversely, nonsignificant noninferiority indicates that the effect at the later time point does not reach equivalence to the effect evident at 1 month.
As a conservative adjustment for missing follow-up data, the p-values of the statistical tests on ASIC scores were calculated setting missing posttreatment values equal to their baseline pretreatment value. Paired t-tests comparing posttreatment values to their pretreatment baseline were performed utilizing the total study N = 30 subjects, setting the missing values of subjects unavailable at the 1-, 3-, 6-, 9-, and 12-month time points to their pretreatment baseline values. The noninferiority tests used 1-month values as the reference comparison, so these tests were performed on only those n = 20 subjects that were available for follow-up at 1 month, with missing subsequent 3-, 6-, 9-, and 12-month values set to their pretreatment baseline values.
Results
Sows
The SOWS score for the entire study sample (a pre- and posttreatment SOWS was returned on 27 subjects) decreased from pretreatment baseline 31.0 ± 11.6 to 14.0 ± 9.8 following ibogaine treatment, a mean reduction of 17.0 ± 12.5 points (t = 7.07, df = 26, p < .001). The mean time interval between the baseline and second SOWS was 76.5 ± 30 hrs.
The original study of the development and validation of the 16-item SOWS by Handelsman et al. (Citation65) found that that the effect of methadone on SOWS scores was smaller in opioid-dependent subjects with comorbid abuse of other substances (alcohol, stimulants, or sedative/hypnotics) relative to those without comorbid substance abuse (Citation65). Accordingly, we compared pre- versus posttreatment change in SOWS scores in subjects with (n = 11), and without (n = 16) comorbid substance use, defined as 5 or more drinking occasions of at least 5 drinks, any stimulant use, or more than 5 days of use of benzodiazepines in the previous 30 days. No significant difference was found, SOWS scores decreased by 16.8 ± 12.4 points in the group with comorbid substance use, versus 17.2 ± 12.7 points in the group without (t = 0.09, df = 20.7, p = .928).
Tests of difference on ASIC scores relative to baseline at 3-, 6-, 9-, and 12-month follow-up
summarizes the results of paired t-tests of difference on ASIC scores comparing 1-, 3-, 6-, 9-, and 12-month values to their pretreatment baseline. In the Table, p-values are adjusted for missing data as described in Methods, and the ASIC scores are unadjusted. Significantly decreased ASIC scores, indicating improvement relative to pretreatment baseline were evident at all posttreatment time points for Drug Use (p < .001 at 1, 3, 6, 9, and 12 months), Family/Social Status (p < .001 at 1, 3, 9 and 12 months; p < .01 at 6 months), and Legal Status (p < .01 at 1, 3, and 12 months; p < .05 at 6 and 9 months).
Alcohol Use and Medical Status scores did not differ significantly from baseline at any time point. The apparent lack of a clear trend regarding the Alcohol Use score across the posttreatment follow-up interval may reflect the relatively low use of alcohol in the study sample. A decrease in Psychiatric Status scores from baseline at 1 month approached significance (p = .053, data not shown in Table), and scores were lower than their pretreatment baseline at 3 months (p < .05) and 6 months (p < .01). The Employment Status score was significantly increased relative to pretreatment baseline only at 1 month (p < .01), without significant differences at other time points, possibly explained by the interruption of employment in order to participate in treatment.
Tests of noninferiority on ASIC scores relative to 1 month over the subsequent posttreatment interval of 3 to 12 months
includes noninferiority tests of ASIC scores at 3-, 6-, 9-, and 12-month posttreatment follow-up compared to 1-month posttreatment, with p-values adjusted for missing subjects as described in Methods. The noninferiority tests were significant for Family/Social (p < .01 at 3 months; p < .01 at 6 and 9 months) and Legal Status scores (p < .01 at 3 and 9 months; p < .05 at 6 and 12 months) indicating that improvement in these measures at the 1-month posttreatment interval was sustained at these subsequent time points.
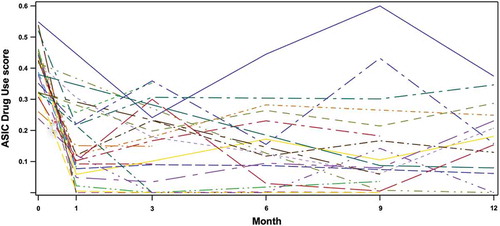
The noninferiority results on Drug Use scores adjusted for missing data were not significant at any time point (). In contrast, without the adjustment for missing data, the noninferiority results on Drug Use scores are significant at p < .001 at all posttreatment time points (data not shown). plots individual trajectories on the Drug Use score across the duration of the study, and appears to indicate an effect at 1 month that persists up to 12 months in a subset of individual subjects. The differential significance of the noninferiority test results utilizing adjusted in contrast to unadjusted Drug Use scores likewise suggests a subset of responders for whom a treatment effect is sustained across the 12-month follow-up interval, although the group noninferiority result becomes nonsignificant under the conservative assumption that all missing subjects have relapsed to their baseline. For the entire sample of subjects, taken together with the results of the paired t-tests on ASIC scores that compare subsequent time points with pretreatment baseline, the noninferiority results suggest sustained reduction in Drug Use relative to pretreatment baseline across the 3- to 12-month posttreatment interval that does not reach equivalence to the effect evident at 1 month.
Figure 1. Individual trajectories of the ASIC Drug Use score in all subjects available at one or more follow-up time points subsequent to treatment (n = 26).
Psychiatric Status scores were significantly noninferior (p < .05), at 3 and 6 months, indicating that the marginal 1-month improvement relative to baseline was sustained at these subsequent time points. Alcohol Use and Medical Status scores yielded some significant results on noninferiority tests, however neither score differed from baseline as indicated by the paired t-tests (), here the significant noninferiority results are consistent with the apparent stability of these scores across the pre- and posttreatment time interval. Similarly, apart from the transient elevation at 1 month noted above, Employment Status scores were unchanged from baseline at all other time points, with the significant noninferiority results consistent with the relative stability of this score at time points other than 1-month posttreatment.
Relationship of baseline characteristics to subsequent outcome
A subset of 12 subjects had favorable outcomes, defined as retention at 9 or 12 months with ≥75% reductions of the Drug Use score relative to pretreatment baseline. This subset of 12 subjects with favorable outcomes did not differ significantly from the remaining subjects (n = 18) with regard to any of the demographic features in . No difference was found between these two groups with respect to their rates of use of non-opioid substances, or opioids other than methadone (see below), although there was a marginally higher rate of benzodiazepine use in the group with less favorable outcomes (Fisher’s Exact p = .072). The two groups did not differ regarding whether the substance they identified as most problematic was heroin (Fisher’s Exact p = .457) or prescription opioid analgesics (Fisher’s Exact p = .622), or whether they reported the intravenous route as the predominant route of heroin self-administration (Fisher’s Exact p = .709).
Exposure to methadone, defined as any use of methadone in the previous 30 days or a prior history of methadone maintenance treatment, was associated with less favorable outcome (Fisher’s Exact p = .002). In contrast, exposure to buprenorphine did not differ between the two groups (Fisher’s Exact p > .999). The subjects with less favorable outcomes averaged 3.8 ± 2.9 prior treatment episodes versus 2.0 ± 1.6 in the favorable outcome group (t = 2.16, df = 27.3, p = .041). Subjects with histories of methadone treatment (n = 13) had a mean of 4.5 ± 3.1 prior treatment episodes, compared to 2.0 ± 1.5 in the subjects (n = 17) without a history of methadone treatment (t = 2.66, df = 16.4, p = .017). The dataset did not permit an analysis of mediation of outcome by methadone exposure per se versus the number of prior treatment episodes.
Subjects receiving other SUD treatment during follow-up
A total of 9 subjects received additional treatment during the 12 months following opioid detoxification with ibogaine, with a total of 10 treatment episodes: drug-free residential (n = 4; mean 2.3 ± 1.1 months, range 0.5 to 3.1 months), opioid agonist maintenance (n = 3; mean 2.3 ± 2.2 months, range 1.0 to 4.8 months), or an additional subsequent ibogaine treatment (n = 3). Two of the 12 subjects in the favorable outcome group received additional treatment, compared to 7 of the 18 remaining subjects (Fisher’s Exact p = .249).
In the statistical analyses subjects in residential treatment were assumed to be missing and their missing ASIC score values set to pretreatment values, and these subjects were not included in the subset who reported no opioid use in the previous 30 days. Previous 30-day use of methadone or buprenorphine in the context of maintenance or from another source was regarded as opioid use and assessed using the ASI as with other subjects and time points.
Adverse events
No clinically significant cardiovascular or other medical events occurred in this study.
Discussion
This observational study reports on follow-up data subsequent to detoxification with ibogaine of 30 individuals with OUD. The data appear to indicate treatment effects in detoxification, and on the use of opioids and other substance use-related outcomes at 1 month, sustained to a variable extent over the subsequent posttreatment interval of up to 12 months.
The reduction in SOWS scores in this study is consistent with prior case series (Citation4,Citation5), and the status of opioid detoxification as the purpose for which ibogaine is most commonly used (Citation1). The clinical effect of ibogaine on opioid withdrawal symptoms appears of a comparable order of magnitude to that of methadone in the study by Handelsman et al. (Citation65), in which subjects withdrawing from opioids were administered the SOWS at pretreatment baseline and following two days of methadone stabilization. In that study, the SOWS decreased by a mean of 18.7 points (from 24.3 to 5.6) in subjects who used opioid exclusively, and 8.7 points (from 23.1 to 14.4) in subjects who additionally abused other, non-opioid substances. In the present study, the mean SOWS decrease following the administration of ibogaine was 17 points (from 31 to 14), without a significant effect related to the use of non-opioid substances.
The effect of ibogaine on drug use at 1 month appears substantive relative to reported outcomes following opioid detoxification. In this study 15 (50%) and 10 (33%) of subjects reported no opioid use during the previous 30 days at 1 and 3 months respectively (). By comparison, a large recent study reported an 8.6% rate of treatment success, defined as self-report of ≤4 days of opioid use in the previous 30 days, at 8 weeks subsequent to tapering and discontinuing buprenorphine with no subsequent pharmacotherapy (Citation68). Recent systematic reviews on follow-up of opioid detoxification without subsequent maintenance treatment report rates of abstaining from illicit opioid use of 18% at 4 weeks following detoxification with buprenorphine (Citation69), and 26% at 6 weeks following detoxification with methadone (Citation70).
Group statistics indicated Drug Use scores were improved relative to pretreatment baseline at all posttreatment time points, although the noninferiority tests suggest that this improvement was sustained over the subsequent 3 to 12 months at levels that did not reach equivalence to the effect observed at 1 month. Treatment responses in clinical populations occur in subsets of individuals, and not as a uniformly equal mean effect across all subjects. suggests treatment effects extending up to 12 months in a subset of individuals, evident as trajectories characterized by large Drug Use score decreases from baseline to 1 month that are sustained at subsequent time points.
The subset of 12 subjects with favorable outcomes averaged 2.0 ± 1.6 prior treatment episodes for OUD, indicating that ibogaine may have provided distinctive benefit for individuals with histories of previously unsuccessful treatment. There was a higher prevalence of a history of methadone maintenance and a greater number of prior treatments in the group with less favorable outcomes. It is not clear whether the effect of exposure to methadone on outcome was mediated by a general association of exposure to methadone as a correlate of a history of more treatment episodes, or a possible pharmacologically mediated effect of exposure to methadone per se, such as the development of relatively more severe physiological dependence.
Although no adverse cardiovascular events were observed in this study, ibogaine has been associated with fatalities, the most common proximal cause of which appears to be cardiac arrhythmia (Citation61,Citation71). Ibogaine has been associated with polymorphic ventricular tachycardia (PVT) including torsade de pointes (TdP), as well as bradycardia, which further potentiates the risk of PVT (Citation72). Both ibogaine and noribogaine cause blockade of the voltage-gated cardiac potassium channel encoded by the human ether-a-go-go-related gene (hERG) (Citation73), which is the most common cause of drug-induced TdP (Citation72). 18-MC, which shares with ibogaine the same ibogamine structural skeleton that defines the iboga class of monoterpene indole alkaloids, produces substantially less hERG blockade than ibogaine or noribogaine (Citation73), and has not produced bradycardia in the animal model (Citation74). This suggests that safer compounds can be designed utilizing systematic substitutions on the iboga alkaloid scaffold.
Qualitative perspective
Some themes are recurrent in comments made in interviews and email correspondence by study participants, who appear to have viewed the subjective experience with ibogaine as psychologically salient. Distinctive features of the subjective experience mediated by ibogaine include what has been termed “the slide show”, a panoramic, rapid readout of long-term visual memory, and equanimity regarding the material retrieved (Citation75,Citation76). Some practitioners who have used classical hallucinogens to assist psychotherapy have utilized ibogaine (Citation1,Citation77,Citation78). Family/Social was the most improved ASIC factor apart from Drug Use, in apparent consonance with the themes of “one heartedness”, and “binding” of the individual to family and ancestors in Bwiti, the African context of sacramental use of T. iboga (Citation76,Citation79). Lotsof has suggested that effects on drug use and interpersonal functioning at later intervals after taking ibogaine may correspond to processing and behavioral integration of a psychodynamically salient psychoactive experience (Citation11,Citation75).
One theme among study participants was the attribution of insight and meaning to the content of the psychoactive state produced by ibogaine. One subject wrote, “I saw my family from young to older and how everything has been and how I affected them.” and, “When I closed my eyes most of the time I had visions from my past… A profound sense of love for my family and their love for me and an intense, almost piercing agony as I was overwhelmed with the remorse and the waste and loss, feeling empathy with my family over all their hopes for me dashed by my relentless pursuit of drugs… I kept seeing clips – real memories, of high-school girlfriends and playing music with friends – but then also clips of the present day in an alternate reality where I hadn’t squandered so much love or compassion that had been offered to me.”
Another theme was the characterization of an interval of diminished posttreatment drug craving as a window of opportunity for personal change, evident in comments such as, “…you could safely say that iboga will give an opiate addict several months to a half a year of freedom from cravings and an expanded awareness. This gives the user a period of time in which to get his/her life together and learn to face things straightforwardly, directly and honestly. Iboga will not do the work for you. However, it will help you do your own work.”
Limitations of this study
This is a study on an unusual pharmacotherapy in a challenging setting for systematic controlled clinical investigation. There were a limited number of subjects and substantial attrition over time. This study lacked a control group and compared individuals to their baseline. This study relied on self-report without laboratory verification, with the ASIC Drug Use score as the primary measure of substance use outcome. Adequately powered randomized controlled clinical studies will be needed to develop ibogaine or its structural derivatives.
Laboratory verification of self-reports are a necessary feature in future clinical research to prevent biasing the data due to underreporting of drug use. Nonetheless, self-reporting in clinical research on SUDs is often accurate (Citation80), particularly when there are no negative consequences to the subject for reporting use. The study inclusion criterion requiring a corroborative source may have provided additional reliability.
The mean time interval between baseline and second SOWS for the entire sample was 76.5 hours, raising a potential concern that in this time frame even an inactive agent might yield falsely positive results on the basis of the eventual resolution of symptoms with unassisted opioid withdrawal. This view would take as evidence for an effect in detoxification the resolution of withdrawal symptomatology within the time window of expected peak symptomatic severity in unassisted opioid withdrawal. Published estimates of the time interval from last use to peak withdrawal symptom severity in unassisted withdrawal from heroin are commonly in a range with a lower bound of approximately 48 hours (Citation81–Citation83), and an upper bound at 72 hours (Citation84,Citation85), or even as long as four days (Citation86,Citation87). The expression of the opioid withdrawal syndrome may be prolonged with severe degrees of physiological dependence (Citation82), which may be relevant here in view of high dosages of heroin and oxycodone used by this study population ().
The subjects in this study had substantial rates of previous 30-day use of buprenorphine (23%) or methadone (30%) (), and 15 subjects (50%) had used at least one of these long-acting opioids in the previous 30 days prior to ibogaine treatment. Withdrawal associated with long-acting opioids evolves and persists over a more extended time interval compared to short acting opioids (Citation88–Citation90), which may also have increased the time interval required for detoxification in this sample. Subjects on long-acting opioids had been instructed to switch to short acting opioids prior to treatment, but likely varied significantly regarding their success in doing so.
Another factor that may have prolonged the treatment is the test-flood-booster opioid detoxification protocol, which utilizes the response to the test dose as a probe to estimate the severity of dependence (Citation62). Subjects in the present study were using amounts of heroin and methadone that are twice as large as those used by subjects in earlier published series of detoxification treatments in the US in the 1960s and the Netherlands in the late 1980s (Citation4,Citation91), and heroin of the present day is of substantially greater purity and lower cost (Citation92). However, the providers in this study utilized total dosages of ibogaine that are very similar to those used in prior treatments (Citation4), suggesting a dose ceiling. There is a relatively greater awareness of medical risk among contemporary treatment providers (Citation1,Citation61,Citation62) compared to the previous era of the initial ibogaine treatments (Citation4). The test-flood-booster ibogaine dosage approach utilized by the providers in this observational study appears to be an adaptation intended toward maximizing efficiency in the face of severe levels of physiological dependence, and contrasts with an earlier era in which opioid detoxification treatments with ibogaine proceeded more rapidly, with nearly all of the total ibogaine dosage administered at once (Citation4).
The qualitative descriptions and comments from the subjects in this study suggest tolerability as another line of clinical evidence for a treatment effect of ibogaine in detoxification. The null hypothesis that ibogaine is ineffective in alleviating withdrawal would equate its tolerability to that of unassisted withdrawal without any ancillary medications for two to five days in individuals with a one gram/day heroin or 200 mg/day oxycodone habit. In view of the lack of a significant placebo effect in opioid detoxification (Citation70,Citation93,Citation94), attributions regarding the effectiveness of ibogaine in the null scenario would be very low to say the least, which did not occur in the present study. Among subjects who spontaneously mentioned withdrawal symptoms when asked to provide descriptions of their ibogaine experience, ibogaine was consistently attributed with alleviating withdrawal.
Conclusion
Within the limitations of an uncontrolled, observational study ibogaine appeared to have a substantive treatment effect in opioid detoxification, and group statistics and individual trajectories appear to indicate an effect of reducing drug use at 1 month, which was sustained up to 12 months in a subgroup of subjects. Ibogaine appears to have had a clinical effect in some subjects with histories of failure of other treatments for OUD. The iboga alkaloid structural skeleton may be accurately regarded as a “privileged scaffold”, a structure of pharmacological significance on which systematic substitutions modulate therapeutic and toxic effects (Citation95,Citation96). With an unknown and likely novel mechanism of action, and a structure that evidently accommodates rational drug design, ibogaine may provide an interesting prototype for discovery and development of fundamentally innovative pharmacotherapy.
Declaration of interest
The authors report no conflicts of interest and have no financial interests to disclose.
Acknowledgments
The Multidisciplinary Association for Psychedelic Studies (MAPS) supported this study. We thank the following individuals for their effort in this project: Jeff Israel, Clare Wilkins, Peter Flom, Ph.D., Julie Denenberg, M.A., Kristin Gorenflo, I.M.F.T., Mayra A. Gomez, Valerie Mojeiko Sonstroem, and Meg Jordan, Ph.D., R.N.
References
- Alper KR, Lotsof HS, Kaplan CD. The ibogaine medical subculture. J Ethnopharmacol 2008;115:9–24.
- Brown TK. Ibogaine in the treatment of substance dependence. Curr Drug Abuse Rev 2013;6:3–16.
- Vastag B. Addiction research. Ibogaine therapy: a ‘vast, uncontrolled experiment’. Science 2005;308:345–346.
- Alper KR, Lotsof HS, Frenken GM, Luciano DJ, Bastiaans J. Treatment of acute opioid withdrawal with ibogaine. Am J Addict 1999;8:234–242.
- Mash DC, Kovera CA, Pablo J, Tyndale R, Ervin FR, Kamlet JD, Hearn WL. Ibogaine in the treatment of heroin withdrawal. Alkaloids Chem Biol 2001;56:155–171.
- Alper KR. Ibogaine: a review. Alkaloids Chem Biol 2001;56:1–38.
- Bastiaans E. Life after ibogaine: an exploratory study of the long-term effects of ibogaine treatment on drug addicts. Doctorandus thesis. Faculty of Medicine Vrije Universiteit Amsterdam 2004. URL https://www.iceers.org/docs/science/iboga/Bastiaans%20E_Life_After_Ibogaine.pdf (accessed April 11, 2017).
- Schenberg EE, De Castro Comis MA, Chaves BR, Da Silveira DX. Treating drug dependence with the aid of ibogaine: a retrospective study. J Psychopharmacol 2014;28:993–1000.
- Cloutier-Gill L, Wood E, Millar T, Ferris C, Eugenia Socias M. Remission of severe opioid Use disorder with ibogaine: a case report. J Psychoactive Drugs 2016;48:214–217.
- Sisko B. Interrupting drug dependency with ibogaine: a summary of four case histories. Multidisciplinary Assoc Psychedelic Stud (MAPS) Newslett 1993;4:15–24.
- Lotsof HS, Alexander NE. Case studies of ibogaine treatment: implications for patient management strategies. Alkaloids Chem Biol 2001;56:293–313.
- Dzoljic ED, Kaplan CD, Dzoljic MR. Effect of ibogaine on naloxone-precipitated withdrawal syndrome in chronic morphine-dependent rats. Arch Int Pharmacodyn Ther 1988;294:64–70.
- Cappendijk SL, Fekkes D, Dzoljic MR. The inhibitory effect of norharman on morphine withdrawal syndrome in rats: comparison with ibogaine. Behav Brain Res 1994;65:117–119.
- Glick SD, Rossman K, Rao NC, Maisonneuve IM, Carlson JN. Effects of ibogaine on acute signs of morphine-withdrawal in rats - independence from tremor. Neuropharmacology 1992;31:497–500.
- Parker LA, Burton P, McDonald RV, Kim JA, Siegel S. Ibogaine interferes with motivational and somatic effects of naloxone-precipitated withdrawal from acutely administered morphine. Prog Neuropsychopharmacol Biol Psychiatry 2002;26:293–297.
- Frances B, Gout R, Cros J, Zajac JM. Effects of ibogaine on naloxone-precipitated withdrawal in morphine-dependent mice. Fundam Clin Pharmacol 1992;6:327–332.
- Layer RT, Skolnick P, Bertha CM, Bandarage UK, Kuehne ME, Popik P. Structurally modified ibogaine analogs exhibit differing affinities for NMDA receptors. Eur J Pharmacol 1996;309:159–165.
- Leal MB, Michelin K, Souza DO, Elisabetsky E. Ibogaine attenuation of morphine withdrawal in mice: role of glutamate N-methyl-D-aspartate receptors. Prog Neuropsychopharmacol Biol Psychiatry 2003;27:781–785.
- Popik P, Layer RT, Fossom LH, Benveniste M, Geterdouglass B, Witkin JM, Skolnick P. NMDA antagonist properties of the putative antiaddictive drug, ibogaine. J Pharmacol Exp Ther 1995;275:753–760.
- Aceto MD, Bowman ER, Harris LS, May EL. Dependence studies of new compounds in the rhesus monkey and mouse (1991). NIDA Res Monogr 1992;119:513–558.
- Koja T, Fukuzaki K, Kamenosono T, Nishimura A, Nagata R, Lukas SE. Inhibition of opioid abstinent phenomena by Ibogaine. 69th annual meeting of the Japanese Pharmacological Society, March 20–23, 1996. Jpn J Pharmacol 1996;71:89.
- Belgers M, Leenaars M, Homberg JR, Ritskes-Hoitinga M, Schellekens AF, Hooijmans CR. Ibogaine and addiction in the animal model, a systematic review and meta-analysis. Transl Psychiatry 2016;6:e826.
- Glick SD, Rossman K, Steindorf S, Maisonneuve IM, Carlson JN. Effects and aftereffects of ibogaine on morphine self-administration in rats. Eur J Pharmacol 1991;195:341–345.
- Glick SD, Kuehne ME, Raucci J, Wilson TE, Larson D, Keller RW Jr., Carlson JN. Effects of iboga alkaloids on morphine and cocaine self-administration in rats: relationship to tremorigenic effects and to effects on dopamine release in nucleus accumbens and striatum. Brain Res 1994;657:14–22.
- Glick SD, Pearl SM, Cai J, Maisonneuve IM. Ibogaine-like effects of noribogaine in rats. Brain Res 1996;713:294–297.
- Dworkin SI, Gleeson S, Meloni D, Koves TR, Martin TJ. Effects of ibogaine on responding maintained by food, cocaine and heroin reinforcement in rats. Psychopharmacology (Berl) 1995;117:257–261.
- Maisonneuve IM, Glick SD. Interactions between ibogaine and cocaine in rats - invivo microdialysis and motor behavior. Eur J Pharmacol 1992;212:263–266.
- Cappendijk SL, Dzoljic MR. Inhibitory effects of ibogaine on cocaine self-administration in rats. Eur J Pharmacol 1993;241:261–265.
- Sershen H, Hashim A, Lajtha A. Ibogaine reduces preference for cocaine consumption in C57BL/6By mice. Pharmacol Biochem Behav 1994;47:13–19.
- Maisonneuve IM, Keller RW Jr., Glick SD. Interactions of ibogaine and D-amphetamine: in vivo microdialysis and motor behavior in rats. Brain Res 1992;579:87–92.
- Rezvani AH, Overstreet DH, Lee YW. Attenuation of alcohol intake by ibogaine in three strains of alcohol-preferring rats. Pharmacol Biochem Behav 1995;52:615–620.
- Maisonneuve IM, Keller RW Jr., Glick SD. Interactions between ibogaine, a potential anti-addictive agent, and morphine: an in vivo microdialysis study. Eur J Pharmacol 1991;199:35–42.
- Maisonneuve IM, Glick SD. Attenuation of the reinforcing efficacy of morphine by 18-methoxycoronaridine. Eur J Pharmacol 1999;383:15–21.
- Maisonneuve IM, Mann GL, Deibel CR, Glick SD. Ibogaine and the dopaminergic response to nicotine. Psychopharmacology (Berl) 1997;129:249–256.
- Glick SD, Maisonneuve IM, Visker KE, Fritz KA, Bandarage UK, Kuehne ME. 18-Methoxycoronardine attenuates nicotine-induced dopamine release and nicotine preferences in rats. Psychopharmacology (Berl) 1998;139:274–280.
- Antonio T, Childers SR, Rothman RB, Dersch CM, King C, Kuehne M, Bornmann WG, et al. Effect of iboga alkaloids on μ-opioid receptor-coupled G protein activation. PlosOne 2013;8:e77262.
- Glick SD, Maisonneuve IM, Szumlinski KK. Mechanisms of action of ibogaine: relevance to putative therapeutic effects and development of a safer iboga alkaloid congener. Alkaloids Chem Biol 2001;56:39–53.
- Schneider JA, McArthur M. Potentiation action of ibogaine (bogadin TM) on morphine analgesia. Experientia 1956;12:323–324.
- Schneider JA. Assigned to Ciba Pharmaceutical Products Inc. Summit NJ. Tabernanthine, Ibogaine Containing Analgesic Compostions. US Patent no. 2,817,623. 1957.
- Bagal AA, Hough LB, Nalwalk JW, Glick SD. Modulation of morphine-induced antinociception by ibogaine and noribogaine. Brain Res 1996;741:258–262.
- Cao YJ, Bhargava HN. Effects of ibogaine on the development of tolerance to antinociceptive action of mu-, delta- and kappa-opioid receptor agonists in mice. Brain Res 1997;752:250–254.
- Bhargava HN, Cao YJ, Zhao GM. Effects of ibogaine and noribogaine on the antinociceptive action of mu-, delta- and kappa-opioid receptor agonists in mice. Brain Res 1997;752:234–238.
- Sunder Sharma S, Bhargava HN. Enhancement of morphine antinociception by ibogaine and noribogaine in morphine-tolerant mice. Pharmacology 1998;57:229–232.
- Pearl SM, Johnson DW, Glick SD. Prior morphine exposure enhances ibogaine antagonism of morphine-induced locomotor stimulation. Psychopharmacology (Berl) 1995;121:470–475.
- Skolnick P. Ibogaine as a glutamate antagonist: relevance to its putative antiaddictive properties. Alkaloids Chem Biol 2001;56:55–62.
- Trujillo KA, Akil H. Inhibition of opiate tolerance by non-competitive N-methyl-D-aspartate receptor antagonists. Brain Res 1994;633:178–188.
- Bisaga A, Comer S, Ward A, Popik P, Kleber H, Fischman M. The NMDA antagonist memantine attenuates the expression of opioid physical dependence in humans. Psychopharmacology (Berl) 2001;157:1–10.
- Panchal V, Taraschenko OD, Maisonneuve IM, Glick SD. Attenuation of morphine withdrawal signs by intracerebral administration of 18-methoxycoronaridine. Eur J Pharmacol 2005;525:98–104.
- Rho B, Glick SD. Effects of 18-methoxycoronaridine on acute signs of morphine withdrawal in rats. Neuroreport 1998;9:1283–1285.
- Deecher DC, Teitler M, Soderlund DM, Bornmann WG, Kuehne ME, Glick SD. Mechanisms of action of ibogaine and harmaline congeners based on radioligand binding studies. Brain Res 1992;571:242–247.
- Sweetnam PM, Lancaster J, Snowman A, Collins JL, Perschke S, Bauer C, Ferkany J. Receptor binding profile suggests multiple mechanisms of action are responsible for ibogaine’s putative anti-addictive activity. Psychopharmacology (Berl) 1995;118:369–376.
- MacInnes N, Handley SL. Characterization of the discriminable stimulus produced by 2-BFI: effects of imidazoline I(2)-site ligands, MAOIs, beta-carbolines, agmatine and ibogaine. Br J Pharmacol 2002;135:1227–1234.
- He DY, McGough NNH, Ravindranathan A, Jeanblanc J, Logrip ML, Phamluong K, Janak PH, Ron D. Glial cell line-derived neurotrophic factor mediates the desirable actions of the anti-addiction drug ibogaine against alcohol consumption. J Neurosci 2005;25:619–628.
- Carnicella S, He DY, Yowell QV, Glick SD, Noribogaine RD. But not 18-MC, exhibits similar actions as ibogaine on GDNF expression and ethanol self-administration. Addict Biol 2010;15:424–433.
- Glick SD, Maisonneuve IM, Kitchen BA, Fleck MW. Antagonism of α3β4 nicotinic receptors as a strategy to reduce opioid and stimulant self-administration. Eur J Pharmacol 2002;438:99–105.
- Pearl SM, Hough LB, Boyd DL, Glick SD. Sex differences in ibogaine antagonism of morphine-induced locomotor activity and in ibogaine brain levels and metabolism. Pharmacol Biochem Behav 1997;57:809–815.
- Baumann MH, Rothman RB, Pablo JP, Mash DC. In vivo neurobiological effects of ibogaine and its O-desmethyl metabolite, 12-hydroxyibogamine (noribogaine), in rats. J Pharmacol Exp Ther 2001;297:531–539.
- Glue P, Lockhart M, Lam F, Hung N, Hung CT, Friedhoff L. Ascending-dose study of noribogaine in healthy volunteers: pharmacokinetics, pharmacodynamics, safety, and tolerability. J Clin Pharmacol 2015;55:189–194.
- Mash DC, Ameer B, Prou D, Howes JF, Maillet EL. Oral noribogaine shows high brain uptake and anti-withdrawal effects not associated with place preference in rodents. J Psychopharmacol 2016;30:688–697.
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders, text revision (DSM-IV-TR); Washington. DC: American Psychiatric Association; 2000.
- Alper KR, Stajic M, Gill JR. Fatalities temporally associated with the ingestion of ibogaine. J Forensic Sci 2012;57:398–412.
- Dickinson J, McAlpin J, Wilkins C, Fitzsimmons C, Guion P, Paterson T, Greene D, Chaves BR. Clinical guidelines for ibogaine-assisted detoxification, 1st edn., Version 1.1. Montreal, Quebec, Canada: Global Ibogaine Therapy Alliance (GITA); 2016. https://www.ibogainealliance.org/guidelines [accessed April 11, 2017].
- Cacciola JS, Alterman AI, McLellan AT, Lin Y-T, Lynch KG. Initial evidence for the reliability and validity of a “Lite” version of the Addiction Severity Index. Drug Alcohol Depend 2007;87:297–302.
- Mcgahan PL, Griffith JA, Parente R, McLellan AT. Addiction severity index composite scores manual. Philadelphia, PA: The University of Pennsylvania/Veterans Administration Center for Studies of Addiction; 1986.
- Handelsman L, Cochrane KJ, Aronson MJ, Ness R, Rubinstein KJ, Kanof PD. Two new rating-scales for opiate withdrawal. Am. J. Drug Alcohol Abuse 1987;13:293–308.
- Castelloe J. Equivalence WD and noninferiority testing using SAS/STAT® software. SAS Paper #1911-2015 2015, 1–23.
- Kaul S, Diamond GA. Good enough: a primer on the analysis and interpretation of noninferiority trials. Ann Intern Med 2006;145:62–69.
- Weiss RD, Potter JS, Fiellin DA, Byrne M, Connery HS, Dickinson W, Gardin J, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry 2011;68:1238–1246.
- Bentzley BS, Barth KS, Back SE, Book SW. Discontinuation of buprenorphine maintenance therapy: perspectives and outcomes. J Subst Abuse Treat 2015;52:48–57.
- Amato L, Davoli M, Minozzi S, Ferroni E, Ali R, Ferri M. Methadone at tapered doses for the management of opioid withdrawal. Cochrane Database Syst Rev 2013;2:CD003409.
- Koenig X, Hilber K. The anti-addiction drug ibogaine and the heart: a delicate relation. Molecules 2015;20:2208–2228.
- Kannankeril P, Roden DM, Darbar D. Drug-induced long QT syndrome. Pharmacol Rev 2010;62:760–781.
- Alper K, Bai R, Liu N, Fowler SJ, Huang XP, Priori SG, Ruan Y. hERG blockade by iboga alkaloids. Cardiovasc Toxicol 2016;16:14–22.
- Glick SD, Maisonneuve IM, Hough LB, Kuehne ME, Bandarage UK. (±)-18-Methoxycoronaridine: a novel iboga alkaloid congener having potential anti-addictive efficacy. CNS Drug Rev 1999;5:27–42.
- Alper KR, Lotsof HS. The use of ibogaine in the treatment of addictions. In: Winkelman M, Roberts T, eds. Psychedelic Medicine, Vol. 2. Westport, CT: Praeger/Greenwood Publishing Group; 2007:43–66.
- Fernandez JW. Bwiti: an ethnography of religious imagination in Africa; Princeton, New Jersey: Princeton University Press; 1982.
- Stolaroff M. The secret chief revealed. Sarasota, FL: Multidisciplinary Association for Psychedelic Studies (MAPS); 2004.
- Naranjo C. The healing journey: new approaches to consciousness. New York: Pantheon, Random House; 1973.
- Fernandez JW, Fernandez RL. “Returning to the path”: the use of iboga[ine] in an equatorial African ritual context and the binding of time, space, and social relationships. Alkaloids Chem Biol 2001;56:235–247.
- Darke S. Self-report among injecting drug users: a review. Drug Alcohol Depend 1998;51:253–263.
- Yu E, Miotto K, Akerele E, Montgomery A, Elkashef A, Walsh R, Montoya I, et al. A Phase. 3 placebo-controlled, double-blind, multi-site trial of the alpha-2-adrenergic agonist, lofexidine, for opioid withdrawal. Drug Alcohol Depend 2008;97:158–168.
- Smolka M, Schmidt LG. The influence of heroin dose and route of administration on the severity of the opiate withdrawal syndrome. Addiction 1999;94:1191–1198.
- Tompkins DA, Smith MT, Mintzer MZ, Campbell CM, Strain EC. A double blind, within subject comparison of spontaneous opioid withdrawal from buprenorphine versus morphine. J Pharmacol Exp Ther 2014;348:217–226.
- Farrell M. Opiate withdrawal. Addiction 1994;89:1471–1475.
- Gerra G, Marcato A, Caccavari R, Fontanesi B, Delsignore R, Fertonani G, Avanzini P, et al. Clonidine and opiate receptor antagonists in the treatment of heroin addiction. J Subst Abuse Treat 1995;12:35–41.
- Loimer N, Linzmayer L, Grunberger J. Comparison between observer assessment and self rating of withdrawal distress during opiate detoxification. Drug Alcohol Depend 1991;28:265–268.
- Gossop M. The detoxification of high-dose heroin-addicts in Pakistan. Drug Alcohol Depend 1989;24:143–150.
- Gossop M, Griffiths P, Bradley B, Strang J. Opiate withdrawal symptoms in response to 10-day and 21-day methadone withdrawal programs. Br J Psychiatry 1989;154:360–363.
- Fudala PJ, Jaffe JH, Dax EM, Johnson RE. Use of buprenorphine in the treatment of opioid addiction. 2. Physiologic and behavioral-effects of daily and alternate-day administration and abrupt withdrawal. Clin Pharmacol Ther 1990;47:525–534.
- San L, Cami J, Fernandez T, Olle JM, Peri JM, Torrens M. Assessment and management of opioid withdrawal symptoms in buprenorphine-dependent subjects. Br J Addict 1992;87:55–62.
- Alper KR, Beal D, Kaplan CD. A contemporary history of ibogaine in the United States and Europe. Alkaloids Chem Biol 2001;56:249–281.
- Drug Enforcement Administration (DEA). National Heroin Threat Assessment Summary - Updated. DEA-DCT-DIR-031-16; 2016
- Gowing L, Ali R, White JM, Mbewe D. Buprenorphine for managing opioid withdrawal. Cochrane Database Syst Rev 2017; 2:Cd002025. doi:10.1002/14651858.CD002025.pub5
- Gowing L, Farrell M, Ali R, White JM. Alpha(2)-adrenergic agonists for the management of opioid withdrawal. Cochrane Database Syst Rev 2016;5: CD002024. doi:10.1002/14651858.CD002024.pub5
- Welsch ME, Snyder SA, Stockwell BR. Privileged scaffolds for library design and drug discovery. Curr. Opin. Chem. Biol. 2010;14:347–361.
- Szychowski J, Truchon JF, Bennani YL. Natural products in medicine: transformational outcome of synthetic chemistry. J. Med. Chem. 2014;57:9292–9308.
