ABSTRACT
Research regarding the effects of high-intensity interval training (HIIT) on executive function has grown exponentially in recent years. However, there has been no comprehensive review of the current state of literature. Therefore, the aim of this systematic review is to summarize previous research regarding the acute and chronic effects of HIIT on executive function across the lifespan and highlight future research directions. The results indicated that acute bouts of HIIT has a positive effect on inhibition in children/adolescents and adults, and further that chronic HIIT benefits inhibition and working memory in children. More research employing chronic interventions, focusing on middle-aged and older adults, and examining the effects on the working memory and cognitive flexibility domains of executive function are needed. Future research should also focus on a) the use of stronger research designs, b) the effects of HIIT dosage/modality, c) consideration of individual differences, d) possible underlying mechanisms, and e) examining the feasibility of translating HIIT to real-word settings.
1. Introduction
A growing body of evidence indicates a positive association of physical activity with cognition and brain function across the lifespan (Hillman et al., Citation2008; Kao et al., Citation2020). The current literature supports the positive influence of acute and chronic moderate-intensity, continuous aerobic exercise (Chang et al., Citation2012; Guiney & Machado, Citation2013; Kamijo et al., Citation2009; Li et al., Citation2017; McMorris et al., Citation2011) and resistance exercise (De Asteasu et al., Citation2017; Soga et al., Citation2018; Wilke et al., Citation2019) on cognitive function across different age groups. In addition, recent findings have bolstered the cognitive benefits associated with high-intensity interval training (HIIT) (Cooper, Dring et al., Citation2016).
HIIT refers to physical activity characterized by relatively brief bursts of vigorous activity (i.e., ~90% of maximal aerobic power for brief intervals), interspersed by short periods of rest or low-intensity physical activity for recovery (Eddolls et al., Citation2017). It has been suggested that HIIT, typically studied using treadmill running or cycling on an ergometer (Eddolls et al., Citation2017), may be an attractive alternative to traditional moderate-intensity continuous exercise or resistance exercise as a means of promoting various health outcomes, such as aerobic capacity (Lee, Hsu et al., Citation2018), lipid metabolism (Lee, Kuo et al., Citation2018), vascular function (O’Brien et al., Citation2020), and balance (Jiménez-García et al., Citation2019). Although HIIT may be associated with higher risk of injury (Rynecki et al., Citation2019), its health-related benefits can be manifested once implemented appropriately. Moreover, the time-efficient nature of HIIT has made it a desirable option in overcoming “lack of time” – one of the most cited barriers to engaging in physical activity (Centers for Disease Control and Prevention, Citation2012).
Despite emerging evidence supporting the efficacy and feasibility of HIIT for improving cognitive and brain health, to date there is only one narrative review included only three studies (Cooper, Bandelow et al., Citation2016). The review did not provide explicit information on either the nature of the HIIT protocols or details of the cognitive assessments used in the studies (Cooper, Bandelow et al., Citation2016), and only focused on young adults (Cooper, Bandelow et al., Citation2016). Considering the rapid growth of this line of research, it is necessary to provide an updated review that summarizes the existing literature in a more comprehensive manner.
The aim of this paper is to systematically summarize the current state of the literature regarding the effects of HIIT on executive function, the most commonly studied aspect of cognition. Given that the literature has indicated cognitive benefits stemming from physical activity across the lifespan (De Asteasu et al., Citation2017; Chang et al., Citation2012; Guiney & Machado, Citation2013; Hillman et al., Citation2008; Kamijo et al., Citation2009; Kao et al., Citation2020; Li et al., Citation2017; McMorris et al., Citation2011; Soga et al., Citation2018; Wilke et al., Citation2019), we categorized studies into children/adolescents, young adults, and older adults. Moreover, though restrictive, we exclusively focused on the effects of HIIT on executive function, a subdomain of cognition involving top-down, multifaceted process underlying goal-oriented behaviour (Diamond, Citation2013). Our emphasis was motivated by two reasons. First, executive function is a central component of cognition known to influence many cognitive processes, which are ecologically relevant to various domains of daily living, including academic achievement (Wang et al., Citation2015; Wang & Gathercole, Citation2013, Citation2015), vocational performance (Bailey, Citation2007), and quality of life (Davis et al., Citation2010). Second, there is a robust conceptual link between physical activity and executive function across the lifespan (Hillman et al., Citation2008; Kao et al., Citation2020), and extension from traditional aerobic/resistance exercise to HIIT is theoretical-driven. For example, acute bouts of HIIT may facilitate executive function via increased levels of circulating lactate, a biomarker that is essential for neural metabolism and neuronal excitability in the brain (Magistretti & Allaman, Citation2018). Other studies indicate that acute HIIT may benefit executive function via a more efficient neuroelectric activation and faster stimulus processing (Kao et al., Citation2017, Citation2018) or higher levels of cerebral oxygenation in the prefrontal cortex (Lambrick et al., Citation2016). Moreover, chronic HIIT may facilitate executive function via decrease in inflammation-related cytokines (Freitas et al., Citation2018), increased expression of BDNF (Freitas et al., Citation2018), and increased uptake of glucose (Robinson et al., Citation2018). Collectively, our goal is that the current review may serve as a reference which guides future research understanding the effectiveness of acute and chronic HIIT on executive function.
2. Methods
2.1. Search strategy
The current review followed the Preferred Reporting Items for Systematic Reviews and Meta‐Analysis (Moher, Liberati, Tetzlaff, Altman, & The PRISMA Group, Moher et al., Citation2009) for literature search and selection. The literature search was performed using three electronic databases, PubMed, Web of Science, and Scopus. While no start date was specified, the search was completed on February 2020 and therefore only included studies published as of that time. The search terms “high-intensity exercise”, “intermittent exercise”,’ interval exercise’, and “sprint” were grouped using the connector “OR” and then were combined using the connector “AND” with search terms “cognition”, “cognitive function”, “cognitive performance”, and “executive function”, resulting in the following search query: ((“high-intensity exercise” OR “intermittent exercise” OR “interval exercise” OR “sprint”) AND (“cognition” OR “cognitive function” OR “cognitive performance” OR “executive function”)).
2.2. Eligibility criteria
Articles were considered eligible if they met all of the following criteria: a) the research involved an intervention study, either acute (single bouts of HIIT) or chronic (repeated bouts of HIIT over days, weeks, or months), where details were given about the nature of the HIIT studied (e.g., the type of movements involved, intensity, number of sessions, duration of each session, duration of recovery between sessions, work-to-recovery ratio (WRR)); b) the reported high intensities were either >80% heart rate reserve (HRreserve), >85% heart rate maximal (HRmax), >90% maximal oxygen consumption (VO2max) or peak power output (Eddolls et al., Citation2017); c) the research focused on different subcomponents of executive function based on the definitions from Diamond (Citation2013): inhibition (i.e., the ability to selectively focus on task-relevant information while resisting attention to a prepotent but undesirable response), working memory (i.e., the ability to hold information in mind and mentally work with it or with information no longer perceptually presented), and cognitive flexibility (i.e., the ability to flexibly change mental representations back and forth between two task demands); d) outcome measures involved executive function-related tasks with explicitly defined performance indices (e.g., response accuracy, response times, items recalled), and executive function-related tasks were identified by referring to the studies from Diamond (Citation2013) and Etnier and Chang (Citation2009); e) participants were healthy individuals without diagnosed disease or disorders; and e) the research was published in English.
2.3. Study selection
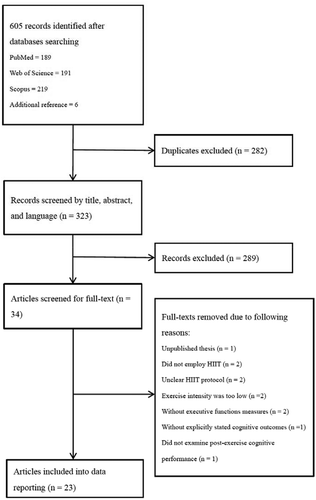
The screening and selection of studies were completed by two of the authors (SSH and TYC) on February 2020. First, titles and abstracts were examined to identify studies that met inclusion criteria after removal of duplicates. Second, the full text of eligible studies based on the screened studies was read to determine their final inclusion. Disagreement between the two reviewers was resolved by a consensus meeting with experts in the field (YKC and TMH) on February 2020. Finally, articles including acute and chronic HIIT interventions and executive function-related tasks were reviewed. provides an overview of the selection process.
2.4. Assessment of risk of bias
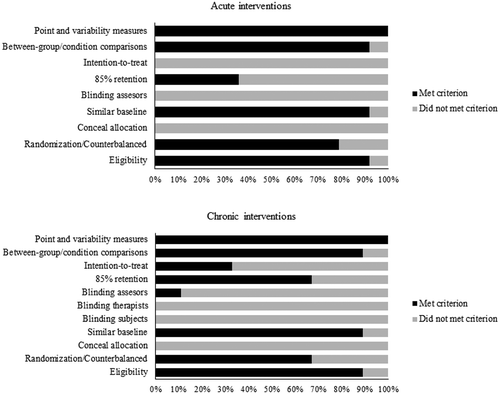
Two authors (SSH and TYC) assessed the study quality according to the PEDro scale (Maher et al., Citation2008). Both reviewers had undergone the PEDro training programme. Any disagreements were discussed with a third reviewer (TMH) until a consensus was achieved. It should be noted that the original version of the PEDro scale was modified to better fulfill the purpose of this systematic review. That is, blinding of participants and researchers were not considered for quality assessment for studies with acute exercise settings (Ludyga et al., Citation2016). These criteria were disregarded because true blinding could not be accomplished in studies with acute exercise settings. The summary of the quality assessment was presented in and Supplement 1.
2.5. Data extraction process
The final studies were examined thoroughly, and the following data were extracted: (1) first authors’ name, publication year, country of data collection; (2) sample size, participants’ age; (3) study design and/or group assignment; (4) details about HIIT intervention; (5) subcomponents of executive function assessed and its assessment paradigm; and (6) main outcomes.
3. Results
3.1. Study selection
A total of 605 studies were initially identified from PubMed, Web of Science, and Scopus. Of these, 282 duplicates were removed. Accordingly, a total of 323 articles were screened for title and abstract. After the first stage of screening, 34 articles were selected for full-text screening, and resulted in a total of 23 full-text articles included in the data extraction and reporting. The flow diagram of the study selection process following PRISMA guidelines is shown in . Also see and for summary of study characteristics and results (all studies in and are numbered for better referencing in the results section).
Table 1. Summary table of studies on children/adolescents
Table 2. Summary table of studies on adults
3.2. Description of studies reviewed
The literature search yielded a total of 23 studies published between February 2014 and January 2020. Studies took place in Europe (Ref 1, 2, 3, 7, 8, 10, 13, 14, 23), North America (Ref 9, 12, 15, 18, 20), South America (Ref 17), Asia (Ref 4, 11, 16), Oceania (Ref 5, 6, 19, 21), and South Africa (Ref 22). These investigations included sample sizes from 6 to 64 participants, ranging in age between 9 and 16 years for children and adolescents (26%, n = 6) (Ref 1–6), and between 19 and >70 years for adults (74%, n = 17) (Ref 7–23).
With regards to study design, 26% (n = 6) of studies examined chronic effects of HIIT using a randomized controlled trial (RCT) design (Ref 5, 6, 19, 21–23), while 13% (n = 3) of studies investigated the chronic effects of HIIT with either a non-randomized design (Ref 4, 18) or a within-group, pre-test-post-test design (Ref 20). Alternatively, 43% (n = 10) of studies investigated the acute effects of HIIT using a within-subjects, crossover design (Ref 2, 3, 9–12, 13, 15–17), and 17% (n = 4) of studies investigated the acute effects of HIIT with either a between-subjects design (Ref 1, 7, 8) or a within-subjects, pre-test-post-test design (Ref 14).
Regarding the modality of HIIT, 78% (n = 18) of studies (Ref 2, 3, 5, 7–13, 15–18, 20–23) employed traditional HIIT, in terms of cycling or running/sprinting, whereas the other 22% (n = 5) (Ref 1, 4, 6, 14, 19) adopted high-intensity functional circuit training (HICT) that consisted of functional, multi-joint movements via both aerobic and muscle-strengthening exercises (Eddolls et al., Citation2017).
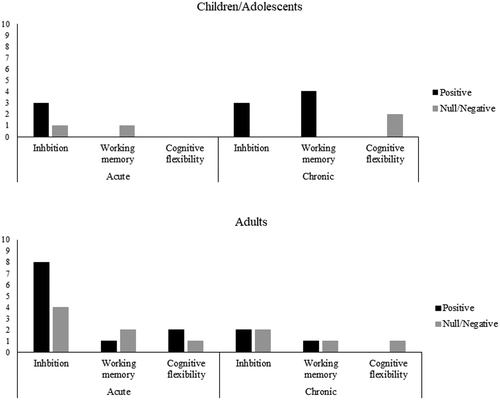
Relative to the different subcomponents of executive function, 70% (n = 16) of studies focused on inhibition (Ref 1–3, 5, 7, 8, 11–18, 21, 22), 35% (n = 8) of studies tapped working memory (Ref 2, 4, 5, 14, 17, 20, 22, 23), and 22% (n = 5) of studies targeted cognitive flexibility (Ref 6–8, 10, 19). demonstrates paradigms employed to assess different subcomponents of executive function. In the following sections, findings from the literature are summarized by age category.
Table 3. Cognitive paradigms used to examine executive functions
3.3. HIIT in children and adolescents
3.3.1. Acute interventions
Of the 23 studies reviewed, 13% (n = 3) focused on the acute effects of HIIT on executive function in children and adolescents (Ref 1–3) (see for details of studies), yielding a total of 5 intervention effects (). All three studies demonstrated a positive effect of acute HIIT on inhibition. Specifically, two studies found positive effects of acute HIIT on inhibition (Ref 2, 3), with the effect sustained for ~30 minutes following exercise cessation (Ref 3). Likewise, one study found positive effect of acute HICT on inhibition, with the beneficial effect sustained for ~60 minutes (Ref 1). Notably, data from one study revealed that HICT with a work-to-recovery ratio (WRR) of 1:1 (i.e., 30-second exercise: 30-second recovery) resulted in improved inhibition performance whereas HICT with a WRR of 2:1 (i.e., 60-second exercise: 30-second recovery) resulted in non-significant effect (Ref 1). In the case of other subcomponents of executive function, one study found a null effect of acute HIIT on working memory in children (Ref 2).
3.3.2. Chronic interventions
Among the 23 studies retrieved, 13% (n = 3) involved chronic HIIT interventions in children and adolescents (Ref 4–6) (see for details) and yielded a total of 9 intervention effects (). One study found improved performance for inhibition and working memory measured via six different executive function tasks (i.e., go/no-go task, flanker task, Stroop test, N-back, digit span backward test, Corsi block tapping test) following a HIIT program in children (Ref 5). Another study found positive effect of HICT on working memory in children (Ref 4). As for cognitive flexibility, one study found null effect from both an aerobic-based HICT and an aerobic + muscular-based HICT after training in adolescents (Ref 6). However, cognitive gains following a HIIT program were moderated by the genotype of brain-derived neurotrophic factor (BDNF), with BDNFmet66 carriers demonstrating larger cognitive gains relative to non-carriers following training (Ref 5).
3.4. HIIT in adults
3.4.1. Acute interventions
Of the 23 studies retrieved, 43% (n = 10) (Ref 7–9, 11–17) investigated the acute effects of HIIT on inhibition in adults (see for details), with a total of 18 intervention effects (). Of these 10 studies, seven of them supported the positive effect of acute HIIT in young (Ref 7, 9, 11, 12, 15, 16) and middle-aged adults (Ref 17), whereas three others found no influence of acute HIIT in young (Ref 8, 13, 14) and middle-aged adults (Ref 14). For working memory, studies found positive effect of a single bout of HIIT in young adults (Ref 14) but not in middle-aged adults (Ref 14, 17). In terms of cognitive flexibility, two studies support the positive effect of an acute bout of HIIT (Ref 7, 10) whereas one study found no benefit (Ref 8).
3.4.2. Chronic interventions
Of the 23 studies retrieved, 26% (n = 6) implemented chronic HIIT with young (Ref 19–21), middle-aged (Ref 23), and older adults (Ref 18, 22) ( for details), generating a total of 7 intervention effects (). Studies reported the positive effect of HIIT on inhibition in young adults (Ref 21) but not in older adults >60 years of age (Ref 18, 22). Alternatively, three studies found that whereas HIIT benefited working memory in older adults (Ref 23), there was no beneficial effect on young adults’ working memory (Ref 20) or cognitive flexibility (Ref 19). However, one study found young adults with higher compliance to the HIIT protocol (peak HR during exercise: 184.2 beats.min−1) had larger improvement in inhibition performance following training relative to those with lower compliance (170.5 beats.min−1) (Ref 21).
4. Discussion
4.1. Summary of the search results
The current systematic review summarizes evidence from studies examining the acute and chronic effects of HIIT as they relate to different subcomponents of executive function. Most studies included in this review were deemed fair quality based on criteria established in the PEDro scale (Maher et al., Citation2008) ( and Supplement 1). Overall, the current state of the literature favours a positive effect of acute HIIT on inhibition in children/adolescents and adults as well as a beneficial effect of chronic HIIT on inhibition and working memory in children.
4.2. Effects of HIIT on children and adolescents
The findings on children and adolescents support a positive effect of acute HIIT on inhibition, with the benefits sustain 30 to 60 minutes following intervention (Lambrick et al., Citation2016; Ludyga et al., Citation2018), whereas it remains unclear if acute bouts of HIIT benefit working memory and cognitive flexibility performance. It is noteworthy that WRR of HIIT may moderate the relation between acute HIIT and inhibition, with a smaller WRR (e.g., 30 second: 30 second) having a positive effect whereas a larger WRR (e.g., 60 second: 30 second) may not (Ludyga et al., Citation2018). To date, there is no study investigating the moderating role of WRR of HIIT in other subcomponents of executive function (i.e., working memory, cognitive flexibility) or research that modulates the WRR to determine why this factor may moderate the benefits to cognitive outcomes.
In terms of the chronic effects of HIIT, the current evidence suggests that both sprint-based and HICT-based interventions may benefit inhibition and working memory in children (Moreau et al., Citation2017; Tottori et al., Citation2019). Moreover, it is possible that children’s BDNF profile moderates the effectiveness of HIIT on inhibition and working memory, with BDNFmet66 carriers benefiting more from HIIT relative to non-carriers (Moreau et al., Citation2017). This is probably because BDNFmet66 carriers have higher post-exercise BDNF secretions (Nascimento et al., Citation2015), which may, in turn, result in stronger neuronal functioning, plasticity, and long-term potentiation (Lipsky & Marini, Citation2007; Zhou et al., Citation2011). In contrast, a chronic HIIT program, regardless of the protocol employed, did not benefit cognitive flexibility performance in adolescents (Costigan et al., Citation2016). However, given there has only been one study delineating this aspect of executive function, more research is needed to verify the findings from Costigan et al. (Citation2016).
Overall, the current evidence relating HIIT and executive function in children and adolescents corroborates the beneficial effects of acute and chronic continuous aerobic exercise (see Kao et al., Citation2020 for review). Of note, considering that the natural physical activity patterns of children and adolescents are similar to HIIT as well as the time-efficient nature of HIIT allows it to be implemented during shorter periods of time throughout the school day, HIIT-based physical activity may be a viable option in school settings to foster brain health (Lopes et al., Citation2006).
4.3. Effects of HIIT on adults
In adults, the majority of the data (7 of 11 studies reviewed; Alves et al., Citation2014; Hashimoto et al., Citation2018; Kao et al., Citation2017, Citation2018; Kujach et al., Citation2020; Miller et al., Citation2018; Tsukamoto et al., Citation2016) supported a positive effect of acute HIIT on inhibition. Findings from three studies (Gmiat et al., Citation2017; Ligeza et al., Citation2018; Schwarck et al., Citation2019) who found null effect of HIIT on inhibition may be confounded by small sample sizes (Gmiat et al., Citation2017), inappropriate timing of cognitive assessment (Gmiat et al., Citation2017), prolonged duration of HIIT (Ligeza et al., Citation2018; Schwarck et al., Citation2019), and/or sexual dimorphism of participants (Ligeza et al., Citation2018). Specifically, the sample size of the Gmiat et al. study was relatively small (i.e., 8 young women and 6 middle-aged women), which may have increased the risks of type 2 error. Furthermore, the one-hour interval between exercise cessation and administration of post-intervention inhibition test in the Gmiat et al. study was much longer than other studies assessing inhibition (typically ≤30 minutes following exercise cessation; Hashimoto et al., Citation2018; Kao et al., Citation2017, Citation2018; Tsukamoto et al., Citation2016), and this raises the possibility that cognitive benefits from exercise may have diminished at the time of cognitive assessment. On the other hand, compared with studies that found improved inhibition following single bouts of HIIT (Kao et al., Citation2017, Citation2018), exercise duration in the Ligeza et al. and Schwarck et al. studies are considerably longer (i.e., >25 minutes). A prolonged duration of HIIT may result in overall fatigue, decreased cerebral oxygenation, and/or decompensated levels of blood lactate, which may, in turn, reduce the facilitative effects of HIIT. Another discrepancy that Ligeza et al. and Schwarck et al. studies hold against other findings (Kao et al., Citation2017, Citation2018) is related to participants’ sexual dimorphism. That is, while the former studies only recruited male participants, the latter studies recruited both men and women. It is plausible that sexual dimorphism may play a moderating role on the magnitude of the effect, as a meta-analysis by Chang et al. (Citation2012) summarizing the effects of acute exercise on cognition indicated that studies with both men and women had larger effect sizes than those studies with either men or women. This moderating role of sexual dimorphism might be accounted for by the fact that men and women have differential secretion of hormones (e.g., dehydroepiandrosterone) relating to arousal (De Menezes et al., Citation2016) and metabolic responses (Ponjee et al., Citation1994) following acute exercise, with women showing greater acute exercise-induced secretion (Heaney et al., Citation2013). Overall, the current evidence supporting the beneficial effect of acute HIIT on inhibition in adults aligns with the effects of single bouts of continuous aerobic exercise (Chang et al., Citation2012; Hsieh et al., Citation2018; Kao et al., Citation2020; Ludyga et al., Citation2016) and resistance exercise (Chang et al., Citation2012; Wang et al., Citation2019), suggesting HIIT may be an alternate means to benefit inhibition during adulthood.
By contrast, there is only a piecemeal of study relating to working memory and cognitive flexibility. While one study favours the positive influence of acute HIIT on working memory in young adults (Gmiat et al., Citation2017), there are two studies found null (Alves et al., Citation2014) and negative effect in middle-aged adults (Gmiat et al., Citation2017). However, considering the high heterogeneity between studies in participants’ age (i.e., young adults vs. middle-aged adults), study design (i.e., within-subjects, crossover design vs. within-subjects, pre-test-post-test design), cognitive tasks selected (spatial working memory task vs. verbal working memory task), and/or HIIT protocols (i.e., HICT vs. HIIT), more research is required to clarify if these methodological differences account for the discrepant findings. Likewise, the current findings relating to cognitive flexibility are also controversial, with two studies reporting positive effects in young adults (Dupuy et al., Citation2018; Kujach et al., Citation2020) and one study indicating no effect (Schwarck et al., Citation2019). However, again, these studies differed in terms of study design, HIIT protocols, and/or cognitive tasks employed; thus, it is difficult to draw conclusions regarding the effects of HIIT based on such limited data. Moreover, it is noteworthy that there is no extant data delineating the acute effect of HIIT on older adults’ executive function.
With regards to the chronic effects of HIIT on executive function, the available results are mixed. Preliminary data support the positive effect of HIIT on inhibition (De Sousa et al., Citation2018), indicating that adults with higher compliance to the HIIT protocol (peak in-exercise HR: 184.2 beats.min−1) had larger cognitive gains following training relative to those with lower compliance (peak in-exercise HR: 170.5 beats.min−1). This finding suggests a moderating role of training fidelity. However, the existing data does not support a positive influence of chronic HIIT to inhibition in older adults (Coetsee & Terblanche, Citation2017; Kovacevic et al., Citation2020), which may suggest further contemplation for implementing HIIT protocols in older adults. The current data on other subcomponents of executive function are scarce, with positive effects on working memory found among middle-aged adults (Connolly et al., Citation2017) but not young adults (Nicolini et al., Citation2019), and null effects also noted for cognitive flexibility in young adults (Eather et al., Citation2019). Nevertheless, more research is needed considering that these three studies differ from each other extensively in participants’ age, study design, HIIT protocol, and/or training duration (i.e., 6 weeks vs. 8 weeks vs. 12 weeks). In sum, the chronic effects of HIIT on executive function during adulthood remains to be clarified given the small number of studies and high heterogeneity in methodology across the literature.
4.4. Future directions
Given emerging evidence showing a positive effect of HIIT on executive function, there are several potential opportunities for future research. First, since most studies (70%) have assessed the effects of HIIT on inhibition, less is known regarding its effects on working memory and cognitive flexibility, especially in adults (). Likewise, more research delineating the chronic effects of HIIT or recruiting middle-aged and older adults are needed. Second, more studies are needed to clarify the dose-response relation between HIIT and different subcomponents of executive function by manipulating WRR. As mentioned earlier, Ludyga et al. (Citation2018) found positive effects of HIIT with smaller WRR but not HIIT with larger WRR. This finding highlights the necessity of clarifying whether different doses of HIIT may lead to different levels of cognitive gain. Third, given that HIIT can be performed in a variety of modalities, including running, cycling, and HICT, it is recommended that future work clarify whether different modalities of HIIT result in similar cognitive changes. In particular, circuit training consists of multi-joint, functional movements (Eddolls et al., Citation2017), which results in greater muscle recruitment than running/cycling, thereby improving cardiovascular endurance, muscular fitness, and body composition (Heinrich et al., Citation2015, Citation2012; Murawska-Cialowicz et al., Citation2015). It is plausible that the joint benefits in cardiovascular endurance, muscular fitness, and body composition lead to larger cognitive benefits compared to running or cycling as the three health dimensions are all associated with executive function (Hsieh et al., Citation2016; Kamijo et al., Citation2012; Kao et al., Citation2020). To date, there is no study delineating the differential or superior effects of HICT relative to HIIT. Fourth, there is a need to examine the moderating effects of individual differences, such as age (Chang et al., Citation2012; Ludyga et al., Citation2016), aerobic fitness (Chang et al., Citation2012), sexual dimorphism (Chang et al., Citation2012), genotype (e.g., BDNFmet66 carriers vs. non-carriers; Moreau et al., Citation2017), and clinical status (e.g., obesity vs. normal weight), as these factors have been suggested as potential moderators in the exercise-cognition relationship. Fifth, while the majority of previous studies only employed behavioural measures, there is still a long way to go in exploring the possible mechanisms underlying the relation. Despite several neurophysiological underpinnings (e.g., lactate, BDNF, cortical activation, cerebral oxygenation) proposed by researchers, other endothelial (e.g., VEGF) or endocrinal markers (e.g., irisin, interleukin-6, testosterone, cortisol) with cognitive implications (Carro et al., Citation2000; Gmiat et al., Citation2017; Huang et al., Citation2014; Hung et al., Citation2018; Tsai et al., Citation2015; Vital et al., Citation2014; Voss et al., Citation2013) should be investigated in future work.
Relative to methodological concerns, it would be useful for future research to include an active control condition, as an inactive comparison may not satisfactorily measure the effectiveness of exercise because it remains unclear whether cognitive gains are a result from locomotion, body composition, or expectations of improvement resulting from participation in a novel intervention (Pontifex et al., Citation2018, Citation2019). Moreover, in order to develop ecologically valid interventions, the feasibility of using HIIT programmes for improving executive function in real-world settings should be explored. With the exception of a few long-term studies conducted in schools (Costigan et al., Citation2016; Leahy et al., Citation2019), most previous studies have been conducted in laboratories, and thus there are questions about the generalizability of current results.
5. Concluding remarks
While HIIT has been proposed as a time-efficient alternative to traditional exercise, we currently have little knowledge regarding its effects on cognitive and brain health, particularly higher-order executive functions. Results of the current review have supported a positive effect of acute HIIT on the inhibition aspect of executive function in children/adolescents and adults as well as a beneficial effect of chronic HIIT on the inhibition and working memory in children. More research on chronic interventions, focusing on middle-age and older adults, and/or assessing working memory and cognitive flexibility are needed. Future research should also focus on a) using stronger research designs, b) the effects of HIIT dosage/modality, c) consideration of individual differences, d) neurobiological mechanisms underpin the HIIT-executive function relationship, and e) examining the feasibility of different modalities of HIIT in real-word settings.
Supplemental Material
Download MS Word (18.5 KB)Acknowledgments
This work was financially supported by the “Institute for Research Excellence in Learning Sciences” of National Taiwan Normal University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education in Taiwan and the Postdoctoral Research Abroad Program by Ministry of Science and Technology in Taiwan, under grant 109-2917-I-564-034.
Disclosure statement
All authors declare that they have no conflicts of interest relevant to the content of this article.
Supplementary material
Supplemental data for this article can be accessed here.
Additional information
Funding
References
- *Alves, C. R., Tessaro, V. H., Teixeira, L. A., Murakava, K., Roschel, H., Gualano, B., & Takito, M. Y. (2014). Influence of acute high-intensity aerobic interval exercise bout on selective attention and short-term memory tasks. Perceptual and Motor Skills, 118(1), 63–72. https://doi.org/10.2466/22.06.PMS.118k10w4
- Bailey, C. E. (2007). Cognitive accuracy and intelligent executive function in the brain and in business. Annals of the New York Academy of Sciences, 1118(1), 122–141. https://doi.org/10.1196/annals.1412.011
- Carro, E., Nuñez, A., Busiguina, S., & Torres-Aleman, I. (2000). Circulating insulin-like growth factor-I mediates effects of exercise on the brain. The Journal of Neuroscience, 20(8), 2926–2933. https://doi.org/10.1523/JNEUROSCI.20-08-02926.2000
- Centers for Disease Control and Prevention. (2012). Prevalence of insufficient physical activity. World Health Organization.
- Chang, Y. K., Labban, J. D., Gapin, J. I., & Etnier, J. L. (2012). The effects of acute exercise on cognitive performance: A meta-analysis. Brain Research, 1453, 87–101. https://doi.org/10.1016/j.brainres.2012.02.068
- *Cooper, S. B., Bandelow, S., Nute, M. L., Dring, K. J., Stannard, R. L., Morris, J. G., & Nevill, M. E. (2016). Sprint-based exercise and cognitive function in adolescents. Preventive Medicine Reports, 4, 155–161. https://doi.org/10.1016/j.pmedr.2016.06.004
- *Connolly, L. J., Bailey, S. J., Krustrup, P., Fulford, J., Smietanka, C., & Jones, A. M. (2017). Effects of self-paced interval and continuous training on health markers in women. European Journal of Applied Physiology, 117(11), 2281–2293. https://doi.org/10.1007/s00421-017-3715-9
- *Coetsee, C., & Terblanche, E. (2017). The effect of three different exercise training modalities on cognitive and physical function in a healthy older population. European Review of Aging and Physical Activity, 14(1), 13. https://doi.org/10.1186/s11556-017-0183-5
- Cooper, S. B., Dring, K. J., & Nevill, M. E. (2016). High-intensity intermittent exercise: Effect on young people’s cardiometabolic health and cognition. Current Sports Medicine Reports, 15(4), 245–251. https://doi.org/10.1249/JSR.0000000000000273
- *Costigan, S. A., Eather, N., Plotnikoff, R. C., Hillman, C. H., & Lubans, D. R. (2016). High-intensity interval training for cognitive and mental health in adolescents. Medicine & Science in Sports & Exercise, 48(10), 1985–1993. https://doi.org/10.1249/MSS.0000000000000993
- Davis, J. C., Marra, C. A., Najafzadeh, M., & Liu-Ambrose, T. (2010). The independent contribution of executive functions to health related quality of life in older women. BMC Geriatrics, 10(1), 16. https://doi.org/10.1186/1471-2318-10-16
- de Asteasu, M. L. S., Martinez-Velilla, N., Zambom-Ferraresi, F., Casas-Herrero, Á., & Izquierdo, M. (2017). Role of physical exercise on cognitive function in healthy older adults: A systematic review of randomized clinical trials. Ageing Research Reviews, 37, 117–134. https://doi.org/10.1016/j.arr.2017.05.007
- de Menezes, K. J., Peixoto, C., Nardi, A. E., Carta, M. G., Machado, S., & Veras, A. B. (2016). Dehydroepiandrosterone, its sulfate and cognitive functions. Clinical Practice & Epidemiology in Mental Health, 12(1), 24–37. https://doi.org/10.2174/1745017901612010024
- *de Sousa, A. F., Medeiros, A. R., Benitez-Flores, S., Del Rosso, S., Stults-Kolehmainen, M., & Boullosa, D. A. (2018). Improvements in attention and cardiac autonomic modulation after a 2-weeks sprint interval training program: A fidelity approach. Frontiers in Physiology, 9, 241. https://doi.org/10.3389/fphys.2018.00241
- Diamond, A. (2013). Executive functions. Annual Review of Psychology, 64(1), 135–168. https://doi.org/10.1146/annurev-psych-113011-143750
- *Dupuy, O., Billaut, F., Raymond, F., Benraiss, A., Theurot, D., Bosquet, L., Fraser, S., & Tremblay, J. (2018). Effect of acute intermittent exercise on cognitive flexibility: The role of exercise intensity. Journal of Cognitive Enhancement, 2(2), 146. https://doi.org/10.1007/s41465-018-0078-z
- *Eather, N., Riley, N., Miller, A., Smith, V., Poole, A., Vincze, L., Morgan, P. J., & Lubans, D. R. (2019). Efficacy and feasibility of HIIT training for university students: The Uni-HIIT RCT. Journal of Science and Medicine in Sport, 22(5), 596–601. https://doi.org/10.1016/j.jsams.2018.11.016
- Eddolls, W. T., McNarry, M. A., Stratton, G., Winn, C. O., & Mackintosh, K. A. (2017). High-intensity interval training interventions in children and adolescents: A systematic review. Sports Medicine, 47(11), 2363–2374. https://doi.org/10.1007/s40279-017-0753-8
- Etnier, J. L., & Chang, Y. K. (2009). The effect of physical activity on executive function: A brief commentary on definitions, measurement issues, and the current state of the literature. Journal of Sport and Exercise Psychology, 31(4), 469–483. https://doi.org/10.1123/jsep.31.4.469
- Freitas, D. A., Rocha-Vieira, E., Soares, B. A., Nonato, L. F., Fonseca, S. R., Martins, J. B., Lacerda, A. C., Massensini, A. R., Poortamns, J. R., Meeusen, R., Leite, H. R., & Mendonça, V. A. (2018). High intensity interval training modulates hippocampal oxidative stress, BDNF and inflammatory mediators in rats. Physiology & Behavior, 184, 6–11. https://doi.org/10.1016/j.physbeh.2017.10.027
- Gmiat, A., Micielska, K., Kozłowska, M., Flis, D. J., Smaruj, M., Kujach, S., Jaworska, J., Lipińska, P., & Ziemann, E. (2017). The impact of a single bout of high intensity circuit training on myokines‘ concentrations and cognitive functions in women of different age. Physiology & Behavior, 179, 290–297. https://doi.org/10.1016/j.physbeh.2017.07.004
- Guiney, H., & Machado, L. (2013). Benefits of regular aerobic exercise for executive functioning in healthy populations. Psychonomic Bulletin & Review, 20(1), 73–86. https://doi.org/10.3758/s13423-012-0345-4
- *Hashimoto, T., Tsukamoto, H., Takenaka, S., Olesen, N. D., Petersen, L. G., Sørensen, H., Nielsen, H. B., Secher, N. H., & Ogoh, S. (2018). Maintained exercise-enhanced brain executive function related to cerebral lactate metabolism in men. The FASEB Journal, 32(3), 1417–1427. https://doi.org/10.1096/fj.201700381RR
- Heaney, J. L., Carroll, D., & Phillips, A. C. (2013). DHEA, DHEA-S and cortisol responses to acute exercise in older adults in relation to exercise training status and sex. Age, 35(2), 395–405. https://doi.org/10.1007/s11357-011-9345-y
- Heinrich, K. M., Becker, C., Carlisle, T., Gilmore, K., Hauser, J., Frye, J., & Harms, C. A. (2015). High-intensity functional training improves functional movement and body composition among cancer survivors: A pilot study. European Journal of Cancer Care, 24(6), 812–817. https://doi.org/10.1111/ecc.12338
- Heinrich, K. M., Spencer, V., Fehl, N., & Carlos Poston, W. S. (2012). Mission essential fitness: Comparison of functional circuit training to traditional army physical training for active duty military. Military Medicine, 177(10), 1125–1130. https://doi.org/10.7205/MILMED-D-12-00143
- Hillman, C. H., Erickson, K. I., & Kramer, A. F. (2008). Be smart, exercise your heart: Exercise effects on brain and cognition. Nature Reviews Neuroscience, 9(1), 58. https://doi.org/10.1038/nrn2298
- Hsieh, S. S., Chang, Y. K., Hung, T. M., & Fang, C. L. (2016). The effects of acute resistance exercise on young and older males’ working memory. Psychology of Sport and Exercise, 22, 286–293. https://doi.org/10.1016/j.psychsport.2015.09.004
- Hsieh, S. S., Huang, C. J., Wu, C. T., Chang, Y. K., & Hung, T. M. (2018). Acute exercise facilitates the N450 inhibition marker and P3 attention marker during stroop test in young and older adults. Journal of Clinical Medicine, 7(11), 391. https://doi.org/10.3390/jcm7110391
- Huang, T., Larsen, K. T., Ried‐Larsen, M., Møller, N. C., & Andersen, L. B. (2014). The effects of physical activity and exercise on brain‐derived neurotrophic factor in healthy humans: A review. Scandinavian Journal of Medicine & Science in Sports, 24(1), 1–10. https://doi.org/10.1111/sms.12069
- Hung, C. L., Tseng, J. W., Chao, H. H., Hung, T. M., & Wang, H. S. (2018). Effect of acute exercise mode on serum brain-derived neurotrophic factor (BDNF) and task switching performance. Journal of Clinical Medicine, 7(10), 301. https://doi.org/10.3390/jcm7100301
- Jiménez-García, J. D., Hita-Contreras, F., de la Torre-Cruz, M., Fábrega-Cuadros, R., Aibar-Almazán, A., Cruz-Díaz, D., & Martínez-Amat, A. (2019). Risk of falls in healthy older adults: Benefits of high-intensity interval training using lower body suspension exercises. Journal of Aging & Physical Activity, 27(3), 325–333. https://doi.org/10.1123/japa.2018-0190
- Kamijo, K., Hayashi, Y., Sakai, T., Yahiro, T., Tanaka, K., & Nishihira, Y. (2009). Acute effects of aerobic exercise on cognitive function in older adults. The Journals of Gerontology Series B: Psychological Sciences and Social Sciences, 64(3), 356–363. https://doi.org/10.1093/geronb/gbp030
- Kamijo, K., Khan, N. A., Pontifex, M. B., Scudder, M. R., Drollette, E. S., Raine, L. B., Evans, E. M., Castelli, D. M., & Hillman, C. H. (2012). The relation of adiposity to cognitive control and scholastic achievement in preadolescent children. Obesity, 20(12), 2406–2411. https://doi.org/10.1038/oby.2012.112
- *Lambrick, D., Stoner, L., Grigg, R., & Faulkner, J. (2016). Effects of continuous and intermittent exercise on executive function in children aged 8–10 years. Psychophysiology, 53(9), 1335–1342. https://doi.org/10.1111/psyp.12688
- *Kao, S. C., Westfall, D. R., Soneson, J., Gurd, B., & Hillman, C. H. (2017). Comparison of the acute effects of high‐intensity interval training and continuous aerobic walking on inhibitory control. Psychophysiology, 54(9), 1335–1345. https://doi.org/10.1111/psyp.12889
- *Kao, S. C., Drollette, E. S., Ritondale, J. P., Khan, N., & Hillman, C. H. (2018). The acute effects of high-intensity interval training and moderate-intensity continuous exercise on declarative memory and inhibitory control. Psychology of Sport and Exercise, 38, 90–99. https://doi.org/10.1016/j.psychsport.2018.05.011
- *Kujach, S., Olek, R. A., Byun, K., Suwabe, K., Sitek, E. J., Ziemann, E., Laskowski, R., & Soya, H. (2020). Acute sprint interval exercise increases both cognitive functions and peripheral neurotrophic factors in humans: The possible involvement of lactate. Frontiers in Neuroscience, 13, 1455. https://doi.org/10.3389/fnins.2019.01455
- *Kovacevic, A., Fenesi, B., Paolucci, E., & Heisz, J. J. (2020). The effects of aerobic exercise intensity on memory in older adults. Applied Physiology, Nutrition, and Metabolism, 45(6), 591–600. https://doi.org/10.1139/apnm-2019-0495
- Kao, S. C., Cadenas‐Sanchez, C., Shigeta, T. T., Walk, A. M., Chang, Y. K., Pontfiex, M. B., & Hillman, C. H. (2020). A systematic review of physical activity and cardiorespiratory fitness on P3b. Psychophysiology, 57(7), e13425. https://doi.org/10.1111/psyp.13425
- Leahy, A. A., Eather, N., Smith, J. J., Hillman, C. H., Morgan, P. J., Plotnikoff, R. C., Nilsson, M., Costigan, S. A., Noetel, M., & Lubans, D. R. (2019). Feasibility and preliminary efficacy of a teacher-facilitated high-intensity interval training intervention for older adolescents. Pediatric Exercise Science, 31(1), 107–117. https://doi.org/10.1123/pes.2018-0039
- Lee, C. L., Hsu, W. C., & Cheng, C. F. (2018). Physiological adaptations to sprint interval training with matched exercise volume. Medicine & Science in Sports & Exercise, 49(1), 86–95. https://doi.org/10.1249/MSS.0000000000001083
- Lee, C. L., Kuo, Y. H., & Cheng, C. F. (2018). Acute high-intensity interval cycling improves postprandial lipid metabolism. Medicine & Science in Sports & Exercise, 50(8), 1687–1696. https://doi.org/10.1249/MSS.0000000000001613
- Li, J. W., O’Connor, H., O’Dwyer, N., & Orr, R. (2017). The effect of acute and chronic exercise on cognitive function and academic performance in adolescents: A systematic review. Journal of Science and Medicine in Sport, 20(9), 841–848. https://doi.org/10.1016/j.jsams.2016.11.025
- *Ligeza, T. S., Maciejczyk, M., Kałamała, P., Szygula, Z., & Wyczesany, M. (2018). Moderate-intensity exercise boosts the N2 neural inhibition marker: A randomized and counterbalanced ERP study with precisely controlled exercise intensity. Biological Psychology, 135, 170–179. https://doi.org/10.1016/j.biopsycho.2018.04.003
- Lipsky, R. H., & Marini, A. M. (2007). Brain‐derived neurotrophic factor in neuronal survival and behavior‐related plasticity. Annals of the New York Academy of Sciences, 1122(1), 130–143. https://doi.org/10.1196/annals.1403.009
- Lopes, V. P., Vasques, C., Pereira, B., Maia, J. A., & Malina, R. M. (2006). Physical activity patterns during school recess: A study in children 6 to 10 years old. The International Electronic Journal of Health Education, 9, 192–201.
- Ludyga, S., Gerber, M., Brand, S., Holsboer‐Trachsler, E., & Pühse, U. (2016). Acute effects of moderate aerobic exercise on specific aspects of executive function in different age and fitness groups: A meta-analysis. Psychophysiology, 53(11), 1611–1626. https://doi.org/10.1111/psyp.12736
- *Ludyga, S., Pühse, U., Lucchi, S., Marti, J., & Gerber, M. (2018). Immediate and sustained effects of intermittent exercise on inhibitory control and task-related heart rate variability in adolescents. Journal of Science and Medicine in Sport, 22(1), 96–100. https://doi.org/10.1016/j.jsams.2018.05.027
- Magistretti, P. J., & Allaman, I. (2018). Lactate in the brain: From metabolic end-product to signaling molecule. Nature Reviews Neuroscience, 19(4), 235–249. https://doi.org/10.1038/nrn.2018.19
- Maher, C. G., Moseley, A. M., Sherrington, C., Elkins, M. R., & Herbert, R. D. (2008). A description of the trials, reviews, and practice guidelines indexed in the PEDro database. Physical Therapy, 2008(88), 1068–1077. https://doi.org/10.2522/ptj.20080002
- McMorris, T., Sproule, J., Turner, A., & Hale, B. J. (2011). Acute, intermediate intensity exercise, and speed and accuracy in working memory tasks: A meta-analytical comparison of effects. Physiology & Behavior, 102(3–4), 421–428. https://doi.org/10.1016/j.physbeh.2010.12.007
- *Miller, M. G., Hanson, N., Tennyck, J., & Plantz, K. A comparison of high-intensity interval training (HIIT) volumes on cognitive performance. (2018). Journal of Cognitive Enhancement, 3(2), 168–173. 2019. https://doi.org/10.1007/s41465-018-0107-y
- Moher, D., Liberati, A., Tetzlaff, J., & Altman, D. G. (2009). Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Annals of Internal Medicine, 151(4), 264–269. https://doi.org/10.7326/0003–4819-151-4-200908180-00135
- *Moreau, D., Kirk, I. J., & Waldie, K. E. (2017). High-intensity training enhances executive function in children in a randomized, placebo-controlled trial. Elife, 6, e25062. https://doi.org/10.7554/eLife.25062
- Murawska-Cialowicz, E., Wojna, J., & Zuwala-Jagiello, J. (2015). Crossfit training changes brain-derived neurotrophic factor and irisin levels at rest, after wingate and progressive tests, and improves aerobic capacity and body composition of young physically active men and women. Journal of Physiology and Pharmacology, 66(6), 811–821.
- Nascimento, C. M. C., Pereira, J. R., Pires de Andrade, L., Garuffi, M., Ayan, C., Kerr, D. S., Talib, L. L., Cominetti, M. R., & Stella, F. (2015). Physical exercise improves peripheral BDNF levels and cognitive functions in mild cognitive impairment elderly with different BDNF Val66Met genotypes. Journal of Alzheimer’s Disease, 43(1), 81–91. https://doi.org/10.3233/JAD-140576
- *Nicolini, C., Toepp, S., Harasym, D., Michalski, B., Fahnestock, M., Gibala, M. J., & Nelson, A. J. (2019). No changes in corticospinal excitability, biochemical markers, and working memory after six weeks of high‐intensity interval training in sedentary males. Physiological Reports, 7(11), e14140. https://doi.org/10.14814/phy2.14140
- O'Brien, M. W., Johns, J. A., Robinson, S. A., Bungay, A., Mekary, S. A. I. D., & Kimmerly, D. S. (2020). Impact of high-intensity interval training, moderate-intensity continuous training, and resistance training on endothelial function in older adults. Medicine and Science in Sports and Exercise, 52(5), 1057–1067. https://doi.org/10.1249/MSS.0000000000002226
- Ponjee, G. A., De, H. R., & Vader, H. L. (1994). Androgen turnover during marathon running. Medicine & Science in Sports & Exercise, 26(10), 1274–1277. https://doi.org/10.1249/00005768-199410000-00015
- Pontifex, M. B., Gwizdala, K. L., Weng, T. B., Zhu, D. C., & Voss, M. W. (2018). Cerebral blood flow is not modulated following acute aerobic exercise in preadolescent children. International Journal of Psychophysiology, 134, 44–51. https://doi.org/10.1016/j.ijpsycho.2018.10.007
- Pontifex, M. B., McGowan, A. L., Chandler, M. C., Gwizdala, K. L., Parks, A. C., Fenn, K., & Kamijo, K. (2019). A primer on investigating the after effects of acute bouts of physical activity on cognition. Psychology of Sport and Exercise, 40, 1–22. https://doi.org/10.1016/j.psychsport.2018.08.015
- Robinson, M. M., Lowe, V. J., & Nair, K. S. (2018). Increased brain glucose uptake after 12 weeks of aerobic high-intensity interval training in young and older adults. Journal of Clinical Endocrinology & Metabolism, 103(1), 221–227. https://doi.org/10.1210/jc.2017-01571
- Rynecki, N. D., Siracuse, B. L., Ippolito, J. A., & Beebe, K. S. (2019). Injuries sustained during high intensity interval training: Are modern fitness trends contributing to increased injury rates? The Journal of Sports Medicine and Physical Fitness, 59(7), 1206–1212. https://doi.org/10.23736/S0022-4707.19.09407-6
- *Schwarck, S., Schmicker, M., Dordevic, M., Rehfeld, K., Müller, N., & Müller, P. (2019). Inter-individual differences in cognitive response to a single bout of physical exercise—A randomized controlled cross-over study. Journal of Clinical Medicine, 8(8), 1101. https://doi.org/10.3390/jcm8081101
- Soga, K., Masaki, H., Gerber, M., & Ludyga, S. (2018). Acute and long-term effects of resistance training on executive function. Journal of Cognitive Enhancement, 2(2), 200–207. https://doi.org/10.1007/s41465-018-0079-y
- *Tsukamoto, H., Suga, T., Takenaka, S., Tanaka, D., Takeuchi, T., Hamaoka, T., Isaka, T., & Hashimoto, T. (2016). Greater impact of acute high-intensity interval exercise on post-exercise executive function compared to moderate-intensity continuous exercise. Physiology & Behavior, 155, 224–230. https://doi.org/10.1016/j.physbeh.2015.12.021
- *Tottori, N., Morita, N., Ueta, K., & Fujita, S. (2019). Effects of high intensity interval training on executive function in children aged 8–12 years. International Journal of Environmental Research and Public Health, 16(21), 4127. https://doi.org/10.3390/ijerph16214127
- Tsai, C. L., Wang, C. H., Pan, C. Y., & Chen, F. C. (2015). The effects of long-term resistance exercise on the relationship between neurocognitive performance and GH, IGF-1, and homocysteine levels in the elderly. Frontiers in Behavioral Neuroscience, 9, 23. https://doi.org/10.3389/fnbeh.2015.0002
- Vital, T. M., Stein, A. M., de Melo Coelho, F. G., Arantes, F. J., Teodorov, E., & Santos-Galduroz, R. F. (2014). Physical exercise and vascular endothelial growth factor (VEGF) in elderly: A systematic review. Archives of Gerontology and Geriatrics, 59(2), 234–239. https://doi.org/10.1016/j.archger.2014.04.011
- Voss, M. W., Erickson, K. I., Prakash, R. S., Chaddock, L., Kim, J. S., Alves, H., Szabo, A., Phillips, S. M., Wójcicki, T. R., Mailey, E. L., Olson, E. A., Gothe, N., Vieira-Potter, V. J., Martin, S. A., Pence, B. D., Cook, M. D., Woods, J. A., McAuley, E., & Kramer, A. F. (2013). Neurobiological markers of exercise-related brain plasticity in older adults. Brain, Behavior, and Immunity, 28, 90–99. https://doi.org/10.1016/j.bbi.2012.10.021
- Wang, C. C., Alderman, B., Wu, C. H., Chi, L., Chen, S. R., Chu, I. H., & Chang, Y. K. (2019). Effects of acute aerobic and resistance exercise on cognitive function and salivary cortisol responses. Journal of Sport and Exercise Psychology, 41(2), 73–81.
- Wang, S., Allen, R. J., Lee, J. R., & Hsieh, C. E. (2015). Evaluating the developmental trajectory of the episodic buffer component of working memory and its relation to word recognition in children. Journal of Experimental Child Psychology, 133, 16–28. https://doi.org/10.1016/j.jecp.2015.01.002
- Wang, S., & Gathercole, S. E. (2013). Working memory deficits in children with reading difficulties: Memory span and dual task coordination. Journal of Experimental Child Psychology, 115(1), 188–197. https://doi.org/10.1016/j.jecp.2012.11.015
- Wang, S., & Gathercole, S. E. (2015). Interference control in children with reading difficulties. Child Neuropsychology, 21(4), 418–431. https://doi.org/10.1080/09297049.2014.918594
- Wilke, J., Giesche, F., Klier, K., Vogt, L., Herrmann, E., & Banzer, W. (2019). Acute effects of resistance exercise on cognitive function in healthy adults: A systematic review with multilevel meta-analysis. Sports Medicine, 49(6), 905–916. https://doi.org/10.1007/s40279-019-01085-x
- Zhou, L. J., Yang, T., Wei, X., Liu, Y., Xin, W. J., Chen, Y., Pang, R. P., Zang, Y., Li, Y. Y., & Liu, X. G. (2011). Brain-derived neurotrophic factor contributes to spinal long-term potentiation and mechanical hypersensitivity by activation of spinal microglia in rat. Brain, Behavior, and Immunity, 25(2), 322–334. https://doi.org/10.1016/j.bbi.2010.09.025