Abstract
In the absence of a direct head-to-head study, we performed an indirect historical comparison of ospemifene 60 mg (Senshio®) vs. local vaginal estrogens in moderate or severe vulvar and vaginal atrophy (VVA). A literature search was carried out of clinical efficacy/safety trials of local vaginal estrogens in VVA approved in Europe. For efficacy comparison, studies had to be placebo-controlled and of 12 weeks’ duration. For safety comparison, studies had to be ≥40 weeks’ duration. Efficacy endpoints were the difference between active and placebo in change from baseline to week 12 for symptoms, vaginal pH, and maturation value (MV). Safety endpoints were endometrial safety, breast safety, thrombosis, and adverse events. The 12-week improvement over placebo in symptom score was not different for ospemifene 60 mg and 17β-estradiol 10 μg and for ospemifene 60 mg and estriol gel. After 12 weeks, the percentages with vaginal pH <5.0 and <5.5 were better for ospemifene 60 mg than 10 μg 17β-estradiol. Week-12 pH changes were comparable with estriol pessaries or gel and ospemifene 60 mg. The 12-week MV improvements over placebo were similar or better with ospemifene 60 mg compared with 10 μg 17β-estradiol and with estriol pessaries or gel. There was no increased vaginal bleeding, endometrial hyperplasia, or carcinoma (including breast cancer) relative to placebo and no signal for increased risk of venous thromboembolism with ospemifene 60 mg or 10 μg 17β-estradiol, but the confidence intervals for both products do not exclude an increased risk. This historical indirect comparison suggests that ospemifene 60 mg has an efficacy, safety, and tolerability profile comparable to or better than local vaginal estrogens in the treatment of VVA.
Introduction
Vulvar and vaginal atrophy (VVA) is a chronic and progressive medical condition that develops because of the decline of estrogen levelsCitation1,Citation2. Symptoms, including vaginal dryness, irritation, soreness, and dyspareunia plus urinary frequency, urgency, and urge incontinence, usually persist or worsen in the absence of treatmentCitation3. Approximately 50% of postmenopausal women suffer from VVA symptomsCitation4. Local vaginal estrogens represent the current standard of care for treatment of symptomatic VVA and are effective in alleviating symptoms of moderate-to-severe VVACitation5–7. However, there are some significant barriers to treatment, including lack of knowledge about VVA, reluctance to discuss symptoms with health-care professionals, safety concerns, contraindications to estrogen use and inconvenienceCitation8,Citation9.
The non-estrogen selective estrogen receptor modulator (SERM) ospemifene (Senshio®) provides a new oral therapy option for postmenopausal women with moderate or severe symptomatic VVA who are not candidates for local estrogensCitation7,Citation10. Ospemifene acts by exerting a tissue-specific effect, including an estrogen agonist effect on the vaginal epitheliumCitation11,Citation12. The efficacy and safety of ospemifene were established in 30 clinical trials, with 2471 subjects exposed to ospemifene. These studies formed the basis for the EU approval of ospemifene for the treatment of moderate-to-severe symptomatic VVA in postmenopausal women who are not candidates for local vaginal estrogen therapyCitation10.
Direct comparisons between oral ospemifene and locally delivered estrogens have not been performed and would be technically and scientifically challenging.
In the absence of direct (head-to-head) comparisons in randomized clinical trials and at the request of EU regulatory authorities, an indirect historical comparison was performed of ospemifene vs. local estrogens that are currently available for VVA treatment in Europe.
Methods
Identification and selection of studies
A literature search of all articles published up to 25 October 2012 was conducted using PubMed to identify publications of clinical trials of local vaginal estrogens for the treatment of VVA. Additional searches were conducted using Cochrane Reviews and www.clinicaltrials.gov.
Studies on the efficacy of local estrogens were selected using the following criteria: (1) the study evaluated monotherapy and was placebo-controlled; (2) relevant endpoints were evaluated at 12 weeks; (3) the formulation was available on the market in Europe.
For the long-term safety evaluation, local estrogen studies were only included if the duration was ≥40 weeks and if the formulation was available in Europe. Trials on systemic hormones or studies on indications other than vaginal atrophy and open-label data were excluded.
Endpoints
The efficacy endpoints considered were symptoms, vaginal pH, and maturation value (MV). The safety endpoints considered were endometrial safety (including vaginal bleeding), breast safety, venous thromboembolism (VTE), and most common adverse events (AEs).
Statistical analyses
Comparisons between local estrogen and ospemifene data were conducted for the three efficacy outcomes of symptoms, pH, and MV, where available. Since baseline data, as well as the study populations, were somewhat different, the comparison is corrected for baseline (difference from baseline) and, as much as possible, for differences between treatment groups (difference in change from baseline between active and placebo arms). All changes/effects discussed are relative to placebo (i.e. the data were normalized to the placebo population to show the effect of the drug regardless of the population/study differences). If the differences from placebo are different between different products, the direction of the difference has been indicated.
For each endpoint, both mean 12-week changes within each treatment arm and 12-week changes relative to placebo were estimated (mean difference between arms = mean in active arm – mean in placebo arm). Two-sided 95% confidence intervals (CIs) for 12-week differences between active and placebo arms were estimated assuming normally distributed data.
For the comparison of safety, incidence proportions were calculated by dividing the number of events by the number of observed cases. Data from different ospemifene studies were pooled. Data from studies on local estrogens could only be pooled for the VTE data.
Results of the indirect comparison
Study identification
Efficacy analysis
Out of 88 clinical studies using a local estrogen as an active comparator, 21 studies were placebo-controlled, including 19 studies with unique data. Of these, 15 were excluded from the efficacy analysis for the reasons listed in Table S1 (Supplementary Material, see http://dx.doi.org/10.1080/13697137.2017.1284780), e.g. the populations or endpoints were not comparable or the active component is no longer available in Europe. Of the remaining studies, two assessed 10 μg 17β-estradiol (Vagifem®)Citation13,Citation14. Details of these studies are summarized in . 17β-Estradiol 25 μg, one of the treatment groups studied by BachmannCitation13, has been largely withdrawn from the European market and was not included in the efficacy evaluation.
Table 1. Study design of four local estrogen trials and three ospemifene trials for indirect comparison of efficacy.
Low-dose estriol vaginal ovulae and estriol gel are now approved in some countries and two studies, one assessing estriol pessaries and one estriol gel, are also includedCitation15,Citation16.
Eight 60 mg ospemifene phase 2/3 studies were identified. Two studies were excluded as they were in healthy postmenopausal volunteers, one because it was only of 6 weeks’ duration, and two because they were long-term extension phases with no efficacy data. Ospemifene 60 mg, 12-week, placebo-controlled efficacy data for comparison were available from the three pivotal trials in women with VVA (Studies 15-50310Citation17, 15-50718Citation18, and 15-50821 for the dyspareunia armCitation19 and for the dryness armCitation20) ().
Safety analysis
Of the 88 clinical studies on local estrogens, 14 studies had a duration ≥40 weeks. Three of these were excluded from the safety analysis because they reported data for an estrogen that is not available in Europe and one small study (n = 68) because the full text article could not be located (published in Czech). The ten remaining studies included five on 10 μg 17β-estradiol and four studies investigated an estradiol ring (). One of the 10 μg 17β-estradiol studiesCitation26 reported the results of a pooled analysis of data from two studiesCitation14,Citation27. For WeisbergCitation23, only data from the Estring arm were included as 25 μg 17β-estradiol is being withdrawn and is no longer available in many EU countries. Data from BachmannCitation13 were excluded because patients were switched from 10 μg 17β-estradiol to 25 μg 17β-estradiol after 12 weeks’ treatment.
Table 2. Local estrogen studies with a duration ≥40 weeks.
Ospemifene 60 mg long-term safety data came from the pivotal 52-week, double-blind, placebo-controlled trial (Study 15-50718Citation18) and the blinded 40-week extension from one of the pivotal 12-week, double-blind, placebo-controlled trials (Study 15-50310xCitation29). As the authors had access to the study reports of the ospemifene studies, the safety data from the 15-50310x study include the safety data from the preceding 15-50310 study for those women who continued in Study 15-50310x, thus representing 52 weeks of observation for safety reporting. In order to allow comparison with the publications of local estrogens, some of the other ospemifene data were also taken from the ospemifene clinical trial database.
Efficacy comparison
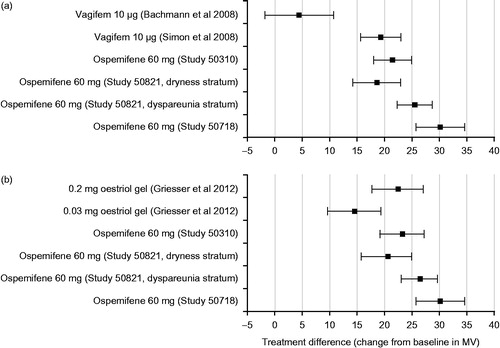
There were differences in the study designs and patient baseline characteristics between the local estrogen and ospemifene trials (e.g. age, time since menopause, and baseline MV) (). At enrolment, all of the women in the ospemifene trials and the Simon 2008 10 μg 17β-estradiol study had a pH >5Citation14, whereas approximately 11% of women had a lower pH value in the Bachmann 10 μg 17β-estradiol studyCitation13. Also in the estriol studies, some women (number unknown) had a pH <5 (range 4.5–7.0 at baseline for GriesserCitation15, 6.4 ± 1.4 (mean ± SD) for CanoCitation16). MVs at baseline varied across studies, ranging from 9.3 to 47.5, despite the fact that the inclusion criteria in most studies specified that superficial cells had to be ≤5%.
Table 3. Baseline data from the four local estrogen trials and three ospemifene trials for indirect comparison of efficacy. Data are given as mean ± standard deviation.
In the two estriol studies, only observed case data are published so, for comparison, observed case data for 60 mg ospemifene are also provided.
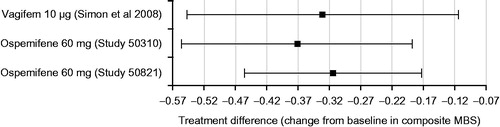
Composite symptom scores, including the most bothersome symptom
A composite score, based on a four-point severity scale (none, mild, moderate, severe) for the most bothersome symptom (MBS) of VVA (which included vaginal dryness, dyspareunia, vaginal soreness, vaginal and/or vulvar irritation/itching, dysuria or vaginal bleeding associated with sexual activity) was used to compare 60 mg ospemifene with 10 μg 17β-estradiolCitation14. The improvement at week 12 relative to placebo was not different for ospemifene and that reported for 10 μg 17β-estradiol (). The improvement relative to placebo could not be calculated for the Bachmann studyCitation13, but the magnitude of improvement was similar to that in the Simon 2008 studyCitation14. The symptom score for estriol, as reported by GriesserCitation15, could not be compared due to a different assessment method (visual analog scale). A global symptom score, based on a composite of the intensity scores of all symptoms of vaginal dryness, dyspareunia, pruritus, burning, and dysuria, was used in the estriol gel studyCitation16, so ospemifene data were recalculated using the same definition. The magnitude in placebo-subtracted improvement from baseline to week 12 was comparable for estriol gel 0.005% (1.07) and 60 mg ospemifene (1.02–1.27).
Figure 1. Difference between active arm and placebo in composite most bothersome symptom (MBS) at week 12. Score based on severity of the most bothersome symptom: none = 0, mild = 1, moderate = 2 and severe = 3. A decrease in score means improvement in symptom severity.
Data for the percentage of subjects cured or improved of their symptom of vaginal dryness and dyspareunia are given in . The improvement with ospemifene relative to placebo is at least comparable to that with estriol 0.005% gelCitation16.
Table 4. Improved or cured proportions and relative proportions for vaginal dryness and dyspareunia at 12 weeks, observed cases.
Vaginal pH Value
In the Bachmann 10 μg 17β-estradiol studyCitation13, only data on the proportion of women with a pH <5 were reported. The percentages of women achieving a vaginal pH <5 with ospemifene relative to placebo and 10 μg 17β-estradiol vs. placebo are shown in .
Figure 2. Difference between active arm and placebo in (a) percentage of women with vaginal pH <5, (b) pH <5.5, and (c) mean change (observed data only) at week 12. Data are mean and 95% confidence interval.
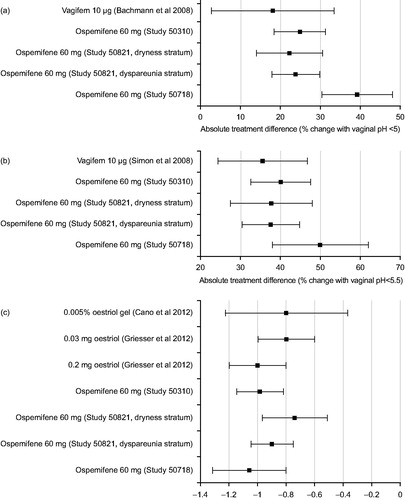
In the Simon 2008 studyCitation14, only data on the proportion of women with a pH <5.5 were reported. The percentages of women achieving a vaginal pH <5.5 with ospemifene relative to placebo and 10 μg 17β-estradiol vs. placebo are shown in .
In the two estriol studies, only change from baseline pH (observed case data) was reported. The placebo-subtracted mean changes from baseline pH with both doses of estriol pessary and with estriol gel were not different to those observed with 60 mg ospemifene ().
Maturation value
The MV was derived from the maturation index according to the formula: MV = 1 × percentage of superficial cells + 0.5 × percentage of intermediate cells.
The improvements in MV vs. placebo over 12 weeks for women treated with 60 mg ospemifene and 10 μg 17β-estradiol in the Simon 2008 studyCitation14 (last observation carried forward), estriol pessary/gel in the GriesserCitation15 and CanoCitation16 studies (observed case data), and 10 μg 17β-estradiol in the Bachmann studyCitation13 are shown in .
Safety comparison
Vaginal bleeding
Three local estrogen studiesCitation21,Citation23,Citation28 reported data on vaginal bleeding, although they have limited comparability to ospemifene data (open label or inspection-only). The rate of vaginal bleeding per 1000 women-years was 21.72 (95% CI 10.41–39.94) for 60 mg ospemifene and 26.34 (95% CI 8.55–61.46) for placebo. There is no increase in the rate of vaginal bleeding for ospemifene compared with placeboCitation10.
Endometrial thickness and biopsy
Endometrial thickness after 1 year was reported in three local estrogen studies (). All showed that mean endometrial thickness remained unchanged over 1 year of treatment, whereas there was a small increase over 52 weeks in mean endometrial thickness of women treated with 60 mg ospemifene ().
Table 5. Change in endometrial thickness. Data are given as mean ± standard deviation.
Endometrial biopsies of women at 12 months were reported in two studies on 10 μg 17β-estradiolCitation26 (see Table S2, Supplementary Material, http://dx.doi.org/10.1080/13697137.2017.1284780). The 1-year histology data for both products were not significantly different from the baseline data (Table S2, Supplementary Material, http://dx.doi.org/10.1080/13697137.2017.1284780).
There was one event of carcinoma and one complex hyperplasia without atypia with 10 μg 17β-estradiol in the Simon 2010 studyCitation26, but no carcinoma with 60 mg ospemifene in any of the studies (Table S2, Supplementary Material, http://dx.doi.org/10.1080/13697137.2017.1284780).
Breast safety
There were no cases of breast cancer in the ospemifene population, but one case of carcinoma-in-situ in the placebo groupCitation30. Breast cancer risk estimates for estrogens were drawn from systemic exposure and it is not known how these apply to local treatments. None of the local estrogen studies reported comparable data on breast safety. A history of breast cancer is a contraindication for all local estrogensCitation31, but once (adjuvant) treatment has been completed, women with a history of breast cancer can use ospemifeneCitation10.
Venous thromboembolism
Hormone replacement therapy (HRT) is associated with a 1.3–3-fold risk of developing VTE, i.e. deep vein thrombosis or pulmonary embolism. The occurrence of such an event is more likely in the first year of HRT than laterCitation31.
There were no cases of VTE in the 10 μg 17β-estradiol clinical database, but the 95% CI provides an indication of the uncertainty due to the small size of the database (upper CIs were calculated for risk using Poisson distribution and test-based methodsCitation32). The incidence of VTE for ospemifene (3.65/1000 women-years) was comparable to the incidence in the placebo population (3.66/1000 women-years)Citation10. Despite the absence of any VTEs in the 10 μg 17β-estradiol database, the CIs for the incidence proportion (IP, in %) of VTE for 10 μg 17β-estradiol (95% CI 0–1.23) were not different to the CIs for the IP of VTE for 60 mg ospemifene (95% CI 0.020–0.581).
Adverse events
The 52-week placebo-controlled study with 10 μg 17β-estradiolCitation14 can be considered the most comprehensive for comparison of AEs, while reports from open-label studies may not be comparable. Most of the other published local estrogen studies either did not report on AEs or reported AEs very selectively.
Hot flushes Although hot flushes have been observed with a higher frequency in patients treated with 10 μg 17β-estradiol compared with placebo, the incidence remains below 1%Citation31. As with other SERMs, there was a trend towards a higher incidence of hot flushes reported as an AE in women treated with 60 mg ospemifene over 52 weeks compared with placeboCitation18.
Vaginal candidiasis For both ospemifene and 10 μg 17β-estradiol, there was a comparable higher incidence of vaginal candidiasis in the active arms than in the placebo arms: 8.3% vs. 2.9% for 10 μg 17β-estradiol and placebo, respectivelyCitation14 compared with 7.7% vs. 1.6% for 60 mg ospemifene and placebo, respectivelyCitation18.
Vaginal discharge The incidences of vaginal discharge in the 60 mg ospemifene and placebo groups were 5.5% and 0%, respectively, in the Goldstein studyCitation18 and 1.4% and 0%, respectively, in study 15-50310/15-50310x. Vaginal hemorrhage, vaginal discharge, or discomfort have been reported in up to 10% of patients using 10 μg 17β-estradiolCitation31,Citation33.
Muscle spasms Muscle spasm is a side-effect that has been observed with other SERMs on the marketCitation34–36. In study 15-50310/15-50310x, the incidence of muscle spasm was 1.4% in the 60 mg ospemifene group and 0% in the placebo group; the corresponding values in study 15-50718 were 8.5% and 6.5%, respectively. The majority of muscle spasms in the ospemifene phase 2/3 study program were reported as leg cramps (50/68, 74%) with 96% reported as mild (n = 40) or moderate (n = 25). No data on muscle spasms could be found for local estrogens.
Headache One local estrogen study reported data on headache, but was not placebo-controlledCitation27. The incidence of headache was lower for 60 mg ospemifene vs. placebo in studies 15-50310/15-50310x and 15-50718Citation18. In the list of AEs observed with a higher frequency in patients treated with 10 μg 17β-estradiol compared with placebo, it is reported that headache occurred in 1–10% of subjects using 10 μg 17β-estradiolCitation31,Citation33.
Discussion
Due to the absence of a direct comparison between ospemifene and local estrogens, we performed an historical indirect comparison of the efficacy and safety of ospemifene with local estrogens in the treatment of VVA. We found that the magnitudes of changes relative to placebo in both subjective and objective efficacy measures were similar to or greater with 60 mg ospemifene than those observed with 10 μg 17β-estradiol or estriol. The improvements in placebo-subtracted composite symptom scores at week 12 were similar for ospemifene vs. 10 μg 17β-estradiol (MBS) and for ospemifene vs. estriol gel 0.005%. The percentages of women achieving a pH <5 or <5.5 with 60 mg ospemifene relative to placebo were greater than that observed with 10 μg 17β-estradiol relative to placebo. In the two estriol studies, we found that placebo-subtracted mean changes from baseline pH with an estriol pessary or estriol gel were comparable to those observed with ospemifene. MV improvements relative to placebo were similar or greater after 12 weeks for 60 mg ospemifene vs. 10 μg 17β-estradiol and vs. estriol pessary/gel. These data suggest that the non-estrogen, ospemifene, is at least as effective as local estrogens in the management of postmenopausal women with VVA symptoms. All treatments included in the analyses were generally well tolerated, with comparable tolerability/safety profiles. The safety of ospemifene compared with local estrogens over 1 year appeared to be comparable in terms of endometrial histology, breast changes, vaginal discharge, and vaginal candidiasis. With the exception of one report of endometrial cancer that was possibly related to 10 μg 17β-estradiol, there was no evidence of an increased risk of cancer, including breast cancer.
The uncertainty around the risk of thrombosis also appeared to be no different to that with local estrogens.
Unfortunately, only the 52-week placebo-controlled study with 10 μg 17β-estradiolCitation14 was suitable for a comprehensive comparison of AEs – the majority of published local estrogen studies either did not report AEs or reported AEs very selectively. Based on the available data, the incidence of headache compared with placebo was not increased with ospemifene, whereas it was with 10 μg 17β-estradiol, which confirms systemic absorption. The incidence of vaginal candidiasis was not different between ospemifene and 10 μg 17β-estradiol.
As noted with other SERMs, the incidence of muscle spasm was higher with 60 mg ospemifene than with placebo and was not reported in the local estrogen publications. The majority of muscle spasms observed in the ospemifene phase 2/3 study program were reported as mild or moderate leg cramps.
Although the methods used in these analyses were as rigorous as possible, this indirect historical comparison does have limitations. The number of studies included in the analysis was small, particularly for efficacy vs. local estrogens. This was expected given the length of time that the products have been on the market in Europe – older studies are rarely of a standard consistent with that needed for a current license submission (in terms of trial design, endpoints, size, level of detail, etc.). Furthermore, there were some differences in study designs and baseline characteristics between studies. For example, lubricant was provided to women in both the ospemifene and placebo arms in the ospemifene studies (a specification of the FDA), thus reducing discomfort due to mild dryness, but does not appear to have been given in the local estrogen studies. Finally, MVs at baseline varied across studies – high baseline MV reduces the amount of improvement that can be made, compared with a lower baseline MV, so the results should be interpreted with caution.
The results of this indirect comparison suggest that the magnitude of the clinical effect observed with ospemifene is comparable to, or better than, that seen with local vaginal estrogens. The safety/tolerability profiles of ospemifene and local vaginal estrogens appear to be similar. The analysis suggests that, for ospemifene indicated in postmenopausal women with VVA who are not candidates for local estrogens, similar efficacy to that observed with local vaginal estrogens can be expected, with a comparable, but slightly different safety profile. Most local estrogens are recommended in the lowest dose for the shortest duration possible. However, ospemifene can be used as long as the benefit outweighs the risk, subject to careful appraisal that is undertaken at least annually.
Conflict of interest
Nico Bruyniks is a consultant for Shionogi Ltd. Nicoletta Biglia is a member of advisory boards and/or consultant with Gedeon Richter, Italfamarco, and Shionogi Ltd. Santiago Palacios acted as a consultant and advisory board member for Servier, Pfizer, GlaxoSmithKline, Abbott, Ferrer, Bioiberica, Shionogi, and Amgen. He has also received grant/research support from Pfizer, Servier, Amgen, Merck, Sharp & Dohme, Preglem, Leon Farma, Gynea, Sandoz, Bayer, and Amgen. Alfred O. Mueck has received funding for research, advisory, and educational activities from manufacturers of HRT, including local estrogen therapy. Financial support was given from Abbott, Bayer, Besins, Dr. Kade, Exeltis, Gedeon Richter, Hexal, HRA, Jenapharm, MSD, Novartis, Pierre Fabre, Pfizer, Serelys, Shionogi, Solvay, TEVA, and Theramex. He serves on the board of several societies and journals covering the field of sexual steroids.
1284780_Suppl_data.doc
Download MS Word (154.5 KB)Acknowledgements
Tasaki Takenobu and Yoshida Yuki of the Biostatistics department of Shionogi & Co Ltd, Osaka, Japan performed the statistical analyses. Susan Crawford of Absolute Healthcare Communications, Ltd, Twickenham, UK assisted in compiling and addressing author comments and was funded by Shionogi Ltd. London, UK.
References
- MacBride MB, Rhodes DJ, Shuster LT. Vulvovaginal atrophy. Mayo Clin Proc 2010;85:87–94
- Archer DF. Efficacy and tolerability of local estrogen therapy for urogenital atrophy. Menopause 2010;17:194–203
- Goldstein IJ. Recognizing and treating urogenital atrophy in postmenopausal women. Womens Health (Larchmt) 2010;19:425–32
- Parish SJ, Nappi RE, Krychman ML, et al. Impact of vulvovaginal health on postmenopausal women: a review of surveys on symptoms of vulvovaginal atrophy. Int J Womens Health 2013;5:437–47
- North American Menopause Society. Management of symptomatic vulvovaginal atrophy: 2013 position statement of The North American Menopause Society. Menopause 2013;20:888–902
- Rahn DD, Carberry C, Sanses TV, et al. Society of Gynecologic Surgeons Systematic Review Group. Vaginal estrogen for genitourinary syndrome of menopause: a systematic review. Obstet Gynecol 2014;124:1147–56
- Palacios S, Castelo-Branco C, Currie H, et al. Update of management of genitourinary syndrome of menopause. A practical guide. Maturitas 2015;82:308–13
- Kingsberg SA, Wysocki S, Magnus L, Krychman ML. Vulvar and vaginal atrophy in postmenopausal women: findings from the REVIVE (REal Women’s VIews of Treatment Options for Menopausal Vaginal ChangEs) survey. J Sex Med 2013;10:1790–9
- Nappi RE, Palacios S, Panay N, Particco M, Krychman ML. Vulvar and vaginal atrophy in four European countries: evidence from the European REVIVE Survey. Climacteric 2016;19:188–97
- Senshio (ospemifene) tablets. European Public Assessment Report. European Medicines Agency. London November 2014. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/002780/WC500182777.pdf [last accessed 6 Sept 2016]
- Rutanen EM, Heikkinen J, Halonen K, Komi J, Lammintausta R, Ylikorkala O. Effects of ospemifene, a novel SERM, on hormones, genital tract, climacteric symptoms, and quality of life in postmenopausal women: a double-blind, randomized trial. Menopause 2003;10:433–9
- DeGregorio MW, Zerbe RL, Wurz GT. Ospemifene: a first-in-class, non-hormonal selective estrogen receptor modulator approved for the treatment of dyspareunia associated with vulvar and vaginal atrophy. Steroids 2014;90:82–93
- Bachmann G, Lobo RA, Gut R, Nachtigall L, Notelovitz M. Efficacy of low-dose estradiol vaginal tablets in the treatment of atrophic vaginitis: a randomized controlled trial. Obstet Gynecol 2008;111:67–76
- Simon J, Nachtigall L, Gut R, Lang E, Archer DF, Utian W. Effective treatment of vaginal atrophy with an ultra-low-dose estradiol vaginal tablet. Obstet Gynecol 2008;112:1053–60
- Griesser H, Skonietzki S, Fischer T, et al. Low dose estriol pessaries for the treatment of vaginal atrophy: a double-blind placebo-controlled trial investigating the efficacy of pessaries containing 0.2 mg and 0.03 mg estriol. Maturitas 2012;71:360–8
- Cano A, Estévez J, Usandizaga R, et al. The therapeutic effect of a new ultra low concentration estriol gel formulation (0.005% estriol vaginal gel) on symptoms and signs of postmenopausal vaginal atrophy: results from a pivotal phase III study. Menopause 2012;19:1130–9
- Bachmann GA, Komi JO; Ospemifene Study Group. Ospemifene effectively treats vulvovaginal atrophy in postmenopausal women: results from a pivotal phase 3 study. Menopause 2010;17:480–6
- Goldstein SR, Bachmann GA, Koninckx PR, Lin VH, Portman DJ, Ylikorkala O; Ospemifene Study Group. Ospemifene 12-month safety and efficacy in postmenopausal women with vulvar and vaginal atrophy. Climacteric 2014;17:173–82
- Portman DJ, Bachmann GA, Simon JA. Ospemifene, a novel selective estrogen receptor modulator for treating dyspareunia associated with postmenopausal vulvar and vaginal atrophy. Menopause 2013;20:623–30
- Portman D, Palacios S, Nappi RE, Mueck AO. Ospemifene, a non-oestrogen selective oestrogen receptor modulator for the treatment of vaginal dryness associated with postmenopausal vulvar and vaginal atrophy: a randomised, placebo-controlled, phase III trial. Maturitas 2014;78:91–8
- Henriksson L, Stjernquist M, Boquist L, Cedergren I, Selinus I. A one-year multicenter study of efficacy and safety of a continuous, low-dose, estradiol-releasing vaginal ring (Estring) in postmenopausal women with symptoms and signs of urogenital aging. Am J Obstet Gynecol 1996;174:85–92
- Naessen T, Rodriguez-Macias K. Endometrial thickness and uterine diameter not affected by ultralow doses of 17beta-estradiol in elderly women. Am J Obstet Gynecol 2002;186:944–7
- Weisberg E, Ayton R, Darling G, et al. Endometrial and vaginal effects of low-dose estradiol delivered by vaginal ring or vaginal tablet. Climacteric 2005;8:83–92
- Gerbaldo D, Ferraiolo A, Croce S, Truini M, Capitanio GL. Endometrial morphology after 12 months of vaginal oestriol therapy in post-menopausal women. Maturitas 1991;13:269–74
- Iosif CS. Effects of protracted administration of estriol on the lower genitourinary tract in postmenopausal women. Arch Gynecol Obstet 1992;251:115–20
- Simon J, Nachtigall L, Ulrich LG, Eugster-Hausmann M, Gut R. Endometrial safety of ultra-low-dose estradiol vaginal tablets. Obstet Gynecol 2010;116:876–83
- Ulrich LS, Naessen T, Elia D, Goldstein JA, Eugster-Hausmann M. Endometrial safety of ultra-low-dose Vagifem 10 μg in postmenopausal women with vaginal atrophy. Climacteric 2010;13:228–37
- Smith P, Heimer G, Lindskog M, Ulmsten U. Oestradiol-releasing vaginal ring for treatment of postmenopausal urogenital atrophy. Maturitas 1993;16:145–54
- Simon JA, Lin VH, Radovich C, Bachmann GA; Ospemifene Study Group. One-year long-term safety extension study of ospemifene for the treatment of vulvar and vaginal atrophy in postmenopausal women with a uterus. Menopause 2013;20:418–27
- Berga SL. Profile of ospemifene in the breast. Reprod Sci 2013;20:1130–6
- Vagifem summary of product characteristics. Vagifem 10 micrograms vaginal tablets. January 2015. Available from: https://www.medicines.org.uk/emc/print-document?documentId=23819 [last accessed 5 Sept 2016]
- Sahai H, Khurshid A. Formulas and tables for the determination of sample sizes and power in clinical trials for testing differences in proportions for the matched pair design: A review. Fundam Clin Pharmacol 1996;10:554–63
- Medicines and Healthcare products Regulatory Agency. Public assessment report. Vagifem 10 micrograms vaginal tablets. February 2010. Available from http://www.mhra.gov.uk/home/groups/par/documents/websiteresources/con078939.pdf [last accessed 5 Sept 2016]
- Nolvadex (tamoxifen) tablets. Summary of product characteristics. Astra Zeneca UK Ltd, Luton, UK. May 2015. Available from: http://www.medicines.ie/medicine/4181/SPC/Nolvadex+D+20mg+Tablets/ [last accessed 5 Sept 2016]
- Conbriza (bazedoxifene) tablets. Summary of product characteristics. Pfizer Limited, Sandwich, Kent, UK. April 2014. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000913/WC500033577.pdf [last accessed 5 Sept 2016]
- Raloxifene Teva (raloxifene hydrochloride) tablets. Summary of product characteristics. Teva B.V., Haarlem, The Netherlands. February 2015. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/001075/WC500091420.pdf [last accessed 5 Sept 2016]