ABSTRACT
Worm predation (WP) on activated sludge leads to increased sludge degradation rates, irrespective of the type of worm used or reactor conditions employed. However, the cause of the increased sludge degradation rates remains unknown. This paper presents a comparative analysis of the physical and biochemical aspects of predated sludge, providing insight into the hydrolytic mechanisms underlying WP. To this end, the sessile worm Tubifex tubifex was used as a model oligochaete and was batch cultivated in an 18-L airlift reactor. Predation on activated sludge showed an average reduction rate of 12 ± 3.8%/d versus 2 ± 1.3%/d for endogenous respirated sludge. Sludge predation resulted in an increased release of inorganic nitrogen, phosphate and soluble chemical oxygen demand (sCOD). The sCOD consisted mainly of polysaccharides; however, fluorescence excitation emission matrix spectroscopy analysis also revealed the presence of Tryptophan-protein-like substances. Results suggest that the released polysaccharides contain a protein-like element. Additionally, soluble iron increased slightly in concentration after WP. The extent of hydrolysis seemed to reach an average plateau of about 40% volatile solids (VS) reduction after 4 days, which is substantially higher than the 29% VS reduction for endogenous decay of activated sludge after 30 days. Furthermore, T. tubifex predominantly consumed the protein fraction of the extracellular polymeric substances. Results suggest that that the worms specifically target a fraction of the sludge that is predominantly biodegradable under aerobic conditions, albeit at significantly higher degradation rates when compared to the endogenous decay of waste activated sludge.
Highlights
Worm predation resulted in an increased sludge reduction rate.
The protein fraction of the sludge was primarily removed.
Soluble COD, inorganic nitrogen and phosphorus were released.
The soluble COD consisted of Tryptophan-protein-like substances.
Results suggest that primarily the aerobically degradable part of sludge is removed.
1. Introduction
Waste activated sludge (WAS) is a by-product of the aerobic treatment of sewage and industrial wastewater. Approximately 10 million ton of WAS is produced annually in Europe [Citation1] (2007 estimate) and has to be disposed of according to the Council Directive of the Commission of European Communities [Citation2]. Sludge disposal has been estimated to account for 50% of the total costs for waste water treatment plants (WWTPs) [Citation3]. To lower these disposal costs, anaerobic digestion is used to reduce the amount of excess sludge and partially recover the biochemical energy stored in sludge as biogas. However, anaerobic digestion removes only about 30–40% of the organic component of biomass, which means that a large amount of undigested solids still needs costly processing.
These costs have stimulated many research projects that aim to minimize the amount of WAS that has to be disposed. Some methods are aimed at increasing the rate and extent of hydrolysis during anaerobic digestion by physical and/or chemical methods or enzymatic pre-treatment of the sludge [Citation4–6]. Other methods are aimed at reducing excess sludge production by increasing the sludge age [Citation7–9] and thus increasing cell lyses and cryptic growth mechanics [Citation10], which in turn results in a decrease in sludge production.
A special case of excess sludge reduction is by means of aquatic worms that naturally inhabit aerobic zones in WWTPs [Citation11]. Several different sessile and free swimming oligochaete worm species have been investigated with a variety of reactor designs for their ability to degrade sludge (see ).
Table 1. Summary of oligochaete worm-related research: worm species, reactor design and important findings.
As shown in , not all studies included appropriate control experiments, which are needed to properly relate worm predation (WP) to the change in sludge characteristics. However regardless of the variability in the different experiments, it is clear that the observed changes in sludge after WP have been similar, irrespective of the worm species and/or reactor setup used. Overall these changes include increased sludge degradation accompanied by the release of mineralization products, an increased in soluble chemical oxygen demand (sCOD), improved settling characteristics and the removal of proteins. These similarities suggest a common mechanism of sludge hydrolysis employed by aquatic worms.
Worm characteristics that could influence hydrolysis rates are peristaltic movement and bioturbation [Citation26–28], although the latter is expected to be of minor importance in a turbulent environment such as a WWTP. It has also been suggested that oligochaete worms consume bacteria [Citation29–32] and change the microbial diversity in natural sediments [Citation30] or the sludge [Citation33] that the aquatic worms inhabit.
The mechanisms that worms use for hydrolysis deserve further attention because hydrolysis is considered to be the rate-limiting step in the degradation of excess sludge [Citation34,Citation35], and sludge predation increases this degradation rate. Even though knowledge regarding WP is steadily increasing, the underlying hydrolytic mechanism of predation has not been investigated specifically. The majority of research concerning aquatic worms has focused on implementation for sludge minimization with the worms depicted as a black-box model for sludge degradation or membrane fouling mitigation (). As a consequence, a complete data set to adequately research WP mechanics is lacking.
By using controlled reactor conditions, this paper presents a comparative analysis of the physical and biochemical components of initial feed WAS, worm predated sludge and endogenously respired sludge. By means of this method, a clear distinction can be made between the effects of the reactor conditions and sludge decay, on the one hand, and WP, on the other hand. With this analysis, further insights can be gained into the hydrolysis mechanics of aquatic worms as well as the general aspects of WP.
2. Materials and methods
2.1. Lab-scale worm reactor
In this study, the sessile worm T. tubifex was chosen as a model oligochaete worm. The aquatic worms were batch-cultivated in a lab-scale reactor and fed with WAS obtained from WWTP Harnaschpolder (Den Hoorn, The Netherlands), which treats municipal wastewater of 1.3 million population equivalents. The reactor was designed as a modified lab-scale version of the full-scale worm reactor that was used by Tamis et al. [Citation21]. The reactor is composed of two identical 18 L compartments. One compartment was used as control to evaluate the conversion due to endogenous respiration (ER) and the effect of applying extended aeration on the structure of WAS. The second compartment was used for WP and contained approximately 40 ± 6 g/L wet weight worms. The amount of worms used was sufficient to make a clear distinction in volatile solids (VS) reduction between the endogenous respirated sludge and worm predated sludge (WP). The worms used in the experiments did not always originate from the same shipment of worms.
The worms were stored in an aerated vessel fed with WAS when not used in experiment. The intestines of the worms contain consumed sludge. Worms were not gut purged at the start of an experiment. This was done to compensate for the sludge solids that were ingested during experiments. These ingested solids would consequently result in an unwanted decrease in sludge solids at the end of an experiment when worms with purged guts would have been used. Both compartments were aerated and mixed by using an airlift system. The average dissolved oxygen concentration was ≥ 5 mg/L, as recommended by Cai et al. [Citation36] and the temperature was maintained at 20 ± 1°C. The pH was left unaltered and was 7.4 ± 0.2 on average. Distilled water was used as make up water in case of evaporation.
2.2. Extended aeration
The ER sludge was transferred from the reactor to an aerated 5 L glass bottle. The sludge was aerated for an additional 30 days (ER-30) at room temperature.
2.3. T. tubifex
T. tubifex was bought at a local wholesales (Aquadip B.V. and De Maanvis B.V., The Netherlands). Upon arrival, the worms were thoroughly rinsed to remove dead specimens and other contaminants. Worms were then transferred to and stored in the aforementioned aerated vessel containing WAS. Fresh worms were stored for at least 1 week in order to adapt to the sludge, before use.
It was observed that clumps of worms tend to concentrate sludge particles around their bodies. To remove these particles, the worms were thoroughly rinsed with tap water in a large beaker. In the beaker, a vortex was created by hand to remove attached particles. The worms were then left to settle for ±1 minute and the upper water phase was discarded. This process was repeated until the water layer was clear and the worms were clean. Worms were spread out and weighed, after most of the adhered water was removed using paper tissues.
2.4. Analytical methods
Total solids (TS) and VS were measured in triplicate. For dissolved compounds determination, mixed liquor samples were filtered over 0.45 µm glass fibre membrane filters prior to analysis. Dissolved nitrate, ammonia and phosphate were measured in duplicate, while COD was measured in triplicate, using Spectroquant photometric test kits (Merck Millipore, Darmstadt, Germany). Analytical methods were in accordance with the standard methods [Citation37].
2.5. Sludge dewaterability
The SVI, the zone settling velocity (ZSV) and capillary suction time (CST) were determined according to the standard methods [Citation37]. For the turbidity analysis, the sludge was left to settle for 30 min and subsequently, the water phase was analysed with a HACH 2100 N Turbidimeter (Hach, Loveland – Colorado, USA). CST measurements were performed using a Type 304 M CST apparatus (Triton Electronics Ltd., Essex, England).
2.6. Particle size distribution
The particle size distribution (PSD) was performed using liquid particle counting device (model HIAC 9703 Hach, Loveland – Colorado, USA) equipped with a HRLD400/HC sensor (Hach Ultra, Grants Pass – Oregon, USA). The operational principle is based on the light-blockage method in the size range of 2–400 µm. Particles are counted in fixed size increments of 0.78 µm. The flow was set to 100 mL/min. Samples were diluted 1000× in demineralized water and subsequently sieved (400 µm mesh) prior to analysis. The presented data were not multiplied with the dilution factor. Data were recorded with WGS Software (version 2.4), which was supplied with the particle counting device, and analysed by using the normalized data, as recommended by APHA [Citation37].
Particle counting data require to be normalized to avoid apparent peaks, due to the variability of the size increments [Citation37]. The obtained normalized PSD was then compared with the theoretically defined PSD of Lawler [Citation38]. A PSD can be presented as a power-law function where the slope of the distribution, referred to as β in literature, can either be constant or variable according to theoretical models.
Normalization of the data consists of dividing the particle count, in a given size range, by the size interval and presenting the data on a logarithmic scale. An example of the difference between raw counts and normalized data is given in Figure S1 (supplementary information section).
2.7. sCOD: quantitative protein and carbohydrate determination
Dissolved carbohydrate-like and protein-like substances in the sCOD fraction were obtained by filtering the mixed liquor using 0.45-µm glass fibre filters. The sCOD fraction was then quantified by using the phenol-sulphuric acid method for carbohydrate determination, with D-glucose as a standard [Citation39]. For protein determination, the Lowry method [Citation40], with Bovine Serum Albumin (BSA) as a standard, was used. The classical Lowry method was chosen, instead of the modified version of Frølund et al. [Citation41], due to the low absorbance values (absorption around 0.1) as proposed by Avella et al. [Citation42].
Within the modified Lowry method, the measured absorbance without using the CuSO4 reagent is a measure for interfering substances for the protein determination. These interfering substances are ascribed to the humic-like fraction [Citation41]. However, it remains questionable whether these interfering compounds are indeed exclusively humic substances. When interfering humic-like substances were intended to be measured, the modified Lowry method [Citation41] was used with humic-acid sodium salts (H16752) as a standard. All the reagents were purchased at Sigma-Aldrich.
2.8. sCOD qualitative: fluorescence excitation emission matrix spectroscopy
Fluorescence excitation emission matrix spectroscopy (FEEMS) can be used to probe the composition, concentration and dynamics of organic matter from various source materials [Citation43–45]. Sludge mixed liquor samples were filtered over 0.2-µm glass fibre filters and stored at 4°C in glass vials prior to analysis. FEEMS were measured on a Horiba Jobin Yvon Fluoromax-4 Spectrofluorometer equipped with a Xenon lamp light source and a 1-cm path length quartz cuvette, following D’Andrilli et al. [Citation46].
Samples were analysed for UV absorbance with a Thermo Scientific Genesys 10 scanning UV spectrophotometer with a 1-cm path length, from 190 to 1100 nm on optically dilutes samples (absorbance values <0.3 at 254 nm). Samples with absorbance values >0.3 at 254 nm were diluted with nanopure water until they were below 0.3 in order to reduce inner filter effects during post processing of the FEEMS [Citation44].
Post-processing of the fluorescence data was completed in MATLAB to generate 3D FEEMS data, which included sample corrections for inner filter effects, Raman scattering and blank water subtraction. Positions and intensities (Excitation and Emission maxima values) for individual fluorophores were determined to gain more information on the composition of the material. Samples were compared to each other for the different wavelength regions. These regions are related to the composition of different substances ().
Table 2. FEEMS excitation and emission wave length regions with the associated substances.
2.9. Divalent and trivalent cations
Total and soluble Al3+, Ca2+, Mg2+, Fe3+ and Na+ were determined using the digestion method described by van Langerak et al. [Citation49]. Samples were analysed using an ICP-MS Xseries II (Thermo Fisher Scientific Carlsbad – California, USA) except soluble Al3+, which was determined using Spectroquant photometric test kits (Merck Millipore, Darmstadt, Germany). Cation measurements were performed in triplicate.
2.10. Extraction of EPS and ALE
Extracellular polymeric substances (EPS) were extracted according to the method used by Frølund et al. [Citation50], using Dowex marathon C cation exchange resin. Extraction was carried out with magnetic stirring at 350 rpm for 17 h at 4°C. An amount of 0.5 g sludge was used per extraction. The EPS extracts were analysed for protein- and humic-like content using the modified Lowry method as proposed by Frølund et al. [Citation41] with BSA and humic sodium salts (H16752) as standards. Carbohydrate content was determined using the aforementioned method. Reagents were obtained from Sigma-Aldrich.
Alginate-like exopolysaccharides (ALE) were extracted using the method described by Lin et al. [Citation51]. About 1 L of feed and processed sludge was used in the extraction. Extractions were performed in triplicate. After extraction, the supernatant was obtained by centrifugation (3500 RCF, room temperature, 20 min) and subsequently filtered over a 0.45-µm glass fibre membrane filter to obtain the ALE extract. The carbohydrate content was determined using the method described earlier.
3. Results and discussion
3.1. Taxonomy of worms
To make sure that the worms used in the experiments were indeed of the tubificid genus, the taxus of 100 individuals were determined. Almost all individuals were of the Tubifex genus with a sporadic presence of L. variegatus. Reproductive organs were not observed. Egg sacs attached to the worms were observed as white/pink perturbations on the segments of the worms. Dispersed cocoons (white/pink) were found throughout the sludge (Supplementary Information Figure S2). External stimuli resulted in ‘curling up’ of the worms, which is distinctly different compared to its similar looking counterpart L. variegatus, who shows a ‘corkscrew’ escape movement. The apparent healthiness of the worms was visually assessed by evaluating the response of a clump of worms after touching. A healthy response is the formation of a firm clump. If the worms were not healthy, the clump was fluffy with an open structure.
3.2. Sludge degradation and physical characteristics
The difference in VS reduction between WP sludge and ER sludge was researched using batch incubations of 4 days. In agreement with the recommendations by Buys et al. [Citation52] regarding WP, approximately 45 g/L wet weight worms were used, which was indeed sufficient to give a clear distinction between the WP sludge and the ER sludge within the duration of the batch experiment.
The presence of T. tubifex during aerobic stabilization of WAS had a significant impact on the extent and rate of WAS degradation ().
Table 3. Solids reduction of activated sludge for worm predated (WP) and endogenous respirated (ER) sludges.
On average, 47% ± 15 of the initially present VS was converted upon WP versus 9% for the endogenous respired sludge. The corresponding averaged VS reduction rates were 12%/d and 2%/d for WP and ER, respectively. Interestingly, extended aeration of WAS for duration of 30 days (ER-30) resulted in a similar 29% ± 1.9 reductions in VS. The large difference in incubation time clearly demonstrates the increased VS reduction rate in the presence of worms.
The VS reduction was accompanied by an increased release rate of the soluble organic fraction (sCOD) and inorganic material, predominantly NH4+-N, NO3--N and PO43--P, as is presented in .
Table 4. Average rates for WP and ER.
These increased release rates upon WP are in line with other studies. For example, Hendrickx et al. [Citation16] found similar values for nitrogen and phosphorus compounds, with 55 mg N/g TSS removed and 17 mg P/g TSS removed versus 30.8 ± 17.6 mg N/g TS removed and 10.5 ± 5.7 mg P/g TS removed in this study. Worm-specific removal rates can be found in the Supplementary Information Table S1.
3.2.1. Changes in EPS upon WP
The changes in EPS composition before and after treatment are shown in .
Table 5. EPS extraction data of waste activated (WAS), endogenous respirated (ER) and worm predated (WP) sludges, after a 4-day batch incubation.
The high release of inorganic nitrogen was accompanied by a large decrease in the protein fraction of the EPS of worm-predated sludge. In contrast, the protein and carbohydrate fraction of WAS and ER remained similar. The EPS-carbohydrate component of WP sludge also decreased, but to a lesser extent than the protein fraction. The increased N release coupled with a decrease in the protein EPS fraction indicates that the aquatic worms predominantly target the protein fraction of the polymers in the sludge. These results are in line with the results of Hendrickx et al. [Citation15], who reported a 35% decrease in worm predated sludge’s nitrogen content.
The humic-like fraction remained relatively stable upon ER and WP treatment (), reflecting the inert behaviour of humic-like substances. Although within the error margins, the average humic fraction slightly increased. This increase might be well ascribed to an increased extraction efficiency, due to an increase in the number of small particles (which will be further discussed in Section 3.2.3). The average humic-like fraction was slightly higher in ER compared to WP, which might suggest that part of the humic-like substances was removed or altered.
Electrostatic interaction between humic-like compounds and proteins have been reported by multiple authors [Citation53–55]. They showed that electrostatic interactions are responsible for protein and humic substance complexation. Additionally, humic substances can contain protein-like elements [Citation56–58]. Shan et al. [Citation59] showed the removal of protein-like elements during vermicomposting, using 14C-labelled proteinaceous components bound to the humic substances. Possibly, during WP, a similar disruption of the electrostatic interactions and subsequent conversion of the humic-protein complexes occurred.
3.2.2. Changes in ALE upon WP
As alginate is an important structural component in (granulated) activated sludge [Citation51], the ALE fractions of the sludge, before and after pre-treatment, were compared (). The amount of ALE that could be extracted from WAS and ER were in a similar range, i.e. 72 ± 6 mg/gVSS, as was found by Lin et al. [Citation60] for suspended activated sludge. ALE concentrations increased for ER and decreased for WP compared to WAS. It seems that aeration and the associated shear forces resulted in smaller particles with a larger total surface area, thus increasing the extractability of ALE.
Table 6. ALE polysaccharide concentrations for WAS, endogenous respirated (ER) and worm predated (WP) sludges after 4 day batch incubation of 2 separate batches.
Irrespective of the increased extractability due to aeration, the concentration of ALE extracted from worm-predated sludge decreased by roughly 40%. Around 62 ± 15 mg ALE per gram of degraded sludge was removed in the presence of worms. It seems that the worms consume part of the extractable ALE. Whether these extracted carbohydrates contain a protein element, which would support the reduced EPS protein fraction, is unknown, as the protein component of the ALE extract was not measured.
3.2.3. PSD and turbidity
The effect of aeration and predation on the PSD of the treated sludge is shown in . It can be seen that the number of large particles, in the range of 30–200 µm, decreased upon treatment of WAS. Shear forces introduced by aeration and sludge decay (e.g. endogenous respiration) are known to break up sludge flocs in smaller particles [Citation61,Citation62]. The difference between ER-30 and the other samples mostly reflects the effect of long-term aeration on particle size reduction. The difference in the number of particles, between WAS, on the one hand, and ER and WP, on the other, reflects the difference in VS removal because both samples were maintained under the same aeration conditions.
Figure 1. Normalized PSD of treated sludges, showing the averages of triplicate measurements. Graph split in two parts: (a) 30–200 µm; (b) 2–30 µm. Error bars are omitted for clarity. Horizontal line around 0.5 counts/mL*µm is due to low particle counts (1–2 counts/mL) in the measurements. The outlier at 1.6 µm* is probably due to a fault in the machine as it is consistent in all the measurements. Particles larger than 200 µm were not observed in the sludges. Batch VS reduction was for WP 42%, ER 18% and ER-30 29%. For visualization, the values plotted were not multiplied by the dilution factor.
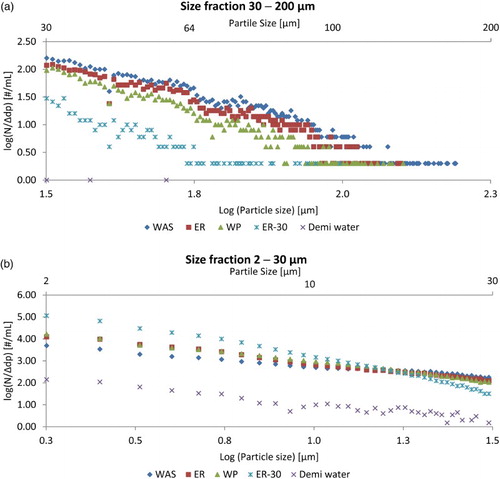
The breakdown of large particles results in an increase in the 2- to 30-µm range, when comparing the samples to WAS ((b)). This increase can be clearly seen for the PSD of ER-30, which shows more small particles and fewer big flocs. Extended aeration clearly breaks up sludge flocs predominantly by prolonged exposure due to mechanical shear. ER and WP were aerated and thus exposed to the same mechanical shear for the same amount of time; the PSD of ER and WP almost overlap and are clearly different from the PSD of WAS, showing higher amounts of small particles.
The original WAS is altered by the activity of the worms that apparently reduced the size of the flocs ((a)) due to their degradation activities, and simultaneously produced a higher amount of small particles (Figure S3 Supplementary information). However, the specific size fraction that the worms consume and excrete cannot be determined from the data due to the large effect that aeration has on floc size. In future work this could be compared to sludge treated in a passively aerated environment.
The increase in smaller particles also becomes apparent in the turbidity measurements presented in and the normalized PSD of the supernatant of settled sludge shown in Figure S3 (Supplementary information).
Table 7. Turbidity and triplicate particle counting measurements of water phase of 30 min settled waste activated (WAS), endogenous respirated (ER), worm predated (WP), 30-day extended aerated (ER-30) sludges and demineralized water (demi water).
It can be seen that with increasing aeration times, the turbidity increases with the amount of particles in the 2- to 30-µm size fraction (Figure S3). Furthermore, the presence of worms increases the number of small particles compared to ER. Figure S4 (Supplementary Information) visually shows the difference in turbidity and settleability between the sludges.
3.2.4. Dewaterability and settleability
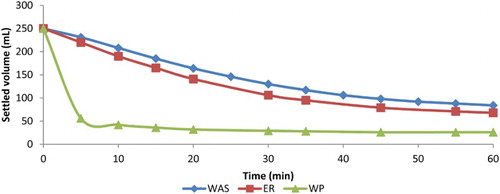
The change in SVI over the duration of the batch assay can be seen in . It is clear that WP improved the settleability of the sludge. The SVI drops to roughly 50% of its starting value for WP, whereas the SVI for ER slowly decreased. To provide information on the velocity at which sludge settles, the sludge blanket volume was monitored over time (). The results show that WP settles almost completely in the first 5 min while WAS and ER needed 60 min to reach similar volumes. On the basis of the data from , the ZSV was calculated. The velocity increased from 0.248 m/h to 0.332 m/h and 2.29 m/h for WAS, ER and WP, respectively, which reflects the improved settling properties of WP sludge.
Figure 2. Change in average SVI over the duration of the batch for endogenous respirated (ER) and worm predated (WP) sludges. Averaged values of 4 batches. Error bars represent standard deviations. Averaged VS reduction percentages were 40% ± 16 and 8% ± 3 for WP and ER, respectively. The data point at day 4 was from one batch only.
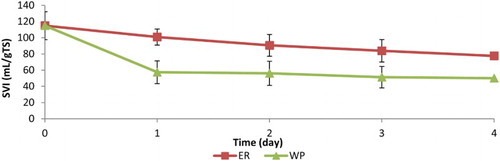
Figure 3. Change in sludge blanket volume versus settling time for WAS, endogenous respirated (ER) and worm predated (WP) sludges. Results are from a 3-day batch experiment with VS reduction of 63% ± 3 and 13% ± 3 for WP and ER, respectively, using a worm/VS ratio of 15 g/g.
The improved settleability of worm predated sludge has been shown by other authors () and is attributed to the increased density of worm faeces [Citation15]. Additionally it was observed that the worms accumulate sludge flocs around their bodies and over time these adhered flocs aggregate into larger particles and remain firmly attached to the worms and to other sludge aggregates (Supplementary Information Figure S5). A similar observation came from Inamori et al. [Citation63], who found that bacterial floc size increased in the presence of the aquatic worm Philodina erythrophthalma.
The change in SVI might also be linked to the significant change in EPS constituents after predation. Jin et al. [Citation64] concluded that the improved settleability was in part correlated to a decrease in EPS concentration, which was also found it this study. More specifically, Chen et al. [Citation65] found that the settleability in granular sludge improved, when loosely bound EPS was removed. Our current results strongly suggest that both the increased density of the faecal matter, the adherence of sludge flocs and the removal of proteins contribute to the decrease in SVI.
The dewaterability also changed in comparison with the feed WAS. The dewaterability of the different sludges were assessed with CST measurements and the results showed that the values for ER and WP slightly increased compared to the starting material: 5.3 ± 0.54, 6.9 ± 0.19 and 7.1 ± 0.31 (in seconds) for WAS, ER and WP, respectively. Unfortunately the CST of ER-30 was not measured. However, CST deterioration was reported by Park et al. [Citation66] who found that the CST increased from 50 to 517 seconds with an extended aeration time of 30 days for sludge stabilization.
The CST increased together with the amount of particles in the 2 µm range (). This suggests that the increase in CST is due to the increase in small particles generated by treatment of WAS, as was mentioned in the previous section. This notion is supported by Hall [Citation67], who found that CST increased with the amount of small particles induced by sonication of activated sludge.
Overall, worm predated sludge exhibited better settling, due to the removal of EPS and faecal pellets, and a slightly worse dewaterability, due to the increase in small particles when compared to WAS and ER. (≤2 µm) particles However when the aeration time is increased to 30 days, which results in a large fraction of ≤2 µm particles, both the settleability and filterability deteriorate.
3.3. Biochemical characterization of extended aerated and worm predated sludges
3.3.1. Soluble COD
In order to study the increased release of sCOD, the protein, carbohydrate and humic-like fractions in the various supernatants were measured. The averaged results of several batches are presented in . The results show that the carbohydrate concentration, of the sCOD in the supernatant, increased more for ER-30, followed by WP and ER when compared to WAS. A similar trend was observed for humic-like substances. The release of soluble carbohydrates and proteins, upon aerobic treatment of WAS, has been reported also by other authors [Citation66,Citation68].
Table 8. Protein and carbohydrate fractions of dissolved COD in batch supernatants of waste activated (WAS), endogenous respirated (ER), worm predated (WP) and 30-day extended aerated (ER-30) sludges.
In contrast to the increase in carbohydrates, the soluble protein concentrations remained relatively constant. Protein concentrations for ER-30 increased more than the other samples. WP had higher VS reduction levels, yet less soluble protein compared to ER-30, which supports the preference for proteins by the worms.
Higgins and Novak [Citation69] showed that the removal of proteins from flocs, by addition of proteases, resulted in the release of carbohydrates and a decrease in particle size. Their results indicate that protein removal from the EPS () by WP will also result in a release of carbohydrates. Although WP resulted in higher VS reduction compared to ER-30, only a limited carbohydrate release was observed in the WP batches compared to the ER-30 batches. This limited release might be due to carbohydrate consumption by the worms ().
To gain a better understanding of the composition of the sCOD fraction, FEEMS analysis was performed ().
Figure 4. EEMS spectra of waste activated (WAS), endogenous respirated (ER) and worm predated (WP) sludges, respectively. Measurements were done in triplicate. The plots are representative off the triplicates. VS degradation percentages of the samples were the following: ER 16%; WP 24%.
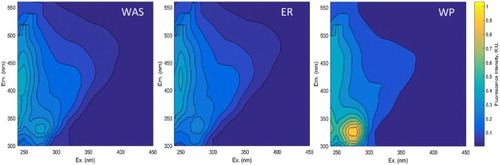
Across all the three samples, protein-like fluorophores (which are believed to be from autochthonous sources [Citation70]) are present, along with less intense humic (Ex/Em 237-260/380-500 nm range [Citation43]) signatures. In the Ex/Em 270-280/320-350 nm range, Tryptophan-protein-like substances (TPLS) increased in concentration after WP, compared to both WAS and ER samples. ER samples showed a lower concentration compared to WAS and WP.
Under WP, aromatic-protein-like substances (APLS) (Ex/Em 220-240/320-350 nm range) showed an increase compared to WAS and ER samples. WAS and ER samples had similar intensities. Additionally, the Tyrosine-like fluorophore (Ex/Em 225-237/309-221 nm range [Citation43]) appears. When dissolved organic matter is degraded, Tyrosine-like residue are exposed [Citation70].
Although TPLS and APLS are referred to as protein-like regions, the soluble protein concentration did not increase as much as the soluble polysaccharides after WP. These differences in concentration could be attributed to the sensitivity of the fluorescent method compared to bulk protein and carbohydrate measurements. Alternatively it is possible that the increase in TPLS and APLS is due to the increase in polysaccharide concentration and that these carbohydrates have a protein-like component.
This protein-like component is partly in line with the proposal of Higgins and Novak [Citation69] that sludge flocs are predominantly hold together by lectin-like polymers, which are proteins with a carbohydrate-binding domain. Another possibility is that these carbohydrates are glycoproteins or lipopolysaccharides as suggested by Park et al. [Citation71], who found that the extracted EPS carbohydrates partly co-precipitated with protein, when exposed to (NH4)2SO4.
In contrast to the FEEMS results presented here, Tian et al. [Citation23] reported a small decrease in TPLS and APLS after 25 days of operation, in a worm reactor that was part of a larger membrane bioreactor setup. Unfortunately, a control worm reactor (e.g. a worm reactor without worms) was not present; so the influence of endogenous respiration and aeration on the release of aforementioned compounds could not be determined.
The concentration of humic- and fulvic-like compounds slightly increased after predation. The fluorescence intensities of both WAS and ER remained similar. The observed increase might be attributed to a release of inert humic- and fulvic-like substances during VS reduction of humic/fulvic-bound substrates. Additionally, as previously discussed in Section 3.2.1, the removal of protein-like components from the humic- and fulvic-like substances could have resulted in the release of these compounds into the supernatant.
A small increase in humic and fulvic concentrations were not reported by Tian et al. [Citation23]. Humic and fulvic substances are thought to inhibit hydrolysis rates by adsorption of enzymes [Citation72]. Therefore, a decrease in the concentrations of these substances could have partly explained the increased hydrolysis rates during WP. In this case, humic and fulvic concentrations slightly increased; however, an inhibitory effect on conversion rates was not observed.
3.3.2. Total and dissolved cations
Multivalent cations are thought to be responsible for the formation and stability of sludge flocs. For this reason, total (sludge bound and dissolved cations) and dissolved cation concentrations were measured for the three studied sludges at the end of experiment; results are depicted in (a) and 5(b), respectively.
Figure 5. (a) Total and (b) dissolved cations for AS, ER and WP. Averaged results from 2 batches. Error bars represent the upper and lower value of the duplicate measurement. For Al3+ only the soluble fraction was measured. Average VS reduction was 10% and 35% for ER and WP, respectively.
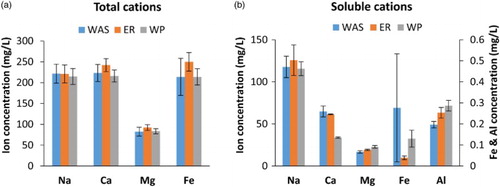
High concentrations of sodium may displace multivalent cations in an EPS matrix [Citation73–75]; therefore, total and soluble sodium concentrations were measured as well. However, sodium concentrations were similar for all sludges and more or less at the same level as the bivalent cations. No impact of sodium at these concentrations is expected.
(b) shows a small distinct increase in soluble Mg2+ and Al3+ after treatment, with WP having the largest increase followed by ER. Total Mg2+ concentrations remained fairly constant ((a)). The increasing soluble cation concentrations probably resulted from a release from the sludge flocs during VS destruction. Therefore, increased VS removal during WP coincided with an increased release of cations compared to the ER results. These observations are consistent with the observations made by Jin et al. [Citation64], who found values in the range of (WAS – ER-30) 18–38 mg Mg2+/L and 64–103 mg Ca2+/L. Furthermore, they concluded that the release of divalent cations was linked to aerobic VS destruction.
Interestingly the soluble Ca2+ concentration showed a large decrease after WP compared to WAS and ER, where a large increase was expected due to the concomitant Ca2+ increase with aerobic VS reduction [Citation66]. The observed Ca2+ decrease was consistent over multiple batches. Several explanations for the Ca2+ decrease are viable. Most probably Ca2+ was taken up by the worms during the batch incubations. The haemoglobin in T. tubifex contains besides iron also calcium. The molar ratios of Fe:Ca were reported to be 160:70 [Citation76].
Additionally precipitation of Ca2+ could have occurred with the increased release of phosphates during predation. Calcium phosphate precipitation was reported to be possible at slightly alkaline conditions and similar calcium concentrations [Citation77,Citation78]. However a decrease in calcium was not observed in ER.
Alternatively, an increased amount of Ca2+ could have been bound to the released humic and fulvic substances that were liberated or made accessible by removal of VS through the action of the worms, as mentioned previously. Azman et al. [Citation79] showed that calcium adsorbs to humic compounds and thereby mitigates the enzyme-binding capacities of these humics. By this mitigation the hydrolysis rates were effectively increased during anaerobic digestion of cellulose [Citation79]. It is not known whether such mechanism is of importance during WP.
The calcium concentrations remain in sharp contrast to what other authors found with regard to the release of soluble Ca2+ [Citation66,Citation68]. A possible explanation for the relatively stable Ca2+ concentrations, when comparing WAS and ER, is the difference in batch duration, which was 30 days in the aforementioned studies versus 4 days in our present study. Unfortunately, the metal content of the worms and ER-30 were not determined and hence, the reason for the lower soluble calcium concentration remains speculative.
Iron is associated with the protein fraction of EPS and iron is released during anaerobic storage of sludge [Citation80,Citation81]. Because of this iron-protein interaction and the removal of protein from the EPS, both total and soluble ferric iron were monitored ((a) and 5(b)). Regardless of the large uncertainty in the soluble WAS measurements, a clear difference between ER and WP is observed. For the total Fe3+ fraction, an average of 36 mg /L iron was removed during WP. Concomitantly, the soluble Fe content in the WP supernatant was higher compared to the ER supernatant.
Results suggest that a part of the protein-bound Fe in the sludge was released during WP and another part absorbed by the worms as an iron source for their iron containing haemoglobin [Citation76]. Unfortunately, the latter could not be verified because the metal content of the worms was not determined. Additionally, due to the possible occurrence of anoxic zones, being formed by clumps of worms, the microbial reduction of Fe3+ to Fe2+ might have had occurred. However, this is not very likely as this would result in ferrous precipitation with soluble phosphate [Citation82,Citation83]. These precipitates would be included in the total iron concentration, which would therefore not change. This is clearly not the case.
4. General discussion
The objective of this study was to gain more insight into the mechanisms of hydrolysis and the general aspects of predation of activated sludge by aquatic worms. Results show that WP of activated sludge has a significant effect on the removal of VS and dewaterability compared to the control without worms. The VS removal is accompanied by improved settling characteristics and an increased release rate of sCOD, inorganic nitrogen and phosphorus. The observed inorganic nitrogen release agreed with the drop in the EPS-protein fraction that T. tubifex specifically seems to target as the substrate.
Furthermore, the results suggest that the release of soluble carbohydrates, cations and humic/fulvic substances is also due to the removal of protein. Concomitantly, sludge flocs disintegrate, resulting in smaller particles and thus increasing turbidity and CST. In contrast with the deteriorating dewaterability associated with ER-30, the settleability increased and the CST did not increase as much as ER-30, which can be attributed to the more compact worm faeces, the removal of VS and the aggregation of sludge particles through sludge–worm interactions. Recalcitrant flock biopolymers that can influence hydrolysis rates such as humic and fulvic substances were not removed but slightly liberated. ALE, on the other hand, was partly consumed during worm treatment of WAS. Additional mechanisms related to sludge hydrolysis were not revealed by researching the biochemical and physical characteristics of WP.
Besides the aerobic removal of proteins and the concomitant release of soluble compounds, Park et al. [Citation66] also showed that WAS conversion, using sequenced aerobic and anaerobic (or vice versa) conditions, reaches the same level of VS reduction of the combined processes, reaching about 63%. Roughly 45–50% of the initial VS is removed in the first stage of either aerobic or anaerobic treatment after 30 days. This aerobic VS reduction is in the same order of magnitude as the averaged results presented in this study, which were about 47% ± 15 for WP and 30% for extended aeration (ER-30). Similar results were reported by Buys et al. [Citation52], who showed that worm predated and endogenously respirated sludge both reached similar VS degradation levels of about 58% with a difference in incubation time of 46 days.
Surprisingly Tamis et al. [Citation21] found 20–30% aerobic VS reduction by WP and an additional 40–55% VS reduction upon anaerobic storage of the worm predated sludges. A total of about 65% of the initial VS was removed during the aerobic and anaerobic treatment of WAS. Comparable results were reported by Hendrickx et al. [Citation15], who showed that WP followed by anaerobic digestion of the worm faeces resulted in a total of 50% VS reduction. In both examples the end point for the aerobic to anaerobic conversion reached similar values as the 63% reduction mentioned previously. The increased VS removal results in a lowering of the biological methane potential of worm predated sludges [Citation84].
In conventional WWTPs where aerobic unit operations are predominantly followed up by anaerobic treatment for sludge digestion, 30–35% of the initial aerobic VS is degraded during digestion. When the findings of Park et al. [Citation66] and Tamis et al. [Citation21] and the results presented here are considered, it seems that the 45–50% of the initial VS, which remains undigested during anaerobic digestion, is digested by additional aerobic (worm) treatment. Furthermore based on the similar VS reduction levels between WP and ER-30, it seems that the worms specifically target a fraction of the sludge that is predominantly biodegradable under aerobic conditions, yet at significantly higher degradation rates when compared to the endogenous decay of WAS.
The presented results call for further research concerning the aerobic and anaerobic biodegradability of predated sludges and a (re-) evaluation of implementing WP as a sludge reduction method. The latter could be of particular interest to WWTPs in which a large VS fraction of WAS seems to be left unaltered in current anaerobic digesters, resulting in large sludge disposal costs associated with the operation of these WWTPs. Note has to be taken of the potential interference with liquid/solids separation in WWTPs due to the increase in small particles which are introduced by predation technologies. In addition to the bioconversion potential of applying WP to activated sludge, the biological cause of sludge reduction deserves further attention, especially to provide insight into the enzymatic activity responsible for the efficient reduction of polymers and possibly the reduction of microbial mass.
5. Conclusions
This research set out to gain more insight into the hydrolytic mechanisms and the general aspects of WP. It was found that worms specifically target the protein fraction of activated sludge. The removal of proteinaceous material from the activated sludge attributed to the increase in sCOD, inorganic nitrogen, the cations Mg2+, Al3+ and Fe3+, fulvic and humic substances as well as the disintegration of particles and partly the improved sludge dewater-ability. Additionally T. tubifex seems to predominantly target the aerobic degradable fraction of activated sludge.
Supplemental_Information
Download MS Word (3.5 MB)Acknowledgements
We would like to thank J. D’Andrilli for her assistance in post-processing the EEMs samples, Marissa Buyers-Basso, Nuria Garcia-Mancha and Gonzalo Villaverde-Durango for all the work they have done with the worms.
Disclosure statement
No potential conflict of interest was reported by the authors.
ORCiD
Steef de Valk http://orcid.org/0000-0002-8383-8110
Christine M. Foreman http://orcid.org/0000-0003-0230-4692
Jules B. van Lier http://orcid.org/0000-0003-2607-5425
Merle K. de Kreuk http://orcid.org/0000-0002-5111-9461
References
- Gendebien A, Davis B, Hobson J, et al. Environmental, economic and social impacts of the use of sewage sludge on land Final Report Part III: Project Interim Reports; 2010.
- Commission of European Communities. Council directive 91/271/EEC of 21 March 1991 concerning urban wastewater treatment amended by the 98/15/EC of 27 February; 1998.
- Tchobanoglous G, Burton FL, Metcalf & Eddy. Wastewater engineering: treatment, disposal and reuse. 3rd ed. New York: McGraw-Hill; 1991.
- Bougrier C, Albasi C, Delgenes J, et al. Effect of ultrasonic, thermal and ozone pre-treatments on waste activated sludge solubilisation and anaerobic biodegradability. Chem Eng Process. 2006;45:711–718. doi: 10.1016/j.cep.2006.02.005
- Jaziri K, Casellas M, Dagot C. Comparing the effects of three pre-treatment disintegration techniques on aerobic sludge digestion: biodegradability enhancement and microbial community monitoring by PCR–DGGE. Environ Technol. 2012;33:1435–1444. doi: 10.1080/09593330.2011.632653
- Pilli S, Yan S, Tyagi RD, et al. Thermal pretreatment of sewage sludge to enhance anaerobic digestion: a review. Crit Rev Environ Sci Technol. 2015;45:669–702. doi: 10.1080/10643389.2013.876527
- Chen G-H, An K-J, Saby S, et al. Possible cause of excess sludge reduction in an oxic-settling-anaerobic activated sludge process (OSA process). Water Res. 2003;37:3855–3866. doi: 10.1016/S0043-1354(03)00331-2
- Novak JT, Chon DH, Curtis B-A, et al. Biological solids reduction using the cannibal process. Water Environ. Res. 2007;79:2380–2386. doi: 10.2175/106143007X183862
- Wang Y, Li Y, Wu G. SRT contributes significantly to sludge reduction in the OSA-based activated sludge process. Environ Technol. 2016;3330:1–11.
- Van Loosdrecht M, Henze M. Maintenance, endogeneous respiration, lysis, decay and predation. Water Sci Technol. 1999;39:107–117. doi: 10.1016/S0273-1223(98)00780-X
- Ratsak CH, Verkuijlen J. Sludge reduction by predatory activity of aquatic oligochaetes in wastewater treatment plants: science or fiction? A review. Hydrobiologia. 2006;564:197–211. doi: 10.1007/s10750-005-1719-7
- Wei Y, Liu J. The discharged excess sludge treated by Oligochaeta. Water Sci Technol. 2005;52:265–272.
- Liang P, Huang X, Qian Y. Excess sludge reduction in activated sludge process through predation of Aeolosoma hemprichi. Biochem Eng J. 2006;28:117–122. doi: 10.1016/j.bej.2005.09.008
- Elissen HJH, Hendrickx TLG, Temmink H, et al. A new reactor concept for sludge reduction using aquatic worms. Water Res. 2006;40:3713–3718. doi: 10.1016/j.watres.2006.08.029
- Hendrickx TLG, Temmink H, Elissen HJH, et al. Aquatic worms eat sludge: mass balances and processing of worm faeces. J Hazard Mater. 2010;177:633–638. doi: 10.1016/j.jhazmat.2009.12.079
- Hendrickx TLG, Elissen HHJ, Temmink H, et al. Operation of an aquatic worm reactor suitable for sludge reduction at large scale. Water Res. 2011;45:4923–4929. doi: 10.1016/j.watres.2011.06.031
- Hendrickx TLG, Temmink H, Elissen HJH, et al. The effect of operating conditions on aquatic worms eating waste sludge. Water Res. 2009;43:943–950. doi: 10.1016/j.watres.2008.11.034
- Guo X, Liu J, Wei Y, et al. Sludge reduction with Tubificidae and the impact on the performance of the wastewater treatment process. J Environ Sci. 2007;19:257–263. doi: 10.1016/S1001-0742(07)60042-4
- Huang X, Liang P, Qian Y. Excess sludge reduction induced by Tubifex tubifex in a recycled sludge reactor. J. Biotechnol. 2007;127:443–451. doi: 10.1016/j.jbiotec.2006.07.035
- Wei Y, Zhu H, Wang Y, et al. Nutrients release and phosphorus distribution during oligochaetes predation on activated sludge. Biochem Eng J. 2009;43:239–245. doi: 10.1016/j.bej.2008.10.004
- Tamis J, van Schouwenburg G, Kleerebezem R, et al. A full scale worm reactor for efficient sludge reduction by predation in a wastewater treatment plant. Water Res. 2011;45:5916–5924. doi: 10.1016/j.watres.2011.08.046
- Wang Q, Wang Z, Wu Z, et al. Sludge reduction and process performance in a submerged membrane bioreactor with aquatic worms. Chem Eng J. 2011;172:929–935. doi: 10.1016/j.cej.2011.07.004
- Tian Y, Li Z, Lu Y. Changes in characteristics of soluble microbial products and extracellular polymeric substances in membrane bioreactor coupled with worm reactor: relation to membrane fouling. Bioresour Technol. 2012;122:62–69. doi: 10.1016/j.biortech.2012.05.009
- Tian Y, Lu Y, Li Z. Performance analysis of a combined system of membrane bioreactor and worm reactor: wastewater treatment, sludge reduction and membrane fouling. Bioresour Technol. 2012;121:176–182. doi: 10.1016/j.biortech.2012.06.071
- Tian Y, Li Z, Ding Y, et al. Identification of the change in fouling potential of soluble microbial products (SMP) in membrane bioreactor coupled with worm reactor. Water Res. 2013;47:2015–2024. doi: 10.1016/j.watres.2013.01.026
- Matisoff G, Wang X. Solute transport in sediments by freshwater infaunal bioirrigators. Limnol. Oceanogr. 1998;43:1487–1499. doi: 10.4319/lo.1998.43.7.1487
- Michaud E, Desrosiers G, Mermillod-Blondin F, et al. The functional group approach to bioturbation: the effects of biodiffusers and gallery-diffusers of the Macoma balthica community on sediment oxygen uptake. J Exp Mar Bio Ecol. 2005;326:77–88. doi: 10.1016/j.jembe.2005.05.016
- Mermillod-Blondin F, Rosenberg R. Ecosystem engineering: the impact of bioturbation on biogeochemical processes in marine and freshwater benthic habitats. Aquat Sci. 2006;68:434–442. doi: 10.1007/s00027-006-0858-x
- Brinkhurst RO, Chua KE. Preliminary investigation of the exploitation of some potential nutritional resources by three sympatric tubificid oligochaetes. J Fish Res Board Canada. 1969;26:2659–2668. doi: 10.1139/f69-258
- Wavre M, Brinkhurst RO. Interactions between some Tubificid Oligochaetes and bacteria found in the sediments of Toronto Harbour, Ontario. J Fish Res Board Canada. 1971;28:335–341. doi: 10.1139/f71-045
- Tsuchiya M, Kurihara Y. The feeding habits and food sources of the deposit-feeding polychaete, Neanthes Japonica (Izuka). J Exp Mar Bio Ecol. 1979;36:79–89. doi: 10.1016/0022-0981(79)90101-1
- Plante CJ, Shriver AG. Differential lysis of sedimentary bacteria by Arenicola marina L.: examination of cell wall structure and exopolymeric capsules as correlates. J Exp Mar Bio Ecol. 1998;229:35–52. doi: 10.1016/S0022-0981(98)00039-2
- Liu J, Zuo W, Zhang J, et al. Shifts in microbial community structure and diversity in a MBR combined with worm reactors treating synthetic wastewater. J Environ Sci. 2016:1–10. DOI:10.1016/j.jes.2016.03.009
- Tiehm A, Nickel K, Zellhorn M, et al. Ultrasonic waste activated sludge disintegration for improving anaerobic stabilization. Water Res. 2001;35:2003–2009. doi: 10.1016/S0043-1354(00)00468-1
- Vavilin VA, Fernandez B, Palatsi J, et al. Hydrolysis kinetics in anaerobic degradation of particulate organic material: an overview. Waste Manag. 2008;28:939–951. doi: 10.1016/j.wasman.2007.03.028
- Cai L, Gao D, Wang K, et al. Sludge reduction using aquatic worms under different aeration regimes. Environ. Technol. 2016;3330:1–7. doi: 10.1080/09593330.2016.1210241
- A.P.H.A. Standard methods for the examination of water and wastewater. 22nd ed. Washington, DC: American Public Health Association, American Water Works Association, Water Environment Federation; 2012.
- Lawler D. Particle size distributions in treatment processes: theory and practice. Water Sci Technol. 1997;36:15–23. doi: 10.1016/S0273-1223(97)00414-9
- DuBois M, Gilles KA, Hamilton JK, et al. Colorimetric method for determination of sugars and related substances. Anal Chem. 1956;28:350–356. doi: 10.1021/ac60111a017
- Lowry OH, Rosenbrough NJ, Farr AL, et al. Protein measurement with the Folin phenol reagent. J Biol. Chem. 1951;193:265–275.
- Frølund B, Griebe T, Nielsen PH. Enzymatic activity in the activated-sludge floc matrix. Appl Microbiol Biotechnol. 1995;43:755–761. doi: 10.1007/s002530050481
- Avella AC, Görner T, de Donato P. The pitfalls of protein quantification in wastewater treatment studies. Sci Total Environ. 2010;408:4906–4909. doi: 10.1016/j.scitotenv.2010.05.039
- Coble PG. Characterization of marine and terrestrial DOM in seawater using excitation-emission matrix spectroscopy. Mar Chem. 1996;51:325–346. doi: 10.1016/0304-4203(95)00062-3
- McKnight DM, Boyer EW, Westerhoff PK, et al. Spectrofluorometric characterization of dissolved organic matter for indication of precursor organic material and aromaticity. Limnol Oceanogr. 2001;46:38–48. doi: 10.4319/lo.2001.46.1.0038
- Hudson N, Baker A, Reynolds D. Fluorescence analysis of dissolved organic matter in natural, waste and polluted waters – a review. River Res Appl. 2007;23:631–649. doi: 10.1002/rra.1005
- D’Andrilli J, Foreman CM, Marshall AG, et al. Characterization of IHSS Pony Lake fulvic acid dissolved organic matter by electrospray ionization Fourier transform ion cyclotron resonance mass spectrometry and fluorescence spectroscopy. Org Geochem. 2013;65:19–28. doi: 10.1016/j.orggeochem.2013.09.013
- Marhaba TF, Lippincott RL. Application of fluorescence technique for rapid identification of DOM fractions in source waters. J Environ Eng. 2000;126:1039–1044. doi: 10.1061/(ASCE)0733-9372(2000)126:11(1039)
- Marhaba T. Rapid identification of dissolved organic matter fractions in water by spectral fluorescent signatures. Water Res. 2000;34:3543–3550. doi: 10.1016/S0043-1354(00)00090-7
- van Langerak EPA, Gonzalez-Gil G, van Aelst A, et al. Effects of high calcium concentrations on the development of methanogenic sludge in upflow anaerobic sludge bed (UASB) reactors. Water Res. 1998;32:1255–1263. doi: 10.1016/S0043-1354(97)00335-7
- Frølund B, Palmgren R, Keiding K, et al. Extraction of extracellular polymers from activated sludge using a cation exchange resin. Water Res. 1996;30:1749–1758. doi: 10.1016/0043-1354(95)00323-1
- Lin Y, de Kreuk M, van Loosdrecht MCM, et al. Characterization of alginate-like exopolysaccharides isolated from aerobic granular sludge in pilot-plant. Water Res. 2010;44:3355–3364. doi: 10.1016/j.watres.2010.03.019
- Buys BR, Klapwijk A, Elissen H, et al. Development of a test method to assess the sludge reduction potential of aquatic organisms in activated sludge. Bioresour Technol. 2008;99:8360–8366. doi: 10.1016/j.biortech.2008.02.041
- Tan WF, Koopal LK, Weng LP, et al. Humic acid protein complexation. Geochim Cosmochim Acta. 2008;72:2090–2099. doi: 10.1016/j.gca.2008.02.009
- Tomaszewski JE, Schwarzenbach RP, Sander M. Protein Encapsulation by Humic Substances. Environ Sci Technol. 2011;45:6003–6010. doi: 10.1021/es200663h
- Wang Z, Cao J, Meng F. Interactions between protein-like and humic-like components in dissolved organic matter revealed by fluorescence quenching. Water Res. 2015;68:404–413. doi: 10.1016/j.watres.2014.10.024
- Brisbane PG, Amato M, Ladd JN. Gas chromatographic analysis of amino acids from the action of proteolytic enzymes on soil humic acids. Soil Biol Biochem. 1972;4:51–61. doi: 10.1016/0038-0717(72)90042-9
- Swift RS, Posner AM. The distribution and extraction of soil nitrogen as a function of soil particle size. Soil Biol Biochem. 1972;4:181–186. doi: 10.1016/0038-0717(72)90009-0
- Knicker H. Nature of organic nitrogen in fine particle size separates of sandy soils of highly industrialized areas as revealed by NMR spectroscopy. Soil Biol Biochem. 2000;32:241–252. doi: 10.1016/S0038-0717(99)00154-6
- Shan J, Brune A, Ji R. Selective digestion of the proteinaceous component of humic substances by the geophagous earthworms Metaphire guillelmi and Amynthas corrugatus. Soil Biol Biochem. 2010;42:1455–1462. doi: 10.1016/j.soilbio.2010.05.008
- Lin YM, Sharma PK, van Loosdrecht MCM. The chemical and mechanical differences between alginate-like exopolysaccharides isolated from aerobic flocculent sludge and aerobic granular sludge. Water Res. 2013;47:57–65. doi: 10.1016/j.watres.2012.09.017
- Abbassi B, Dullstein S, Räbiger N. Minimization of excess sludge production by increase of oxygen concentration in activated sludge flocs; experimental and theoretical approach. Water Res. 2000;34:139–146. doi: 10.1016/S0043-1354(99)00108-6
- Stricot M, Filali A, Lesage N, et al. Side-stream membrane bioreactors: influence of stress generated by hydrodynamics on floc structure, supernatant quality and fouling propensity. Water Res. 2010;44:2113–2124. doi: 10.1016/j.watres.2009.12.021
- Inamori Y, Kuniyasu Y, Hayashi N, et al. Monoxenic and mixed cultures of the small metazoa Philodina erythrophthalma and Aeolosoma hemprichi isolated from a waste-water treatment process. Appl Microbiol Biotechnol. 1990;34:404–407. doi: 10.1007/BF00170069
- Jin B, Wilén B-M, Lant P. A comprehensive insight into floc characteristics and their impact on compressibility and settleability of activated sludge. Chem Eng J. 2003;95:221–234. doi: 10.1016/S1385-8947(03)00108-6
- Chen H, Zhou S, Li T. Impact of extracellular polymeric substances on the settlement ability of aerobic granular sludge. Environ Technol. 2010;31:1601–1612. doi: 10.1080/09593330.2010.482146
- Park C, Abu-Orf MM, Novak JT. The digestibility of waste activated sludges. Water Environ Res. 2006;78:59–68. doi: 10.2175/106143005X84521
- Hall T. Sonication induced changes of particle size and their effects on activated sludge dewaterability. Environ Technol Lett. 1982;3:79–88. doi: 10.1080/09593338209384102
- Novak JT, Sadler ME, Murthy SN. Mechanisms of floc destruction during anaerobic and aerobic digestion and the effect on conditioning and dewatering of biosolids. Water Res. 2003;37:3136–3144. doi: 10.1016/S0043-1354(03)00171-4
- Higgins MJ, Novak JT. Characterization of Exocellular Protein and Its Role in Bioflocculation. J Environ Eng 1997;123:479–485. doi: 10.1061/(ASCE)0733-9372(1997)123:5(479)
- Chari NVHK, Sarma NS, Pandi SR, et al. Seasonal and spatial constraints of fluorophores in the midwestern Bay of Bengal by PARAFAC analysis of excitation emission matrix spectra. Estuar Coast Shelf Sci. 2012;100:162–171. doi: 10.1016/j.ecss.2012.01.012
- Park C, Novak JT, Helm RF, et al. Evaluation of the extracellular proteins in full-scale activated sludges. Water Res. 2008;42:3879–3889. doi: 10.1016/j.watres.2008.05.014
- Fernandes TV. Hydrolysis inhibition of complex biowaste. Wageningen, The Netherlands: Wageningen University; 2010.
- Bruus JH, Nielsen PH, Keiding K. On the stability of activated sludge flocs with implications to dewatering. Water Res. 1992;26:1597–1604. doi: 10.1016/0043-1354(92)90159-2
- Higgins MJ, Novak JT. The effect of cations on the settling and dewatering of activated sludges: laboratory results. Water Environ. Res. 1997;69:215–224. doi: 10.2175/106143097X125371
- Sobeck DC, Higgins MJ. Examination of three theories for mechanisms of cation-induced bioflocculation. Water Res. 2002;36:527–538. doi: 10.1016/S0043-1354(01)00254-8
- Rokosz MJ, Vinogradov SN. X-ray fluorescence spectrometric determination of the metal content of the extracellular hemoglobin of Tubifex tubifex. Biochim Biophys Acta – Protein Struct Mol Enzymol. 1982;707:291–293. doi: 10.1016/0167-4838(82)90364-8
- Füredi-Milhofer H, Purgarić B, Brečević L, et al. Precipitation of calcium phosphates from electrolyte solutions. Calcif Tissue Res. 1971;8:142–153. doi: 10.1007/BF02010131
- Mañas A, Pocquet M, Biscans B, et al. Parameters influencing calcium phosphate precipitation in granular sludge sequencing batch reactor. Chem Eng Sci. 2012;77:165–175. doi: 10.1016/j.ces.2012.01.009
- Azman S, Khadem A, Zeeman G, et al. Mitigation of humic acid inhibition in anaerobic digestion of cellulose by addition of various salts. Bioengineering. 2015;2:54–65. doi: 10.3390/bioengineering2020054
- Nielsen PH, Keiding K. Disintegration of activated sludge flocs in presence of sulfide. Water Res. 1998;32:313–320. doi: 10.1016/S0043-1354(97)00235-2
- Murthy SN, Novak JT. Factors affecting floc properties during aerobic digestion: implications for dewatering. Water Environ. Res. 1999;71:197–202. doi: 10.2175/106143098X121879
- Ivanov V, Stabnikov V, Zhuang WQ, et al. Phosphate removal from the returned liquor of municipal wastewater treatment plant using iron-reducing bacteria. J Appl Microbiol. 2005;98:1152–1161. doi: 10.1111/j.1365-2672.2005.02567.x
- Gutierrez O, Park D, Sharma KR, et al. Iron salts dosage for sulfide control in sewers induces chemical phosphorus removal during wastewater treatment. Water Res. 2010;44:3467–3475. doi: 10.1016/j.watres.2010.03.023
- Serrano A, Hendrickx TLG, Elissen HHJ, et al. Can aquatic worms enhance methane production from waste activated sludge? Bioresour Technol. 2016;211:51–57. doi: 10.1016/j.biortech.2016.03.061
