Abstract
This article makes a case for a positive role of tension in the creative process in chemistry. I begin with an argument that there is an inherent tension in what makes molecules interesting—their positioning along various polar axes. One of these, the age-old differentiation between useful (to society and for personal profit) commercialization and pure understanding of molecules and their reactions is characteristic. The question of whether there are any bad molecules then leads me to ethical concerns in chemistry, and a particular working out of these in interactions of chemists in the Middle East. An analysis is made of the special tensions involved in publishing, especially in citation ethics; chemists publish a lot, so this is situation ethics worked out on a daily basis. I then find in the literature of psychology good evidence for the positive value of moderate stress in stimulating creativity. It is obvious that too much tension leads to distress, and there are some institutional aspects of chemistry that do not come out well here. But all in all, the dynamic middle is alive, and it leads to good new science.
Editor’s Note
Those who have the good fortune to read any of Roald Hoffmann’s writings are blessed. In 1963, he published several quantum chemical papers on what is called the “Extended Hückel Method.” These publications catapulted theory into mainstream organic chemistry. In 1965, Hoffmann—a Junior Fellow at Harvard—and his Harvard colleague R. B. Woodward published five short communications that explained a third of organic chemical reactions and led to Hoffmann’s Nobel in 1981. (Woodward died in 1979; else, he would have received his second Nobel Prize. That very same year, 1965, Woodward received the Nobel Prize for his research in synthetic organic chemistry.) To this day, at 77, Hoffmann continues to publish original chemical research—along with essays (regularly in American Scientist), plays, poems, and other literature. Several things characterize Hoffmann: his breadth of interests, his open and searching mind, his facility to bring diverse ideas and information together in novel and meaningful fashion, and his communication style. And the idea-density and scholarship of his writings. I wanted readers of this special issue of Accountability in Research to have the opportunity to attend a smorgasbord of intellectualism and individualism from Hoffmann’s deeply personal perspective as they relate to ethics and responsible conduct of research. I wish you all a hearty appetite.
Jeffrey I. Seeman
Guest Editor
University of Richmond
Richmond, Virginia 23173, USA
E-mail: [email protected]
Most of us do not view tension positively. Every day of our lives seems to fill with deadlines, obligations. The prevailing sense is that we cannot possibly do all that we want to or others expect us to do. These feelings generate stress and make us feel psychologically uncomfortable, if not unhappy.
I would like to argue that there is another way to view tension than for its discontents, and this is as natural, even a blessing in disguise, as a source of creativity: That our profession, our chemistry, has an innate set of tensions. And that the everyday practice of chemistry generates still other tensions that (a) are in fact transmuted into greater knowledge and better practice, and (b) are important in honing ethical judgment.
POLARITIES SHAPE CHEMISTRY
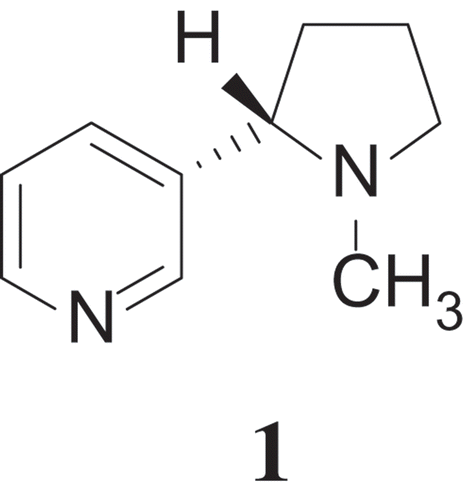
My proposition in The Same and Not the Same was that every chemical fact is poised on the axes of not one, but several polarities, balances between extremes (Hoffmann, Citation1995). So the molecule of nicotine has so much more to it than its stoichiometry and atom connectivity, than it being (S)-3-[1-Methylpyrrolidin-2-yl]pyridine (1):
Nicotine is a liquid with a low melting point, one that can be easily dispersed in an aerosol (ergo the recent interest in and concern about electronic cigarettes). This naturally occurring alkaloid is easy to make or not, and is a stimulant or a relaxant (it is both). Nicotine is there as a microscopic molecule, which we can “see” (or try to) with scanning tunneling microscopy or, as its salts, by X-ray crystallography. Nicotine is addictive and is used (in some smoking cessation devices) to control and eliminate smoking. A chemist might want to patent a synthesis of a derivative. Or publish it. Or both.
So many ways to think about a single molecule! Here is a partial list of the polarities along which nicotine, or any molecule, is aligned:
macro–micro
simple–complex
symmetry–asymmetry
harm–benefit
natural–unnatural
creation–discovery
static–dynamic
symbolic–iconic
synthesis–analysis
pure–impure
to reveal–to conceal
trust–skepticism
the same and not the same
Molecules reside on the many axes of chemical interest. But they most certainly do not rest easily on any. I have been thinking about writing a book that might formulate a philosophy of life out of chemical concepts. First among these, for sure, would be that most characteristic and distinguishing aspect of chemistry, that the molecule (or the property of the molecule that interests us) can change. And does change. Heraclitus was right—everything flows, nothing stands still. Change, or the potential of change, define chemistry.
The next motive idea in that far-out book might be equilibrium—which, of course, for chemists does not mean lying there, quietly. Yes, A + B → C + D. But at the same time, all the time, C + D → A + B. We are not talking of two molecules—in any drop of a solution there are 1019 of them, all moving madly, colliding frequently. At equilibrium there are some A and B molecules, some C and D ones; there may be little of any one molecule, but it is there. In its sealed ampoule, the compound just looks placid and static at that middle point, the concentrations of what made it up, and what it may react to not changing. But it is a dynamic middle, the world of those molecules, and not just in their chaotic molecular motions, but also ripe with potential. Do you want to have the reaction go one way, or the other? We can perturb that equilibrium.
Change, or the potential of change (the molecule modified to be more or less addictive, more or less soluble) creates tension—the molecule refuses to sit still in reality, and in our mind’s eyes. And still other tensions may take shape in the minds of the practitioners of chemistry, as they go about their professional labors.
Pure and Applied
Those attributes are enshrined in the name of IUPAC, the International Union of our science. They describe a particular tension that animates chemistry, in that the products of our science have a direct impact on the human condition. One can expend many nice words in justifying knowledge for the sake of knowledge, but from the beginning, before there ever were chemists, people did chemistry for profit. So Egyptian blue was used in brickwork of the Gate of Ishtar, and Chinese blue, a related pigment (both with Cu2+ as a chromophore) was employed to decorate the Xi’an army (Berke, Citation2002). Protochemistries of winning metals from their ores, making alloys, cooking, preserving food, fermentation, distillation, of preparing medications, ceramics, dyes, textile preparation, cosmetics, tanning, mummification, in time turned into industries.
The economic and military interests of governments or patrons always were a driving force for research. We can see this in Duke Augustus of Saxony financing the remarkable European rediscovery of Chinese porcelain in the first decade of the 18th century (Hoffmann, Citation2004b), or in Lavoisier’s work on assuring a supply of nitrate for gun-powder for royal and republican France (Poirier, Citation1996). An argument could be made for American laser chemistry benefitting from “star-wars” ideas (the Strategic Defense Initiative) in the 1980s. And today some pretty chemistry rides unrealistically, with easily recognizable, almost comic verbiage decorating the beginning and endings of papers, on the tails of present concerns about hydrogen storage or high energy capacity materials.
But then there is another thread of argument in our time, that of doing science, and chemistry in particular, for the purpose of advancing knowledge—the “pure” in “pure and applied.” Science for science’s sake is possibly a 19th century idea, connected to the growth of universities away from being religious institutions. In modern times the justification is often made in terms of eventually commercialized advances, such as the transistor, lasers, or polymers, which had their origins in fundamental research. Lives have been saved, our condition ameliorated, by molecules that emerge from a basic research program.
It is a tension I feel personally. For I made a conscious choice for understanding rather than practice. But I feel guilty about it, in that I could be doing something more useful to humanity. My concerns are lifted, barely so, when I see that many pharmaceuticals patented, synthesized, and marketed using ideas in their synthesis of which reactions might go easily and which might not be based on the Woodward-Hoffmann rules or frontier orbital thinking. I, who do not “own” a single patent, can then feel that I am helping, without running after the money. But underneath I know that what keeps me thinking up new molecules is just the fun of it. And that I can parlay that fun into a broader framework of chemical bonding that (a) is useful, and (b) can be taught.
It is easy to see that the current rush to commercialization of discovery out of chemical (and medical and engineering) laboratories can lead to other tensions. I worry, and so do others (Kovac and Coppola, Citation2000; Coppola, Citation2001) that the graduate students working in a group with a professor who has one or more startup companies may have their education distorted by that, even if the commercial activities are separated from the government-sponsored academic research. I know my worries are those of an outsider. But, as Jeff Seeman reminded me, that self-description itself is a tension—I am also an insider, very much in the chemical community, reflective and willing to speak out on how chemistry is taught and used.
Are There Any Bad Molecules?
The first answer is “no, there aren’t any bad molecules”—only bad people. The point is more interestingly made in the context of molecules that both heal and harm—of ozone and morphine. Ozone is a harmful pollutant at sea level, and a saving filterer of UV radiation in the stratosphere. Morphine is our most wonderful painkiller, and very addictive.
The “no bad molecules, only bad people” slogan, of course, evokes the argument of the anti-gun-control lobby. Is it right to ban guns, or to ban molecules? My opinion is that under our unspoken social contract, society has the right to ban both. It should exercise that right with care.
Let me be specific, and speak about thalidomide. You know that terrible story of the unscrupulous marketing of a potent teratogenic agent. I describe it in my book The Same and Not the Same (Hoffmann, Citation1995). In the sequel, in the 1960s the world spoke clearly “never again,” and put into place laws and regulatory regulations to ensure that.
And now the FDA has approved the manufacture of thalidomide in the United States. It can be prescribed for complications of hanseniasis (leprosy) and there are indications of antitumor activity. The approval came with the most stringent warnings attached to its use, and precautions against misuse. My personal opinion, with which some surely disagree, is that I think the molecule should have been banned. Why?
Well, another country (Brazil) has tried the experiment of limited licensed use against leprosy, while taking measures to warn and monitor patients against use by pregnant women in the first trimester. Nevertheless, the drug has been misused, in a variety of circumstances, and there are apparently several dozen documented recent thalidomide-deformed births (Estado de S. Paulo, Citation1994).
Perhaps the medical system in the U.S. is better than in Brazil, so misuse could not occur here. I am not that sure. My opinion is that some potential effects of a pharmaceutical have moral consequences of a nature such that the normal and agonizing risk equation (benefit to some vs. potential harm to others) just cannot be used. Risks to infants and children, the repositories of the future of humanity, to me constitute such an unacceptable risk. This drug should not be licensed, in my opinion, under any circumstances.
Instead, pharmaceutical researchers should be given incentives to develop thalidomide-related molecules which are effective but do not cause birth malformations.
You need not agree with me. But I think there are very few extreme libertarians who would argue that governments do not have the authority to constrain the making, sale, or consumption of one molecule or another. Think of angel dust. Think of hydrogen cyanide. There are no evil molecules, but governments can forbid people to make certain molecules.
Ethical Concerns
The previous two sections have shifted subtly from scientific tensions to ethical ones. These are ably discussed by Jeff Kovac elsewhere in this issue of Accountability in Research; the community owes Jeff much for his dedicated discussion of ethics in scientific research, and in particular for writing the one and only case book in our field (Kovac, Citation2003).
It has been my fortune to be faced with thinking about not one but many ethical situations—how to deal with E. J. Corey’s claim to have given R. B. Woodward the first, simplest frontier orbital explanation of the specificity of electrocyclic reactions (Hoffmann, Citation2004a); what to do with the claims of Peter Debye’s having cooperated with the Nazi regime (Hoffmann, Citation2006; Abruña, Citation2006); how to react to a IUPAC-sponsored conference in Jordan that excluded Israelis (for a report I view as biased, see Rouhi, Citation2010). I do not say “my fortune” ironically; it is a statement of fact. Even as there are moments when I think that I wish I had been spared, actually I am grateful to have been impelled, by the circumstances in each case, to think. For ethics needs exercise; it needs discussion among friends and with people who disagree. Ethical judgments are deeply personal and existential. And easy only in textbooks. In the real world, ethical concerns need to be worked out in personal conversations.
I have discussed elsewhere the Corey claim, and the Debye case, and written more generally of ethics in science as we enter the third millennium (Kovac and Weisberg, Citation2012). Here, I want to enter some of the ethical tensions that arise from science being a collegial international practice, yet one moored in the real world.
Collegiality and the Middle East
One of the unresolved political tragedies of our time is the inability of Israel, Palestine, the surrounding Arab countries, and Iran to reach a modus vivendi. The matter is of personal interest as I am from a Polish Jewish background, and have many relatives in Israel—some who went there in the 1920s and 1930s, some after World War II. Believing in the legitimacy of Israel, and with pride in its achievements, I have watched with great disquiet its political actions in occupied Palestine. And like many others, I have wanted to do something, be it small, to help bring about peace in the region.
So it has been a great satisfaction to take part in the Malta conferences, running for over a decade now, existing only by the dint of Zafra Lerman’s great energy (Everts, Citation2013; Hoffmann Citation2014). The conferences, held not only in Malta, bring together chemists from the Arab countries, Israel, and Iran. The plenary lectures were mostly by Nobel Laureates—that affectation was deemed a necessity—workshops, discussion groups, and posters came from all countries.
Cordial, interactive, and informative as they were, there was naturally some tension in the air at the meetings, for politically charged issues, such as the control of water rights, were discussed. Descriptions were given of the indignities to which Palestinians were subjected in passing from one part of their land to another. And there were chilling reports at the last meeting of a Syrian colleague killed, of students trying to do labs with gun fire around them.
Encouraged by the Malta conferences, I organized three workshops in 2006 and 2007 in the Middle East, with 15 young (under 35) scientists in each, mostly graduate students, from the Arab countries, Israel, and Iran. I was helped in this by Pere Alemany, Vanessa Buisson, Steve Lippard, Ralph Nuzzo, and George Whitesides. And hindered by politics, of course. We had to have the workshops in places where Israelis and Arab country nationals could meet, and that meant Jordan or Egypt; we almost talked Qatar into hosting one, but in the end they would not allow it. But we could not get Iranians into Egypt, so poor were the relations between the two countries.
The attraction to the participants was the stellar science proffered—who would not want to learn bioinorganic chemistry from Steve Lippard? There was a bonding mechanism, the Marine Corps principle—work the group so hard that they forget their differences and bond through survival. I think it worked. But in the years that followed, the political situation in the region deteriorated, and I could not raise the money I needed for further workshops from the Arab sources that were needed for legitimacy.
In 2010, I noticed that the 11th Eurasian Conference on Chemical Science was to be held in Jordan, and had a hundred speakers listed but not a single Israeli. That was a conspicuous piece of politicized prejudice; it was as if there were a chemical congress in Belgium, and no French scientists were invited. You cannot imagine that, right? If I raised a stink about this, there would be a severe risk to my efforts to raise support for my workshops. But that is what I did.
In the process I encountered incredible moral blindness by others to what the obvious (at least to me) political exclusion of Israeli chemists meant. So Nicole Moreau, then the President of IUPAC, wrote to me, “explaining” why IUPAC had no problem with sponsorship of the meeting:
In no case is the choice of invited speakers an element that has to be taken as a criterion for sponsorship. On another hand, if we look at the list of invited speakers, many countries are not represented, for instance Spain, which could be surprising for an Eurasia conference: we noted no complaint from any Spanish chemist! This could be put in parallel to your example about Korea and Japan. And there is nobody from Latin America …
I do not intend to ask the organizers why they did not invite speakers from Israel. I trust you to understand that this is not a personal decision, but that this is because it is not the role of IUPAC to interfere with the organizers’ choices and decisions. I realize, however, that these choices and decisions cannot give satisfaction to everybody, considering the very delicate situation in the region. (Moreau, Citation2010)
My small efforts in the Middle East are, in the end, inconsequential. In the context of my theme of tensions, there is precious little life-giving here, mostly the petty political perversion of the internationalist aspiration to scientific knowledge being shared by humanity. I still feel that the shared quest for knowledge to benefit humanity spiritually and practically is one light that can lead us out of the dark spirals of prejudice that politicians are all too good at swirling in our souls. It is worth trying, as tense as it gets, for the middle way.
The Continual Exercise of Situation Ethics in Chemical Publishing
We are not yet done with ethics; how can we be? I would, however, like to shift to the positive aspects of the tensions pervading science, with a case for the particularity of chemistry in the shaping of ethics by our publication practices.
Chemists publish much and often. On the scale of science, I suspect only molecular biologists can “compete,” but then they are really chemists as well. Chemical Abstracts indexes over 700,000 articles per year in chemistry and related fields. I suspect that a good half of these are what people would agree is chemistry, i.e., about one thousand a day. My RSS (Rich Site Summary) aggregator brings in the abstracts of ˜100 journals, about 1,500 articles weekly; this is what I feel I must read (well, glance at) to keep up. All of us know colleagues who have published 500 to 1,000 articles. Our unit of publication in chemistry is pretty small, 4 to 10 printed—whoops, not much is “printed” today—pages, with 25–75 endnotes.
Each article is a small exercise in publication ethics. My claim is that being forced to go through the exercise, say 6 times a year, is a salutary experience for the writer/researcher, naturally shared with her or his postdoctoral visitors or graduate students. And it is an experience in which reviewers of the articles play an important role.
The situation ethics is that of giving proper credit. Jeff Seeman and Mark House have carried out an important study of the realistic practices of crediting advice and discussion (House and Seeman, Citation2010; Seeman and House, Citation2010a,b). Journal codes of ethical practice give wise but general guidelines that apparently are not widely used, at least by chemists (House and Seeman, Citation2010). But in the end, the discussion of acknowledging prior history of a problem and forerunners in theory or experiment is a very personal one, often taken by the lead author, sometimes in consultation.
External pressures may influence the decision. The journal Nature, for instance, limits the numbers of references per article. You can be a very good, recognized scientist without ever publishing in Nature, but go tell a young Chinese chemist that! There are also special problems (mostly in the past) with of Americans ignoring literature published in other languages, and of nothing more that laziness making people cite reviews rather than source articles.
Jeff Seeman also reminded me of the idea of “obliteration by incorporation,” introduced by our mutual hero, Robert K. Merton (Merton Citation1968). Some ideas in science (and phrases in writing) are as intellectually attractive and/or useful as to become seemingly self-evident, needing no citation. Languages clearly grow that way. As long as there is an occasional reminder that a real live person thought of that first, no great harm is done in science by this phenomenon. Perhaps one could even aspire to be obliterated in this way!
Let me interpose here a personal experience to show the complexity of the problem of just citation. Maosheng Miao and I have just written what I think is an important article on high pressure electrides, which is in Accounts of Chemical Research (Miao and Hoffmann, Citation2014). Accounts of Chemical Research is a place for personal reviews of mainly one’s own work—a situation of the research in the surrounding literature is expected, but not a systematic review. Nevertheless, I felt the pressure to reference previous work. Here was, for instance, Jim Dye’s beautiful work on alkali metal ambient condition electrides. But our article was on high pressure materials, beasts of a different stripe. I felt guilty (and said so in the article) about having to relegate most of the references to Dye’s work to the Supplementary Information of the article. Another beautiful body of work by Arndt Simon on alkali metal suboxides and subnitrides, Swiss-cheese metals, was also not directly relevant, and mentioned only briefly by us. But other people had more or less ignored the Simon work—was it our place to correct in our brief tour through previous work the unjustified omission of Simon’s work by others? We found a right word to hint that this had transpired.
When we sent a preprint to a colleague whose work we had mentioned, a prime researcher in the field, he told us straight up that he was unhappy that we had not mentioned that he was the first to mention the electride concept in the high pressure literature. Fortunately, his complaint came as we were reading proof. We fixed it, went to second proof. At this point, by tracing a reference we should have followed earlier, we found another article in which the idea of an interstitial electron density was forwarded by a still different group, prior to the complainant. The matter was not expressed in direct chemical language, i.e., the entity was not called an electride by name, but it was clear to me they had the idea. That too had to go in.
I stopped at third proof. And thank you, ACS Publications Department, for not billing us for the work we created via the extra galleys. It is not that we did a poor literature job to begin with; my collaborator in this work probably got tired (or at least expressed some surprise) at the time I put in to go through the literature. It is just that the literature is large, and the chains of antecedents almost divergent. And there are always forerunners, partial understandings, and inklings. Let there be no doubt: one should bend over backwards to cite these.
At times I have wondered about the peer review process. So much time spent by the community, so much aggravation generated for the authors, all to either occasion a revision, or, at the extreme, keep out a few poor articles from being published. After all, we train our students to tell the difference between a routine article and a good one. But I am now convinced that peer review is important for many reasons—most for the intangible feeling by an expert that this work makes sense, and is important or is routine, or worse (Hudlický, Citation2005; Carlson and Hudlický, Citation2012). And for making sure that the illustrations communicate—that, sadly, is where many authors fail. And, relevant to this section, that others’ work is cited. To do otherwise, not to cite relevant work, especially if it disagrees with your own theories, violates no laws, but is destructive of the underlying ethics of our scientific microsociety. Reviewers are the front line on ethics of citation.
Creative Tension
The idea that a certain amount of tension is conducive to creation is commonplace. One finds it often expressed in the inspirational literature of management. The usual prescription is that by articulating a vision, and contrasting it with reality, one formulates more clearly a goal, in whose determined pursuit creativity is unleashed.
The vision may be aspirational, of the heart as much as it is of the mind, as in Martin Luther King’s Citation1963 “I have a Dream” speech, which in part reads as follows:
I have a dream that my four little children will one day live in a nation where they will not be judged by the color of their skin but by the content of their character. I have a dream … I have a dream that one day down in Alabama, with its vicious racists, with its governor having his lips dripping with the words of “interposition” and “nullification,” that one day right there in Alabama little black boys and black girls will be able to join hands with little white boys and white girls as sisters and brothers. (King, Citation1963)
Or it may be direct, and chemical, as in the goal posed to U.S. automotive engineers and scientists by California’s emission controls in the sixties.
King’s voicing of a challenge to the United States, not just Alabama, has been met, but only in part. And catalytic chemists in Detroit, their bosses crying to high heaven that it could not possibly be done, came up with the remarkable three-way-catalyst, reducing exhaust unburned hydrocarbons, CO, and NOx by factors of 10 to 40 between 1966 and 1993 (Calvert et al., Citation1993).
That is inspiration. Is there any evidence in the psychological literature for creativity enhancement under tension? A place to start is with the Yerkes–Dodson law, the recognition that there is a dome-shaped relationship between arousal and optimal performance on a variety of physical and intellectual tasks (Yerkes and Dodson, Citation1908). The hypothalamic-pituitary-adrenal system reacts to stress (read perceived tension) by releasing corticosteroids, epinephrine, and, secondarily, neurotransmitters. There is an initial increase in performance with stress level, and then a leveling off, followed by a decrease. So … some tension is good; too much is definitely not.
At the same time, there is a world of writing that recognizes the reality or potential of growth through extreme suffering, stress that no human being should experience. My immediate reaction, as a childhood survivor of the Holocaust, is that much of this is ex post facto romanticization. But one cannot deny the first-hand, honest accounts of many whose lives have been changed by traumatic events. Tedeschi and Calhoun provide a sensitive analysis of the phenomenon in their article “Posttraumatic Growth: Conceptual Foundations and Empirical Evidence” (Tedeschi and Calhoun, Citation2004; see also Park and Helgeson, Citation2006). I quote from their conclusion:
[Posttraumatic growth] is likely to involve a powerful combination of demand for emotional relief and cognitive clarity, that is achieved through construction of higher order schemas that allow for appreciation of paradox. Metaphorical and narrative elements are likely to serve trauma survivors well as they take on a life that has become surprising, complicated beyond expectation, and painful. (Tedeschi and Calhoun, Citation2004; Park and Helgeson, Citation2006)
I range too far afield from what I seek—a connection between tension, a stressor, and scientific creativity. To return in stages, consider first the work of Hans Selye, the prolific Hungarian-Canadian endocrinologist, who identified “eustress,” literally “good stress.” Selye documented the way our bodies cope with stress, the hormones and organs involved. The response to tension, he argues, is not only negative, distress, but may be positive (Selye, Citation1976). An example might be the reward system I experience on going longer up and down our Ithaca hills on my bicycle than I ever did before. But it also may be the satisfaction of seeing B,N substitution on a naphthalene skeleton work to meet a criterion for singlet fission (no, not a new way of asexual reproduction).
Selye’s perception of the potential responses of an individual to a tension-producing stimulus by eustress or distress puts matters in a somewhat different light (LeFevre et al., Citation2003). There appears to be a growing literature on the positive psychological effects of stressors. I found very useful the rethinking of stress by Crum, Salovey, and Achor (Crum et al., Citation2013), which emphasizes the role of mindsets (that can be modified) in the formation of a “stress-is-enhancing” response. Previous studies have shown that there may be an enhancement of physical thriving in response to stress (Epel et al., Citation1998), and in taking personal initiative (Fay and Sonnentag, Citation2002). There is also some evidence in organizational psychology that individuals experiencing ambivalence of an emotional sort (feeling simultaneously positive and negative emotions) recognize better unusual relationships between concepts, and are more sensitive to associations (Fong, Citation2006). Recognizing relationships, forming associations—that is a lot of what the best scientists do.
I found especially informative an account by Mark A. Runco of the ways in which “disturbance and anxiety can facilitate creative effort” (Runco, Citation1994; see also Smith and van der Meer, Citation1994). Runco reviews a variety of psychological studies that point to a moderate amount of stress in a way fertilizing creativity. Important guidelines here are provided by two great 20th century developmental psychologists, Lev Vygotsky and Jean Piaget. Vygotsky described creativity as “a benefit of conflict between realistic and imaginative conditions” (Runco, Citation1994, p. 112; Smolucha, Citation1992); and Piaget had an imaginative theory of psychic disequilibrium between what experience offers and the cognitive structures in our mind (and our imagination) proffer.
Let me be parochial: I have a schema of frontier orbital thinking in my mind; this ornery molecule behaves opposite to my “prediction.” This does not feel good, for I want to understand. I look for an explanation, of course: I want to reach the point where I could kick myself in the behind for not having seen it. That is a theory of theory formation.
Runco makes an important point, that tension can be “both a cause and an effect of creativity” (Runco, Citation1994, p. 119).
There is also a substantive literature on cognitive dissonance, i.e., how one deals with incompatible observations or sensations (Festinger, Citation1962; Aronson, Citation1995). Denial and self-delusion are one negative outcome. Creative adjustment is a positive one—I view Bohr’s complementarity principle as such. Bohr said that we are obliged to describe phenomena in ways that may seem contradictory (e.g., the particle and wave nature of light), always aware that these descriptions are tied to specific experiments that force those seemingly incompatible interpretations.
The history of chemistry has a number of such productive adjustments. Consider the progression from 19th century lines in chemical formulas, symbols of a chemically manipulable replacement of one atom/functional group for another, to G. N. Lewis’s shared electron pair, then reinterpreted by Linus Pauling as a quantum mechanical valence bond. The sequence of these representations is hardly an example of a Kuhnian revolution. These world views of the chemical bond were grafts of one idea onto another, by such smart people. They improvised, sans malice—to escape the imprisoning dichotomous world of the either/or. Almost seamless, these appropriations of a previously existing concept (the chemical bond) were carried out with the faith that the world is one underneath, and that one day there will come a new way of seeing that will reconcile seemingly irreconcilable perspectives. I see it also in the establishment of the equivalence at a higher level of approximation of simple valence bond and molecular orbital pictures of the chemical bond, due to Charles Coulson.
Finally, I see a piece of the relationship between tension and creation in Blume Zeigarnik’s older studies, not undisputed, of people remembering better unresolved activities (Baumeister and Bushman, Citation2008), or remembering them in a different way. Most of us have had the experience of an unsolved problem loitering, so to speak, in our mind, and then surfacing, unexpectedly, to be solved or resolved, at some later date, even in a dream.
And Excessive …
I began this article writing “Most of us do not view tension positively.” The previous section could be brought up as evidence that I see the world through rosy glasses. So I would like to reiterate here the multitude of ways in which the excessive tensions of the modern scientific world cause “distress,” to use Selye’s term (Selye, Citation1976).
We have, for instance created in the U.S. chemistry (and not just chemistry) an incredible machine for innovation, the Assistant Professorship. At great psychological cost—a young man or woman has to learn to teach, to attract graduate students, to do first class research, to “sell” that research and be “seen,” to serve his university and profession to get research funding, all while beginning a family. I think one of the reasons the increasing pool of Ph.D. women chemists moves disproportionately more into the industrial rather than the academic workplace is just because of the inhumane tensions of our innovation machine.
A special tension of the great recession out of which we are working our way is that in a cure, spurring employment, American Recovery and Reinvestment Act funds increased the research pot for academic chemists. But at the same time, private universities lost parts of their endowment, and state budgets were constricted. The outcome was a 5–15% decrease in education budgets. Research up, education down, and no way to shift funds from one category to the other (Hoffmann, Citation2009).
One disease of the biomedical profession we have avoided in academic chemistry is that we do not have to raise our salaries from our research grants, as people in our wonderful medical schools do. Nevertheless, the pressure to get research grants, and to publish in prestigious journals is great, and in its own way, destructive of the spirit.
The Tense Middle
I return to equilibrium, and the idea of a tense, productive middle (Hoffmann, Citation2008). Productive, because it is tense. The middle is interesting. It may not be what the world wants, least of all what journalists (and that includes NPR journalists) want. “Is that hurricane the worst you’ve experienced?” Strong opinions, extremes make a good story. And … for the teller, the extreme is a haven, where the water is calm, where your back is secure against a wall.
What chemical equilibrium teaches me is that the middle is not static, but dynamic. And so it is tense. The middle has the potential; I have the potential; you have the chance—of going one way or another.
I like that. Yes, I also want stability. But I believe that extreme positions—all reactants, all products, all people A bad, all people B good, no taxes at all, taxed to death—are impractical, unnatural, boring, the refuge of people who never want to change. The world is not simple, though, God knows, political ads (on every side) want to make it so. I like the tense middle, and am grateful for a world that offers me the potential for change. Chemistry does.
ACKNOWLEDGMENT
I am grateful to Deena Weisberg for steering me to Alia Crum’s research on mindsets, and to Jeff Seeman for some guidance.
FUNDING
The author thanks the Innovative Grant Program (IGP) of the American Chemical Society; the IGP has provided the funds that permit permanent open access to this article and all articles in this special issue of Accountability in Research, titled, “Ethics and Responsible Conduct of Research within the Chemical Community. Ideas and Experiences Worth Sharing.” Over the years, the National Science Foundation has supported my chemical research, from which these reflections have come.
REFERENCES
- Abruña, H. D. 2006. Peter Debye. Chemical & Engineering News, 84:4–5.
- Aronson, E. 1995. The social animal. New York, NY: W.H. Freeman and Co.
- Baumeister, R. F., and B. J. Bushman. 2008. Social psychology and human nature. 122. United States: Thompson Wadsworth.
- Berke, H. 2002. Chemistry in ancient times: The development of blue and purple pigments. Angewandte Chemie, International Edition 41(28):2483–2487.
- Calvert, J. G., J. B. Heywood, J. F. Sawyer, and J. H. Seinfeld. 1993. Achieving acceptable air quality: Some reflections on controlling vehicle emissions Science, 261:37–45.
- Carlson, R., and T. Hudlicky. 2012. On hype, malpractice, and scientific misconduct in organic synthesis. Helvetica Chimica Acta 95:2052–2062.
- Coppola, B. P. 2001. The technology transfer dilemma: Preserving morally responsible education in a utilitarian entrepreneurial academic culture. HYLE: International Journal for the Philosophy of Chemistry 7:155-167.
- Crum, A. J., P. Salovey, and S. Achor. 2013. Rethinking stress: The role of mindsets in determining the stress response. Journal of Personality and Social Psychology 104:716–733.
- Epel, E. S., B. S. McEwen, and J. R. Ickovics. 1998. Embodying psychological thriving: Physical thriving in response to stress. Journal of Social Issues 54:301–322.
- Estado de S. Paulo. 1994, and a Brazilian thalidomide victims association, A.B.P.S.T., http://www.talidomida.org.br/. (accessed May 20, 1994)
- Everts, S. 2013. Middle East meeting of scientific minds. Chemical & Engineering News 91:59–61.
- Fay, D., and S. Sonnentag. 2002. Rethinking the effects of stressors: A longitudinal study on personal initiative. Journal of Occupational Health Psychology 7:221–234.
- Festinger, L. 1962. Cognitive dissonance. Scientific American, 207:93–107.
- Fong, C. T. 2006. The effects of emotional ambivalence on creativity. Academy of Management Journal 49:1016–1030.
- Hoffmann, R. 1995. The Same and Not the Same. New York, NY: Columbia University Press.
- Hoffmann, R. 2004a. A claim on the development of the frontier orbital explanation of electrocyclic reactions. Angewandte Chemie, International Edition 43:6586–6590.
- Hoffmann, R. 2004b. Meissen chymistry. American Scientist 92:312–315.
- Hoffmann, R. 2006. Peter Debye. Chemical & Engineering News 84:4–6,39.
- Hoffmann, R. 2008. The tense middle. In This I Believe II, ed. J. Allison and D. Gediman, 112–114. New York, NY: Henry Holt.
- Hoffmann, R. 2009. Cost cuts should come from research, not just education, Chronicle of Higher Education, Commentary. 55:A26.
- Hoffmann, R. 2014. Maltese reflections. Chemical & Engineering News 91:5.
- House M. C., and J. I Seeman. 2010. Credit and authorship practices. Educational and environmental influences. Accounts of Chemical Research 17:223–256.
- Hudlický, T. 2005. What a novel idea. Chemical & Engineering News 83:6.
- King, M. L. 1963. In A Call to Conscience: The Landmark Speeches of Dr. Martin Luther King, Jr. Carson, C., Shepard, K., eds. New York, NY: IPM/Warner Books, 2001.
- Kovac, J. 2003. The Ethical Chemist: Professionalism and Ethics in Science. Upper Saddle River, NJ: Pearson Prentice Hall.
- Kovac, J., and B. P. Coppola. 2000. Universities as moral communities. Soundings: An Interdisciplinary Journal 83:765–779.
- Kovac, J., and M. Weisberg. 2012. Roald Hoffmann on the philosophy, art and science of chemistry. New York, NY: Oxford University Press.
- Le Fevre, M., J. Matheny, and G. S. Kolt. 2003. Eustress, distress, and interpretation in occupational stress. Journal of Managerial Psychology 18:726–744.
- Merton, R. K. 1968. Social theory and Social Structure. Enlarged edition. New York, NY: Free Press.
- Miao, M., and R. Hoffmann. 2014. High pressure electrides, Accounts of Chemical Research 47:1311–1317.
- Moreau, N. 2010, private communication, 15 August 2010.
- Park, C. L., and V. S. Helgeson. 2006. Introduction to the special section: Growth following highly stressful life events—current status and future, Journal of Consulting and Clinical Psychology 74: 791–796.
- Poirier, J. - P. 1996. Lavoisier, Chemist, Biologist, Economist. Translated by R. Balinski. Philadelphia, PA: University of Pennsylvania Press.
- Rouhi, A. M. 2010. Eurasian conference begins confidently, Chemical & Engineering News 88:11.
- Runco, M. A. 1994. Creativity and its discontents. In Creativity and Affect, ed. Shaw, P. P. and M. A Runco, 104–125. Norwood: Apex.
- Seeman, J. I., and M. C .House. 2010a. Influences on authorship issues. An evaluation of giving credit. Accounts of Chemical Research 17: 146–169.
- Seeman, J. I., and M. C House. 2010b. Influences on authorship issues: An evaluation of receiving, not receiving, and rejecting credit. Accountability in Research: Policies and Quality Assurance 17: 176–197.
- Selye, H. 1976. The Stress of Life. Revised Edition. New York, NY: McGraw-Hill.
- Smith, G. J. W., and G. van der Meer. 1994. Generative Sources of Creative Functioning. In Creativity and Affect, ed. Shaw, P. P. and M. A. Runco. 147–167. Norwood: Apex.
- Smolucha, F. 1992. The relevance of Vygotsky’s theory for research on creativity and play, Creativity Research Journal 5: 8–67.
- Tedeschi, R. G., and L. G.Calhoun. 2004. Posttraumatic growth: Conceptual foundations and empirical evidence. Psychological Inquiry 15:1–18.
- Yerkes, R. M., and J. D. Dodson. 1908. The relationship of strength of stimulus to rapidity of habit-formation. Journal of Comparative Neurology and Psychology 18:459–482.