ABSTRACT
Introduction: Early dry powder inhalers (DPIs) were designed for low drug doses in asthma and COPD therapy. Nearly all concepts contained carrier-based formulations and lacked efficient dispersion principles. Therefore, particle engineering and powder processing are increasingly applied to achieve acceptable lung deposition with these poorly designed inhalers.
Areas covered: The consequences of the choices made for early DPI development with respect of efficacy, production costs and safety and the tremendous amount of energy put into understanding and controlling the dispersion performance of adhesive mixtures are discussed. Also newly developed particle manufacturing and powder formulation processes are presented as well as the challenges, objectives, and new tools available for future DPI design.
Expert opinion: Improved inhaler design is desired to make DPIs for future applications cost-effective and safe. With an increasing interest in high dose drug delivery, vaccination and systemic delivery via the lungs, innovative formulation technologies alone may not be sufficient. Safety is served by increasing patient adherence to the therapy, minimizing the use of unnecessary excipients and designing simple and self-intuitive inhalers, which give good feedback to the patient about the inhalation maneuver. For some applications, like vaccination and delivery of hygroscopic formulations, disposable inhalers may be preferred.
1. Introduction
The history of inhalation therapy has been described before [Citation1–Citation4]. It dates back to more than 4000 years ago when in India powdered Durata stramonium and Durata ferox, containing potent alkaloids with anticholinergic bronchodilating properties, were smoked after mixing with other compounds such as ginger and pepper [Citation1]. Drug inhalation gained interest for the treatment of respiratory diseases particularly after Philip Stern acknowledged in 1764, ‘the only possible way of applying medicines directly to the lung is through the windpipe’ [Citation4]. This resulted in the development of a variety of ceramic inhalers in the eighteenth century for drawing the inhaled air through infusions of plants and other ingredients [Citation1]. In the early 1900s, hand-bulb nebulizers became popular for supplying adrenaline chloride as bronchodilator, and in the twentieth century, electric and ultrasonic nebulizers were developed [Citation1,Citation2]. In 1956, a completely new principle for wet aerosol generation was introduced: the ‘Metered Dose Inhaler’ (MDI, Riker Laboratories), driven by propellants [Citation2]. The MDI became rapidly popular because this type of device is small (portable), inexpensive, theoretically easy to use, fast, and silent compared to nebulizer equipment. In spite of their advantages over nebulizers, many patients had difficulties in operating an MDI correctly. In addition, a great concern about the future for MDIs was raised on account of the discovery in the early 1970s that CFC propellants contribute to the depletion of the ozone layer in the stratosphere [Citation5,Citation6]. This discovery resulted in the Montreal protocol on substances that deplete the ozone layer, which entered into force in January 1989 to phase out production of the substances that are responsible for this depletion [Citation7,Citation8]. The uncertain situation about MDIs became a major driver for dry powder inhalation, and dry powder inhalers (DPIs) are currently considered as the type of pulmonary drug administration device with the greatest potential for improved and new therapies.
The aim of this review was to evaluate the past, present, and (expected) future of DPI development. It will be discussed that a lot of effort is currently put in formulation research to obtain acceptable performance with poorly designed DPI concepts from the past. For DPI innovation in the future, breaking with this tradition is necessary. Safer, simpler, cheaper, and yet more effective DPIs are necessary to make high expectations about mass vaccination and TB therapy, particularly in developing countries, come true.
2. Dry powder inhalation: past
DPIs are already known since mid-eighteenth century from Vincent Alfred Newton’s UK patent 1161 [Citation9]. His device, meant to deliver pulverized potassium chloride, was never manufactured on an industrial scale, however. This, in contrast with the Aerohalor of Abbott, which was launched nearly 100 years later in 1948 and used for the delivery of penicillin and norethisterone, a bronchodilator [Citation4]. The Aerohalor served as a prototype for the various capsule inhalers developed in the period of 1950–1980. These inhalers contained a lactose-based drug formulation in small capsules or ‘sifters’. For the development of the first device marketed in this period, the Fisons Spinhaler® (1967), the high dose (20 mg of cromoglycate sodium, which is too high for MDIs) was the main driver [Citation4,Citation10]. Bell et al. tested a lactose-based formulation (1:1 with the drug by weight) for this drug too and concluded that the emptying efficiency of the capsule depends very much on the size fraction of the lactose used [Citation11]. Several patents for different inhaler concepts were filed between 1950 and 1980 e.g. [Citation12–Citation17], but only the inventions that made use of the same basic design principles reached the market. They all had gelatin capsules as dose container and a lactose (carrier)-based formulation for the drug [Citation18]. Differences were primarily confined to the means for opening or piercing the capsule and the way in which the capsule is spinning, vibrating, or wobbling to release its contents during inhalation. In 1969, the Rotahaler® was launched (Allen & Hanbury’s) [Citation19], followed by the Inhalator Ingelheim in the 1980s (Boehringer Ingelheim) [Citation20] and the Cyclohaler® in the early 1990s (Pharmachemie) [Citation21]. The latter is also known as the ISF®-inhaler and was patented in 1974 [Citation14]. The choice for hard gelatin capsules as dose compartment for the drug seemed obvious, as filling equipment for this dosage form was available and did not need to be developed. Drugs against asthma and chronic obstructive pulmonary disease in the microgram range, for which the capsule inhalers were developed, have to be diluted, and their flow properties improved for reproducible capsule filling. Therefore, using carrier-based formulations for such low doses was a logical choice too. Jones and Pilpel were among the first to describe the strong adhesion of micronized particles to the surfaces of much larger host (carrier) particles during mixing [Citation22], and this phenomenon was studied more systematically in 1971 by Shotton and Orr [Citation23] and Travers and White [Citation24]. It was recognized by Hersey in 1975 that this type of mixing is basically different from random mixing, and for that reason, he named it ordered mixing [Citation25]. More than 10 years later and after intensive debate about the nomenclature, Staniforth (1987) presented very good arguments to replace the name ‘ordered’ by ‘adhesive’ [Citation26]. In tablet manufacturing for very potent drugs, the objective was to obtain maximal mixture homogeneity and stability. First, when the relevance of adhesive mixtures to inhalation increased, improving dislodgement of the micronized drug particles from the carrier crystals during inhalation became the main objective (see paragraph 3.1). After the first capsule-based DPIs, multidose reservoir inhalers were introduced from the 1990s onwards, having the same adhesive powder formulations for the drug. They were designed with various dose measuring principles, including slides (e.g. Novolizer®, Meda Pharma) [Citation27–Citation29], cylinders (e.g. Easyhaler®, Orion Pharma) [Citation30], and disks (e.g. Pulvinal®, Chiesi Farmaceutici) [Citation31] with dose cavities, or hollow shafts (e.g. Taifun®, Leiras Oy) [Citation32,Citation33], operated by the patient. Similar multidose DPIs developed in the same period are the Twisthaler® (Merck,Sharp & Dohme Ltd) [Citation34], Clickhaler® (Vectura) [Citation35], SkyeHaler® (SkyePharma), marketed as Certihaler® by Novartis [Citation36], and Airmax® (Norton Healthcare) [Citation37], the latter currently known as Spiromax® (TEVA) [Citation38]. Adhesive mixtures were also applied in the first multiple unit-dose blister inhalers from the 1990s, the Diskhaler® [Citation39] and Diskus® [Citation40] of Glaxo(Smith Kline). The only exception to the rule is the Turbuhaler®, introduced by Astra(Zeneca) in the late 1980s [Citation41]. Micronized drugs for delivery with this multidose reservoir inhaler are transformed by spheronization into soft aggregates (originally without micronized lactose) to obtain a carrier-free formulation. Nearly all DPIs developed before 2010 were so-called passive, or breath-actuated devices.
3. Dry powder inhalation: present
New inhaler concept developments may easily stretch over periods of 10-15 years. What was started several years ago may not reach the patient in the next half a decade. The patent literature may tell what is going on but cannot reveal which developments will be successful; only few of the patented inventions reach the market. Therefore, the boundary between past and present is blurred, and for the sake of continuity, the period from approximately 1990 onwards will be considered (at least partly) as the present. Currently, at least five mainstream developments can be distinguished related primarily to dry powder inhalation:
‘repair actions’ for the design weaknesses of the early inhaler concepts
formulation of high-dose drugs (‘particle engineering’)
development of inhalers and formulations for vaccines and systemically acting drugs
understanding (and controlling) pulmonary drug deposition and distribution from DPIs
miscellaneous innovative developments.
Additionally, one of the main concerns in relation to the use of these breath actuated DPIs is that patients may not be able to generate a sufficient flow rate for extracting the dose and good powder dispersion and hence, achieving the desired therapeutic effect [Citation42]. This resulted in the development of active DPIs, like the inhalers of the Spiros® platform (Dura Pharmaceuticals), which have a battery-operated propeller [Citation43] and the Exubera® insulin inhaler (Pfizer) which utilizes pressurized air from a hand piston for active powder dispersion [Citation44]. Such inhalers basically deliver a patient (i.e. flow rate) independent fine particle aerosol [Citation45], which has long been considered an advantage [Citation46,Citation47]. An increasing delivered fine particle dose with increasing flow rate can compensate for the increased oropharyngeal losses, however, thus yielding a more constant lung deposition and higher distribution uniformity [Citation29,Citation48–Citation50]. Besides, both active inhalers were not successfully marketed. They were complex and, therefore, expensive, vulnerable to failure in case of flat batteries (Spiros), and user-unfriendly because of the large number of operational steps and the large size of the apparatus (Exubera). For these reasons, active DPIs will not further be discussed in this review.
Finally, the therapeutic and commercial success of combination products results in one of the most noticeable therapy changes of the present [Citation51]. This also concerns dry powder inhalation.
3.1. ‘Repair actions’ for design weaknesses of early DPI concepts
The initiative to develop DPIs may have been the result of some well-defined needs [Citation18,Citation42,Citation52–Citation54]; the choices made for their design indicate that the development itself was driven by existing expertise. The availability of capsule filling equipment and the emerging expertise in dry powder formulation of potent drugs enabled to keep DPI development cheap and simple and made DPIs cost compatible with MDIs. However, currently, these choices can be considered the DPI design weaknesses. Both in vitro and in vivo deposition studies show that delivered drug fractions in the required aerodynamic size fraction from early capsule-based DPIs with adhesive mixtures are quite low () [Citation55,Citation56]. The carrier-free formulation from the Turbuhaler generally performs much better, even in comparison with MDIs that deliver the same drug [Citation57,Citation58]. The explanation for this poor performance of the early capsule-based DPIs is in the strong interparticulate forces in the adhesive mixtures and the absence of efficient dispersion principles in these inhalers. To have some dispersion of the formulation at all, the inhaler resistance is kept low to enable high flow rates for sufficient dispersion energy [Citation59]. This has the consequence of high mouth–throat depositions [Citation60,Citation61]. As a response to the inefficient dispersion of adhesive mixtures, an abundance of studies into understanding and controlling the variables and mechanisms involved in the drug–drug and drug–carrier interaction (and dispersion) was started. Many of these studies have recently been reviewed [Citation62,Citation63], and most of them focus on selecting the most appropriate carrier size fraction [Citation64,Citation65], controlling or modifying the carrier (surface) morphology [Citation66–Citation68], and varying the amount of (drug and/or excipient) fines in the mixture [Citation69,Citation70]. Additionally, the effects of carrier shape, grade, and type and the influence of the mixing conditions on the dispersibility of the inhalation powders have been studied intensively [Citation63]. An interesting theoretical approach for a better understanding of the performance of carrier-based formulations is also the cohesion–adhesion balance [Citation71]. According to this approach, drug particles exhibit a preference for attachment to each other, resulting in drug particle agglomeration, or to the carrier surface, depending on the balance between cohesiveness to adhesiveness of the particles present in the mixture. When the ratio of cohesion to adhesion is >1, a tendency for drug particle agglomeration during blending exists, which promotes dispersion during inhalation. None of the aforementioned studies has really resulted in significant improvement so far, however. Complicating factors are not only the many variables involved, but in particular, also the interactions between these variables. Mostly, only one or two of these variables are controlled and studied for their effect, but when (some of) the other variables are chosen differently between studies, comparison of data is difficult, and seemingly contradicting results may be obtained [Citation62,Citation63]. Therefore, alternative approaches have been introduced. As has been mentioned already, the use of a lactose carrier has been avoided for the formulations in the Turbuhaler [Citation41]. An alternative way to refrain from the use of a particulate carrier is to store the drug on a microstructured carrier tape [Citation72]. This Taper DPI (3M™) is another example of an active inhaler releasing the dose by vibration of the tape [Citation73]. Jethaler® (Ratiopharm) and MAGhaler® (Mundipharma) are DPIs with the carrier-based drug formulation compacted by isostatic pressure technique into a ring-shaped tablet [Citation74]. Measuring a dose is performed by scraping an amount of powder from the ring, and the high mechanical forces also overcome part of the interparticulate forces between the carrier and the drug [Citation75]. In addition to controlling the carrier (surface) morphology, also drug particle engineering processes have been applied. Examples are co-crystallization of drugs and excipients by standard spray drying [Citation76], spray drying using supercritical enhanced atomization [Citation77], or supercritical fluids crystallization (SEDs: solution-enhanced dispersion by supercritical fluids) [Citation72,Citation78]. Such particles generally exhibit improved dispersion performance [Citation79,Citation80]. Another approach to obtain better dispersion performance of adhesive mixtures finds its origin in the work of Staniforth [Citation81]. He used small amounts (not more than 0.1–5%) of antiadherent or antifriction additive in carrier-based mixtures to reduce the adhesive force between the carrier and active particles. This concept has been elaborated into the PowderHale® technology by the University of Bath and Vectura, using magnesium stearate or leucine as additives, later referred to as Force Control Agents (FCAs). The technology, based on a dry mechanical fusion (mechanofusion) process [Citation82,Citation83], is licensed by Vectura to GlaxoSmithKline (for the formulations in the Ellipta®) [Citation84] and Novartis (for the Breezhaler®) [Citation85]. Also the Chiesi powder formulation for beclomethasone dipropionate and formoterol fumarate (Foster, or Fostair), administered with the NEXThaler® device, contains magnesium stearate. Chiesi was granted their own proprietary technology on the use of magnesium stearate on the basis of improved dispersion as well as improved resistance to moisture which leads to a higher physical and chemical stability of the powder mixture [Citation86,Citation87].
Figure 1. In vitro delivered fine particle doses as percent of label claim (FPFs < 5 µm) from various DPIs tested at 4 kPa (high resistance devices) or 2 kPa (low resistance devices): range of flow rates 40–75 L/min using a Next Generation Impactor. Data derived from different comparative in vitro evaluation studies performed by the authors at the same conditions for direct comparision. Grey bars are for inhalers carrying adhesive mixtures; dark grey bars with force control agents in the formulation. Shaded bars are for carrier free (Turbuhaler) formulations. FPFs are for budesonide (Easyhaler, Cyclohaler, M2 and M3 Turbuhaler and Novolizer), fluticason propionate (Diskus and Elpenhaler), beclometason dipropionate (NEXThaler), aclidinium bromide (Genuair), tiotropium bromide (Handihaler), indacaterol maleate (Breezhaler) and disodium cromoglycate (Spinhaler).
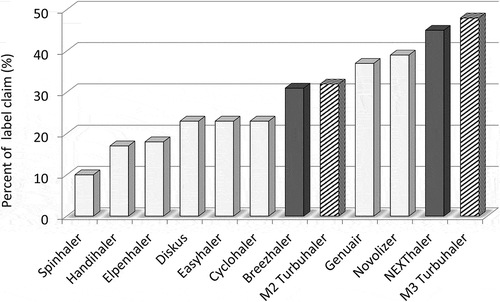
All previously mentioned (drug and carrier) particle modifying and engineering techniques and the addition of FCAs are meant to decrease the high interparticulate forces in adhesive mixtures for inhalation. This is needed because of the absence of efficient dispersion principles in most early inhaler concepts, and these developments may, therefore, be considered ‘repair actions’ for poor inhaler device design. The NEXThaler device also delivers a so-called ‘extrafine beclomethasone and formoterol aerosol’ with particles micronized to a mass median aerodynamic diameter (MMAD) <1.5 μm. Chiesi claims to achieve higher lung deposition with better penetration into the small airways with such particles, compared to inhalation powders with a larger MMAD, due to lower oropharyngeal losses [Citation88,Citation89]. The magnesium stearate in the formulation facilitates effective dispersion of such small drug particles. The delivery of such extrafine aerosols is in essence to compensate for the high flow rate from (most) early DPIs. All these repair actions for the weaknesses of early DPI design make the drug formulations complex, more expensive, and less safe due to the use of excipients. This could have been avoided by designing more effective inhaler devices instead. It should, therefore, be recommended that in the future, more energy is put in designing better devices as part of formulation-device integrated developments.
3.2. Formulation of high-dose drugs
The problems encountered with high-dose drug formulations for inhalation are essentially the same as those with adhesive mixtures, because they are tested and administered with the same capsule-based inhalers such as Spinhaler, Aerolizer®, Eclipse®, and Turbospin® [Citation90–Citation95]. The interparticulate cohesive forces are too high, which makes high flow rates necessary for acceptable dispersion, whereas capsule emptying can be problematic too. It may, therefore, not be surprising that at least partly the same powder processing and particle engineering techniques as used for low doses are also applied for dispersion improvement of high-dose drug formulations. Improvement can often be obtained by preparing low-density (high-porosity) particles. Different methods can be applied to decrease particle density, such as creating hollow particles (void space on the inside), particles with corrugated or wrinkled surfaces (void space on the outside), or solid foam particles. The conditions to obtain such particles by spray drying and the excipients needed have been explained in an excellent review by Vehring [Citation96]. Such particles decrease the powder bulk density (i.e. the number of contact points per particle) and also have a smaller surface area per contact point, which decreases the number and magnitude of the interparticulate forces in the powder. One of the most successful technologies in this respect is PulmoSphere™ (Novartis, developed by Nektar Therapeutics) [Citation95]. Particle engineering is also needed to achieve stabilization of large molecules (biopharmaceuticals) in the dry state with sugar glasses (cryoprotectants) [Citation97–Citation101], prolonged drug release or therapeutic effect (e.g. by mucoadhesion) [Citation102–Citation107], and enhanced drug absorption [Citation106,Citation108]. Particle processing can furthermore be used to prevent or retard moisture uptake by hygroscopic drugs [Citation94,Citation109,Citation110], to escape macrophage clearance [Citation111], or, in contrast, to target specifically the alveolar macrophages [Citation112,Citation113], even though the conditions that influence particle uptake by macrophages are still widely unknown [Citation114]. Also, particle coprocessing with hygroscopic excipients, such as sodium chloride and mannitol, to achieve excipient enhanced particle growth may be desired for enhanced deposition in the deep lung [Citation115,Citation116]. Moisture absorption by such particles during transport through the airways results in a higher particle mass, and this increases the sedimentation velocity. The plethora of publications regarding these particle processing techniques for inhalation powders makes it virtually impossible to provide a complete survey, but interesting reviews on formulation strategy and excipient use for (high dose) inhalation powders were recently written by Pilcer and Amighi [Citation117] and Hoppentocht et al. [Citation118,Citation119].
When previously mentioned effects such as drug stabilization, macrophage targeting, or sustained release are not needed, however, and particle engineering is applied for dispersion improvement only, its use should be critically evaluated for high-dose drugs. Such powders are frequently highly voluminous because of their extreme porosity, particularly when being prepared by freeze drying [Citation120], spray-freeze drying [Citation98,Citation121], or spray drying with volatile or blowing agents [Citation94,Citation122]. The excipients used furthermore increase the dose weight to be administered, and for high-dose drugs, this may also increase the number of inhalations per dose. In some studies, the drug-loaded particles contain only a few percent of the active, which, for the example of inhaled antibiotics, can bring the inhaled powder dose into the gram range [Citation107]. Many particle engineering processes also require multiple unit operations, which makes such powders expensive and increases the risk of batch variation. Finally, a variety of excipients are used of which some may pose an immediate risk to the patient or be harmful in the long term. Only safe excipients should be used in the smallest possible amounts in high-dose drug formulations; even small fractions of an excipient in a high-dose drug formulation may still be administered in significant amounts. Alternatively, dispersion performance improvement can also be achieved with improved inhaler design. For example, recently, it has been shown that the use of excipients may not even be needed for problematic drugs such as amikacin in capsule-based inhalers when the spray drying conditions are optimized to the product [Citation123].
3.3. DPI design and pulmonary drug deposition and distribution
A consistent lung dose is considered a prerequisite for an adequate and efficient therapy. The efficiency (and safety) of pulmonary drug administration depends on the aerosol properties and the way in which the aerosol is delivered to the respiratory tract. The drug formulation and administration device plays crucial but different roles in this. The formulation has to contain the drug particles in the desired aerodynamic size distribution for deposition in the target area. Formulation design may be needed to achieve previously mentioned effects [Citation97–Citation116] or a better product stability. DPI devices need to disperse the formulation effectively into this size distribution, preferably at the lowest possible flow rate within the first 0.5 L (children) or 1 L (adults) of inhaled air to achieve drug distribution over the entire lung. Fine-tuning of the primary particle size distribution and the inhalation maneuver enables to target different sites of action with such an inhaler.
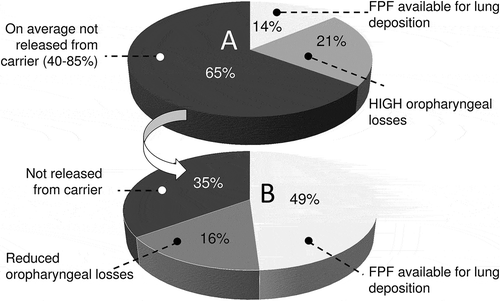
Similar to other pulmonary administration devices, the lung dose from a DPI is considerably lower than the label claim. This is due to inhaler losses and substantial deposition in the mouth–throat region (oropharynx), which often amounts to more than 50% of the delivered dose () [Citation49,Citation50,Citation55,Citation56,Citation61]. does not show the extreme variation in oropharyngeal losses between different inhalers, however, which is strongly influenced by the inhaler resistance. Oropharyngeal losses also vary with the inhalation maneuver through the same inhaler. The variability and uncertainty of lung deposition are mostly the result of variations in extrathoracic deposition, and it has been hypothesized that throat deposition is the major determinant for lung deposition of inhaled aerosols [Citation124]. It may be clear that what is ‘lost’ in the mouth–throat region does not contribute to the therapeutic effect and can cause adverse local (or systemic) side effects. Hence, understanding and reducing mouth–throat deposition are of utmost importance for DPI design.
Figure 2. Fine particle fraction of the delivered dose and part of the delivered dose deposited in the mouth and oropharynx from DPIs carrying adhesive mixtures as drug formulation. A: current situation. The fraction not released from carrier and the fine particle fraction were averaged for the DPIs presented in and the oropharyngeal losses were estimated from in vivo deposition studies with radiolabel drugs in various marketed DPIs. B: desired situation to be obtained by improved dispersion (from 35% to 65% drug detachment) at a lower flow rate (from ≫ 60 L/min to < 40 L/min).
For the experimental assessment of drug deposition in the human throat, different anatomical models are used [Citation125–Citation127] to replace the standard USP induction port for more realistic deposition. Using such a throat, DeHaan and Finlay obtained a sigmoidal (averaged) relationship between extrathoracic (monodisperse) aerosol deposition (from different devices) and the logarithm of an impaction parameter (IP), showing that mouth–throat deposition can approach 100% at high IP values [Citation128]. The two DPIs in their study showed substantially higher deposition than the nebulizers and an MDI (with holding chamber). In a follow-up study, they presented the rather extreme differences in oral cavity deposition for monodisperse aerosol particles (at the same flow rate) between different DPI designs [Citation61]. They concluded that the influence of turbulence on oral deposition is relatively small and that inertial impaction is the major, but not the unique, deposition mechanism in the oral cavity. In a study with an idealized pediatric throat model, the difference in oropharyngeal deposition from different DPIs was confirmed, but the difference between idealized adult and pediatric models was almost negligible [Citation129]. Studies such as these show that DPI design is a major determinant, not only for the properties of the delivered aerosol but also particularly for the ‘losses’ in between the mouthpiece exit and the target area below the trachea. Even for particles with the same aerodynamic diameter released at the same flow rate, differences in extrathoracic deposition may be rather extreme from different DPI concepts [Citation61]. For these reasons, DPI device design should focus on minimizing the extrathoracic losses as well as on reducing the patient variability in the inhalation maneuver. The first is a function of the DPI’s mouthpiece design rather than that of total device design, whereas the latter is served best with a medium to high air flow resistance and an increasing fine particle dose with increasing flow rate to compensate for the higher oropharyngeal losses.
Assessing lung deposition and distribution of the aerosol fraction passing the oropharyngeal region is more challenging than measuring oropharyngeal deposition. Lung deposition has to be known for dose finding, fine-tuning of the particle size distribution of the aerosol, and optimizing the inhalation maneuver for the inhaler used. In contrast with the mouth–throat region, in vitro replicas of the entire bronchial tree do not exist. Therefore, lung deposition assessment relies on other methods such as deposition modeling, pharmacokinetic method, and scintigraphy. Particle deposition modeling in the respiratory tract is based on impaction and sedimentation probability equations for single particles. They are derived for simplified situations [Citation130] and make use of standardized lung models, for instance, as defined by Findeisen or Weibel [Citation131]. Although such computations teach about the effects of particle size and velocity on drug deposition in the lungs, they bear a significant degree of uncertainty for drug deposition in patients, as diseased lungs may deviate considerably from the standardized models (and conditions) used for the computations. For lung deposition studies, radiolabeling techniques have greater value than PK methods, although both have been used successfully to estimate total lung dose [Citation132]. PK data do not allow estimating regional deposition, and the absorbed dose may be underestimated when the drug is metabolized. Pharmacokinetic evaluations are also hindered by a lack of understanding of the relation between deposition and absorption. For example, a high plasma peak concentration (Cmax) may be the result of either a high drug concentration in the upper airways or that of a more even drug distribution with lower concentrations over the central and lower airways. The first situation yields a high driving force for absorption, whereas the second provides a large area for absorption (low resistance). Even more important is the rather extreme intersubject variability in absorption of the drug, as has been found for instance in different studies with tobramycin in different patient groups [Citation95,Citation133,Citation134]. Therefore, PK data may lead to false conclusions about the real dose deposited. Radiolabeling techniques for inhaled aerosols from MDIs were first presented in the 1980s using two-dimensional gamma cameras [Citation135]. They yielded limited information due to an overlay of the structures of interest [Citation136]. Three-dimensional techniques such as single photon emission computed tomography and positron emission tomography (PET) provide more detailed data, although particularly PET poses greater logistical and technical difficulties [Citation136–Citation138]. Also, the lack of standardization and the differences in procedures between different centers have been a concern, as they make comparison of data complex and limit acceptance by the regulatory authorities [Citation137,Citation139–Citation141]. As a response to that, the Regulatory Affairs Networking Group of the International Society for Aerosols in Medicine (ISAM) presented practical guidance to the methods and techniques for standardizing radiolabel validation, image acquisition, and analysis [Citation142]. Recent developments are to coimage anatomical structures from high-resolution computer tomography (CT) scans with deposition data from radiolabeling technique [Citation137,Citation143]. CT scans at functional residual capacity and total lung capacity are also used as boundary conditions for computational fluid dynamics (CFD) simulation to predict flow behavior and drug deposition [Citation144]. It is anticipated that this functional respiratory imaging may become a tool for personalized respiratory treatment [Citation145].
3.4. Development of inhalers and formulations for vaccines and systemically acting drugs
As early as 1958, the feasibility of pulmonary vaccination was presumed when the inhalation of very small numbers of living attenuated Mycobacterium tuberculosis by guinea pigs resulted in development of acquired resistance against airborne infection with virulent bacilli [Citation146]. Today, pulmonary vaccination is still in its infancy, and of all possible applications for pulmonary drug delivery, DPI development for vaccination is probably the most challenging. Pulmonary vaccination differs at least in two distinct ways from the administration of most locally and systematically acting drugs: the delivery is one–off (with at most one or two booster doses after considerable time), and there is no immediate measurable therapeutic response. This implies that (nearly) all vaccinees are inhalation naive, and there is no check on whether the vaccine administration was successful or not. This poses high demands on the delivery system for pulmonary vaccines. DPIs offer the advantage over wet nebulization systems (or MDIs) that the vaccine is administered in the dry state. Therefore, stability is less of an issue, and reconstitution from powdered material is not necessary. But the administration is in one single inhalation act. This requires that the inhaler performs well and consistent over a wide range of flow rates and that operating the inhaler correctly must be intuitive. In addition, practicing of the inhalation maneuver under supervision of a health-care expert with an empty, instrumented inhaler must be recommended, and correct vaccine delivery must be verified with a feedback system showing the relevant inspiratory parameters. Recently, the (pre-)clinical pulmonary vaccine studies were reviewed in which it was shown that inducing an immune response is possible indeed [Citation147,Citation148]. However, not all pulmonary vaccines produced and tested were equally effective in these studies, and this could be due to differences in the formulation properties (particularly the aerodynamic size distribution of the delivered aerosols) and delivery efficiencies from different inhalation devices. More research into the desired site of deposition, the intrinsic immunogenicity of the antigen to activate the innate immune system, and the choice of adjuvants is necessary. Special attention has to be given also to DPI development for school children, after infants and toddlers probably the most relevant age group for pulmonary vaccination. For children, not only respiratory but also cognitive skills may be limiting factors for correct DPI use [Citation149]. It has recently been shown that most children from 5 years on can understand and comply well with the inhalation instruction given [Citation150]. However, inhaled volumes and flow rates were low and require that the majority of the dose is delivered within 0.5 L of inhaled air and that dispersion of the powder formulation is good at a peak flow (PIF) of only 25–40 L/min [Citation150]. Obviously, inhaled volume and PIF depended on the DPI resistance, but the minimal values (0.5 L and 25 L/min) obtained in the study did not vary so much between the different age groups for the highest airflow resistance. More worrying is the observation in this study that a high incidence of narrowing of the passageway for the aerosol (by teeth, tongue, or cheeks) was observed in 90% of the inhalations, depending on the DPI mouthpiece design (). Also, exhalation through the DPI prior to inhalation occurred. This may require special features for a pediatric DPI.
Figure 3. The oral cavity during DPI use of a child. a: Captures from a video recording of the changes in the passageway for the aerosol during a single inhalation manoeuvre; b; examples of a narrowed passageway by elevated tongue and inwards directed displacement of the cheeks (captures from different video recordings of different children showing some extreme situations). The recordings were made using a bronchoscope in the mouthpiece of a test inhaler with exchangeable mouthpieces and air flow resistances [Citation150].
![Figure 3. The oral cavity during DPI use of a child. a: Captures from a video recording of the changes in the passageway for the aerosol during a single inhalation manoeuvre; b; examples of a narrowed passageway by elevated tongue and inwards directed displacement of the cheeks (captures from different video recordings of different children showing some extreme situations). The recordings were made using a bronchoscope in the mouthpiece of a test inhaler with exchangeable mouthpieces and air flow resistances [Citation150].](/cms/asset/a3b07d4f-e9e4-4a1f-8377-6ac4a2d5cab3/iedd_a_1224846_f0003_oc.jpg)
DPIs for systemic drug delivery are not fundamentally different from devices for local therapies in the lungs. Inhalation seems attractive as noninvasive route for the delivery of peptides and proteins that otherwise must be injected, because the respiratory tract offers an enormous absorptive surface area for drug absorption [Citation151,Citation152]. It has long been believed that deposition in the most distal airways is needed for a high and reliable bioavailability. This would require an appropriate particle size distribution (approx. 1–3 μm), deep inhalation of the aerosol at a low flow rate (after previous exhalation to residual volume), and a long breath hold time. And even then, drug deposition on all different epithelial surfaces is inevitable. However, it is now recognized that the preferred site of deposition may depend on the type of drug, e.g. certain antibodies are preferentially absorbed in the upper airways [Citation152].
3.5. Miscellaneous innovative developments
The profusion of developments and the diversity of applications for pulmonary drug delivery (as dry powder) make it impossible to give a complete survey. A fair conclusion is that formulation developments outnumber the device developments. Many trends in powder formulation have already been mentioned in the paragraphs 3.1 and 3.2. Relatively new is the interest in nanoparticulate systems for inhalation [Citation153,Citation154], and an abundance of articles on this subject has been published particularly since 2005. The application of nanosystems has been investigated for drug, protein, and gene delivery through the respiratory tract because of their ability to escape (alveolar) macrophages and mucociliary clearance mechanisms [Citation154]. This prolongs their residence time. They can also enter intercellular compartments to increase bioavailability but exhibit poor lung deposition efficiency and high formulation instability. Another method proposed for evading macrophage uptake in the deep lung is by making liposomal particles, using phospholipids similar to endogenous lung surfactant. Such particles are less foreign to alveolar macrophages [Citation155]. Liposomal formulations are also used for controlled drug release and enhanced formulation stability, but scaling-up the manufacturing process to an industrial scale is often problematic. Highly innovative is the principle of micromolding particles with defined uniform shape, size, and morphology, termed PRINT® (Liquidia) [Citation156]. The technique is compatible with biopharmaceuticals such as proteins and oligonucleotides and yields particles in a size range suitable for inhalation. In the field of DPI development, the need for high-dose delivery devices becomes urgent, particularly for the delivery of antibiotics in cystic fibrosis (CF), TB, and other infectious lung diseases. Noticeable in this respect is that all known device developments are from academia, such as the Twincer™ [Citation157], the Cyclops [Citation158], and the Orbital® multibreath DPI [Citation159,Citation160]. Also, a new high-dose DPI based on fluidized bed design is developed in the academic setting [Citation161]. The industry, in contrast, has chosen for existing capsule inhalers for dry powder tobramycin (Novartis, TOBI®), ciprofloxacin (Bayer Pharma AG), colistin sulfomethate (Forest Laboratories, Colobreathe®), and mannitol (Pharmaxis, Aridol®) using the Turbospin (named Podhaler® for TOBI) and RS01 (for Aridol), a high-resistance version of the ISF inhaler, respectively. All these devices have a prefabricated powder formulation in common. A completely different approach is the Staccato® system of Alexza Pharmaceuticals. The basic concept of this system is rapid and efficient vaporization of a thin film of pure drug coated on a metallic substrate inside a passageway for the inhaled air [Citation162]. Heating to 400°C for evaporation of the drug in approx. 0.2 s is triggered by the inhaled air stream and followed by condensation to produce the inhaled aerosol particles before the drug is inhaled. The system is developed for loxapine. Innovative are also the Technosphere® technology of MannKind and the iSPERSE™ platform of Pulmatrix. The Technosphere® technology makes use of self-assembling (fumaryl diketopiperazine, FDKP) carrier particles with a high surface area for absorption of large active molecules [Citation163]. The carrier particles are in the micron size range and small enough to be inhaled. This has the advantage that drug and carrier need not to be separated before inhalation. The principle has been used to develop Technosphere Insulin (AFREZZA®) which is inhaled with a new DreamBoat™ inhaler [Citation164]. iSPERSE™ powders consist of dense salt-containing particles in an appropriate size range for inhalation. They are highly dispersible without blending with lactose and contain typically less than 20% excipients in formulations with the drug, or drug combination. iSPERSE™ particles enable processing of both large-molecule and low-potency drugs [Citation165].
On the application site, new dry powder bronchial challenge tests have recently been presented for methacholine and adenosine [Citation110,Citation166] in addition to the mannitol test [Citation167].
4. Dry powder inhalation: future
In 1995, it was written: ‘it is possibly the “power assisted” multidose dry powder inhalers which represent the real “new wave” of powder aerosol delivery’ [Citation18]. Today, 20 years later, no active DPI has been successful on the market yet. In the early 1990s, also the first manuscripts about the promising perspectives of aerosolized medication for gene and peptide therapy were published [Citation168]. It was anticipated that ‘in the near future, optimal formulations will be combined with modified aerosol delivery devices to achieve reproducible dosing’. Again 20 years later, it has to be acknowledged that still more efficient aerosol devices are needed, that formulations have to be safer and bioavailability has to be improved [Citation169]. McElroy et al. compared the results for inhaled proteins from different studies, showing that systemic bioavailability for compounds with mol weights (MWs) <10 kDa can vary from almost 0% to 100%, whereas for MWs >10 kDa, 60% is maximal (so far) [Citation170]. Therefore, bioavailabilities are often too low for cost-effective and reliable treatments [Citation152]. Besides, pulmonary administration of one of the most interesting candidates at the time, insulin, may be less relevant today because of the refinement of subcutaneous injection devices and new pharmacological strategies for patients with type 2 diabetes [Citation171]. These examples show how difficult it is to predict the future of DPI therapy. Many large biopharmaceuticals may eventually appear to be unsuitable for inhalation. Smaller molecules, on the other hand, such as levodopa, loxapine, and (locally acting) iloprost or sildenafil (MWs ≪1 kDa) seem to have considerably greater future perspective. Currently, the treatment of infectious lung diseases with inhaled antibiotics is in the spotlight. An advantage is the deposition directly at the site of infection, which makes higher local concentrations, and thus, a more effective therapy possible without increasing the adverse systemic effects. Higher concentrations also could make drug-resistant organisms susceptible to the antibacterial drugs again. Some inhaled dry powder antibiotics are on the market already (e.g. TOBI from Novartis and Colobreathe from Forest Laboratories for CF therapy) or are expected to obtain approval soon (Ciprofloxacin DPI from Bayer HealthCare for bronchiectasis therapy). For all these drugs, classic capsule inhalers are used. Therapy for diseases such as TB is much more challenging because of the higher doses involved. For the future, the success of pulmonary TB treatment may depend on the development of efficient novel high-dose DPIs and synergistic drug combinations to minimize the number of inhalations. Some studies with proven synergistic effect of antibiotic combinations have recently already been reviewed e.g. [Citation118]. Particularly for inhaled antibiotics, the need for good patient compliance with the instructions, minimizing patient errors, and good adherence to the therapy are prerequisites for an effective treatment. Also, the expectations for dry powder pulmonary vaccination are currently high. The advantages are improved stability (in the dry state), no reconstitution needed (requiring sterile water), and noninvasive administration (no needle technology) [Citation148]. However, inhaled vaccination as one-off administration depends on a high reliability regarding delivered fine particle dose on the target area. This requires highly efficient inhaler technology which also needs to be disposable, simple, and cheap to facilitate mass vaccination programs in developing countries [Citation172]. It may also be of utmost importance to develop reusable instruction and feedback systems for such disposable inhalers in order to practice the inhalation maneuver and check correct inhalation during vaccine delivery. Disposable DPIs are furthermore recommended for hygroscopic drug formulations and antibiotics [Citation172]. For a realistic future expectation, it may be wise not to think too much in terms of feasible applications, however, but rather in terms of DPI technique itself and what is possible with this technique. Future developments will make use of new tools. New DPI design and development will benefit from computer-aided design. CFD and discrete element method may assist optimizing the flow field and particle behavior inside, but also ex-mouthpiece of the inhaler into the oral cavity [Citation173–Citation177]. New 3D printing techniques facilitate rapid DPI prototyping and making casts of (upper) parts the respiratory tract based on high-resolution CT scans [Citation178]. Coimaging may enable to draw better conclusions about respiratory tract deposition in individual patients with diseased lungs and eventually facilitate personalized treatment with highly expensive drugs [Citation143–Citation145]. But most important of all may be reducing the cost of inhaled therapy considering the pressure on health budgets and the rapidly growing demand for inhaled therapies in developing and newly industrialized countries. Some of the challenges and objectives for future DPI design and development are listed in .
Table 1. Future challenges and objectives for DPIs.
5. Expert opinion
Many reviews on dry powder inhalation have been published since 1985, but clear visions or strategies for improved DPI design and future DPI development were scarcely presented. Most reviews are confined to a state-of-the-art description with in vitro and in vivo performance data e.g. [Citation179,Citation180] or performance data in combination with functionality and production considerations [Citation181]. Also, new tools and techniques for DPI device and formulation design have been reviewed [Citation182], and a few have provided characteristics of an ideal DPI [Citation183] without presenting a strategy for achieving these characteristics. Many reviews do have the same key design factors in common, however, such as simplicity, cost-effectiveness, safety, and flow rate-independent lung deposition. Also, the needs for reduction of patient errors and device-formulation integrated development are frequently mentioned. How different is daily practice. The many generic devices on the market increase the number of different inhalers used by the same patient, and it is known that this contributes to the number of patient errors [Citation184]. Future improvement of the inhalation therapy is, therefore, primarily served by reducing the number of different inhalers used by the same patient for the same therapy. However, in practice, several plastic manufacturers and consultant agencies continue to develop new DPIs without knowing what type of drug or drug formulation they can or will be used for. This frequently entails a need for unnecessary powder processing or particle engineering to obtain an acceptable performance of the combination. This makes development and production of such inhalers more expensive and, thus, less cost-effective and also less safe. It also often requires the use of excipients of which we assume that they are safe because they are endogenous, or because we cannot imagine (yet) how they can interact with the physiological processes in the lungs. It should be considered that the fact that the excipients are endogenous does not make them safe by definition, however. Their functionality in the human body does not depend on their presence but on their concentration and location. Any deviation from the normal physiological concentration can potentially be harmful. For instance, sodium chloride and cholesterol are endogenous, and an occasional high oral intake may not immediately cause safety problems, but on the long term, they can become a serious health risk. For cholesterol, it took more than 40 years to acknowledge this after a link between cholesterol and vascular damage in rabbits was already discovered in the early 1900s. Obviously, the health risk of exogenous excipients, such as poly-lactic acid (PLA), chitosan, carrageenan, hydroxypropyl methylcellulose, magnesium stearate, etc., in inhalation products is much greater. Parlati et al. recently prepared drug solutions with sodium stearate for spray drying and observed micelle formation when this adjuvant was added in amounts >1% [Citation185]. It can be computed for spray aerosols in the size range needed for inhalation powders that amounts slightly over 1% indeed exceed the critical micelle concentration (CMC) for this salt, and examples such as these show what might happen locally in the lungs for magnesium stearate (and other exogenous surfactants) too when they exceed their CMC. The risks of such nanoparticulate micelles are still widely unknown. Only for achieving special effects, such as product stabilization, improved absorption, sustained action, or macrophage targeting, the use of excipients should be considered, but their selection and application have to be weighed carefully against the achieved therapeutic improvement from their use. A logical consequence of refraining from the use of excipients is that the inhaler design must be adapted to the physicochemical properties of the drug (class) [Citation118]. Only device-formulation integrated development can prevent unnecessary use of excipients and powder formulation and knowing what the inhaler will be used for is a prerequisite for a good device design. Practically, device-formulation integrated development for the different drugs needed by the same patient may be a difficult problem to solve because different manufacturers, having their own strategies and inhalers, may produce the drugs. They most likely use different inhalers for that. Generic companies could bring the solution by making it their strategy to develop the complete range of inhaled medicines for a particular disease in the same inhaler.
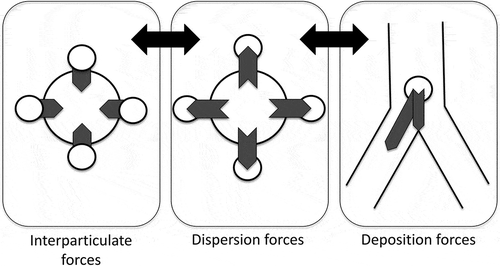
Innovation starts with a better understanding of how lung deposition can be influenced and controlled and which inhaler characteristics are most critical in that respect. One of the most persistent misconceptions in inhalation therapy with DPIs is the belief that a constant lung deposition depends on a flow rate-independent fine particle dose [Citation75,Citation76,Citation185]. As mentioned earlier, a flow rate-independent lung deposition is served best with a higher fine particle dose at a higher flow rate to compensate for a higher oropharyngeal deposition. Additionally, a low flow rate limits the oropharyngeal deposition, and this requires a medium-high to high inhaler resistance. Innovation also requires breaking with the tradition of making complex formulations for poorly designed DPIs. More effective inhalers are needed that are less dependent of the interparticulate forces in the drug formulation and generate high fine particle doses at low flow rates without needing excipient containing particle engineered powders. Achieving a proper balance between the interparticulate forces in the mixture, the dispersion forces generated by the inhaler and the (inertial) deposition forces during inhalation can be the solution () [Citation118]. Inhalers such as the Novolizer and Genuair® with air classifier technology as dispersion principle clearly show that this is very well possible (). They generate high inertial dispersion forces at a medium resistance (limited flow rate), which makes these inhalers largely independent of the interparticulate forces in the formulation. Such effective inhalers also deliver the desired increasing fine particle dose with increasing flow rate.
Figure 4. The desired balance between the interparticulate force in the powder formulation, the dispersion generated by the inhaler and the lung deposition forces during inhalation. Appropriate balancing of these forces is needed for optimal DPI performance.
For future DPI developments also the use of hard gelatin or hydroxypropyl methylcellulose capsules should be reconsidered. Capsules require a threshold value for the flow rate to achieve adequate emptying by rotation, spinning, or vibration during inhalation. The high risk of fractioning during piercing requires that the inhaler is designed with an appropriate screen in between the capsule chamber and the mouthpiece to prevent that large capsule fragments are inhaled. Capsules also provide poor moisture protection and need appropriate blister packaging to guarantee long-term stability of moisture-sensitive drugs and drug formulations. Moreover, the possibility must be excluded that capsules for inhalation are mistaken for oral capsules or that oral capsules are inhaled. For future applications (e.g. hygroscopic formulations), it also has to be considered whether disposable or reusable inhalers are the best option [Citation172]. Finally, new inhaler technology should enable training of the proper inhalation technique by providing feedback about the inhalation maneuver during drug administration to the patient or the health-care professional during inhalation [Citation186]. By showing to the patient when the correct flow rate is achieved and sufficient volume has been inhaled, the maximal fine particle dose from a DPI and optimal deposition pattern of that fine particle dose may be obtained. Recording of these data enables the health-care professional to check compliance with the instructions for inhaler use and adherence to the therapy.
Article highlights
Early dry powder inhalers have a poor drug delivery performance due to design weaknesses
Formulation studies currently outnumber device design and development studies and are meant to make old inhaler concepts perform better
Large amounts of excipients in high dose formulations are unwanted: they increase the number of inhalations of a single dose, whereas their safety in the long term is uncertain
New applications, particularly pulmonary vaccination, require more effective inhalers
Future inhalers have to be simple (but effective) and give good feedback to the patient to increase adherence to the therapy
Several applications (e.g. delivery of hygroscopic drug formulations and vaccination) are served better with disposable inhalers
This box summarizes key points contained in the article.
Declaration of interest
The contents of this article is the authors’ responsibility and neither COST nor any person acting on its behalf is responsible for the use which might be made of the information contained in it. AH de Boer, P Hagedoorn and HW Frijlink receive royalties from the Novolizer and Genuair sales and are involved in the development and production of the Twincer platform by pureIMS. F Grasmeijer and M Hoppentocht are part-time employees of pureIMS and F Buttini receives research grants from Chiesi Pharmaceutici SpA, Qualicaps® Europe S.A.U. and Plastiape SpA for inhalation projects. The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Additional information
Funding
References
- Anderson PJ. History of aerosol therapy: liquid nebulization to MDIs to DPIs. Respir Care. 2005;50:1139–1149.
- Dessanges J. A history of nebulization. J Aerosol Med. 2001;14:65–71.
- Grossman J. The evolution of inhaler technology. J Asthma. 1994;31:55–64.
- Sanders M. Inhalation therapy: an historical review. Primary Care Respir J. 2007;16(2):71–81.
- Cutchis P. Stratospheric ozone depletion and solar ultraviolet radiation on earth. Science. 1974;184:13–19.
- Lovelock JE. Halogenated hydrocarbons in the atmosphere. Ecotoxicol Environ Saf. 1977;1:399–346.
- Noakes TJ. CFCs, their replacement, and the ozone layer. J Aerosol Med. 1995;8:S3–S7.
- D’Souza S. The montreal protocol and essential use exemptions. J Aerosol Med. 1995;8:S13–S17.
- The London Gazette. 1864 Aug vol. 16. 4044.
- Robson RA, Taylor BJ, Taylor B. Sodium cromoglycate: Spincaps or metered dose aerosol. Br J Clin Pharm. 1981;11:383–384.
- Bell JH, Hartley PS, Cox JS. Dry powder aerosols. I: a new inhaler device. J Pharm Sci. 1971;60:1559–1564.
- Hall VC. Inhaler. US patent 2517482. 1950.
- Ellis MC. Device for dispensing medicaments. US patent 2573918. 1951.
- Cocozza S. Inhaling device for medial powder compositions. US patent 3807400. 1974.
- Mathes S. Inhalation device. US patent 3938516. 1976.
- Wilke GA, Henderson NC, Stansel SM, et al. Inhalation device. US patent 3948264. 1976.
- Valentini L, Maiorano N. Inhalation device. US patent 4069819. 1978.
- Clark AR. Medical aerosol inhalers: past, present and future. Aerosol Sci Technol. 1995;22:374–391.
- Pover GM, Dash CH. A new modified form of inhaler (“Rotahaler”) for patients with chronic obstructive lung disease. Pharmatherapeutica. 1985;4:98–101.
- Kladders H. Powdered pharmaceutical inhaler. US patent 4889114. 1989.
- Cocozza S. Inhaler for powdered medicaments. US patent 3991761. 1976.
- Jones TM, Pilpel N. Some physical properties of lactose and magnesia. J Pharm Pharmacol. 1965;17:440–448.
- Shotton E, Orr NA. Studies on mixing cohesive powders. J Pharm Pharmacol Suppl. 1971;23(S1):260S.
- Travers DN, White RC. The mixing of micronised sodium bicarbonate with sucrose crystals. J Pharm Pharmacol Suppl. 1971;23(S1):260S–261S.
- Hersey JA. Ordered mixing: a new concept in powder mixing. Powder Technol. 1975;7:41–44.
- Staniforth JN. Order out of chaos. J Pharm Pharmacol. 1987;39:329–334.
- de Boer AH, Hagedoorn P, Gjaltema D, et al. Air classifier technology (ACT) in dry powder inhalation Part 3: Design and development of an air classifier family for the Novolizer multi-dose dry powder inhaler. Int J Pharm. 2006;310:72–80.
- de Boer AH, Hagedoorn P, Gjaltema D, et al. Air classifier technology (ACT) in dry powder inhalation Part 4: Performance of air classifier technology in the Novolizer multi-dose dry powder inhaler. Int J Pharm. 2006;310:81–89.
- Newman SP, Pitcarin GR, Hirst PH, et al. Scintigraphic comparison of budesonide deposition from two dry powder inhalers. Eur Respir J. 2000;16:178–183.
- Nieminen MM, Vidgren M, Laurikainen K, et al. Easyhaler, a novel multiple dose powder inhaler: clinically equivalent to salbutamol metered dose inhaler and easier to use. Respiration. 1994;61:37–41.
- Meakin BJ, Ganderton D, Panza I, et al. The effect of flow rate on drug delivery from the Pulvinal, a high-resistance dry powder inhaler. J Aerosol Med. 1998;11:143–152.
- Overhoff KA, Clayborough R, Crowley M. Review of the TAIFUN multidose dry powder technology. Drug Dev Ind Pharm. 2008;34:960–965.
- Lankinen T. Device intended for measuring a dose of powdered medicament for inhalation. US patent RE35552. 1997.
- Ambrosio TJ, Ashley CR, Bilanin AJ, et al. Inhaler for powdered medications. WO patent 94/14492. 1994.
- Newhouse MT, Nantel NP, Chambers CB, et al. Clickhaler (a novel dry powder inhaler) provides similar bronchodilation to pressurized metered-dose inhaler, even at low flow rates. Chest. 1999;115:952–956.
- Dahl R, Creemers JP, Van Noord J, et al. Comparable efficacy and tolerability of formoterol (Foradil) administered via a novel multi-dose dry powder inhaler (Certihaler) or the aerolizer dry powder inhaler in patients with persistent asthma. Respiration. 2004;71:126–133.
- Middle MV, Terblanché J, Perrin VL, et al. Bronchodilating effects of salbutamol from a novel inhaler Airmax. Resp Med. 2002;96:493–498.
- Canonica GW, Arp J, Keegstra JR, et al. Spiromax, a new dry powder inhaler: dose consistency under simulated real-world conditions. J Aerosol Med Pulm Drug Deliv. 2015;28:309–319.
- Sumby BS, Churcher KM, Smith IJ, et al. Dose reliability of the serevent diskhaler system. PharmTech Int. 1993;5:20–27.
- Brindley A, Sumby BS, Smith IJ, et al. Design, manufacture and dose consistency of the serevent diskus inhaler. Pharm Technol Eur. 1995;7:14–22.
- Wetterlin K. Turbuhaler: a new dry powder inhaler for administration of drugs to the airways. Pharm Res. 1988;5:506–508.
- Crompton GK. Dry powder inhalers: advantages and limitations. J Aerosol Med. 1991;4:151–156.
- LiCalsi C, Christensen T, Bennett JV, et al. Dry powder inhalation as a potential delivery method for vaccines. Vaccine. 1999;17:1796–1803.
- White S, Bennett DB, Chey S, et al. EXUBERA: pharmaceutical development of a novel product for pulmonary delivery of insulin. Diabetes Technol Ther. 2005;7:896–906.
- Han R, Papadopoulos G, Greenspan BJ. Flow field management inside the mouthpiece of the spiros inhaler using particle image velocimetry. Aerosol Sci and Technol. 2002;36:329–341.
- Virchov JC. What plays a role in the choice of inhaler device for asthma therapy. Curr Med Res Opin. 2005;21:S19–S25.
- Dolovich MB, Dhand R. Aerosol drug delivery: developments in device design and clinical use. Lancet. 2011;377:1032–1045.
- Laube BL, Janssens HM, De Jongh FHC, et al. What the pulmonary specialist should know about the new inhalation therapies. Eur Resp J. 2011;37:1308–1331.
- Usmani OS, Biddiscombe MF, Barnes PJ. Regional lung deposition and bronchodilator response as a function of beta2-agonist particle size. Am J Respir Crit Care Med. 2005;172:1497–1504.
- Demoly P, Hagedoorn P, de Boer AH, et al. The clinical relevance of dry powder inhaler performance for drug delivery. Resp Med. 2014;108:1195–1203.
- Norman P. Developments in inhaled combination therapies: patent activity 2013-2014. Expert Opin Ther Patents. 2015;25:1239–1245.
- Flenly DC. Today’s treatment of airway obstruction.and tomorrow’s? Respiration. 1989;55(Suppl.2):4–9.
- Prime D, Atkins PJ, Slater A, et al. Review of dry powder inhalers. Adv Drug Deliv Rev. 1997;26:51–58.
- Smith IJ, Parry-Billings M. The inhalers of the future? A review of dry powder devices on the market. Pulm Pharmacol Ther. 2003;16:79–95.
- de Boer AH, Gjaltema D, Hagedoorn P. Inhalation characteristics and their effects on in vitro drug delivery from dry powder inhalers. Part 2: effect of peak flow rate (PIFR) and inspiration time on the in vitro drug release from three different types of commercial dry powder inhalers. Int J Pharm. 1996;138:45–46.
- Pauwels R, Newman S, Borgström L. Airway deposition and airway effects of antiasthma drugs delivered from metered dose inhalers. Eur Respir J. 1997;10:2127–2138.
- Olsson B. Aerosol particle generation from dry powder inhalers: can they equal pressurized metered dose inhalers? J Aerosol Med. 1995;8:S13–S18.
- Borgström L, Derom E, Ståhl E, et al. The inhalation device influences lung deposition and bronchodilating effect of terbutaline. Am J Respir Crit Care Med. 1996;153:1636–1640.
- Frijlink HW, de Boer AH. Dry powder inhalers for pulmonary drug delivery. Exp Opin Drug Deliv. 2004;1:67–86.
- Clark AR, Newman SP, Dasovich N. Mouth and oropharyngeal deposition of pharmaceutical aerosols. J Aerosol Med. 1998;11:S116–S121.
- DeHaan WH, Finlay WH. Predicting extrathoracic deposition from dry powder inhalers. Aerosol Sci. 2004;35:309–331.
- de Boer AH, Chan HK, Price R. A critical view on lactose-based drug formulation and device studies for powder inhalation; Which are relevant and what interactions to expect. Adv Drug Deliv Rev. 2012;64:257–274.
- Grasmeijer F, Grasmeijer N, Hagedoorn P, et al. Recent advances in the fundamental understanding of adhesive mixtures for inhalation. Curr Phar Design. 2015;21:5900–5914.
- Islam N, Stewart P, Larson I, et al. Effect of carrier size on the dispersion of salmeterol xinafoate from interactive mixtures. J Pharm Sci. 2004;93:1030–1038.
- Guenette E, Barrett A, Kraus D, et al. Understanding the effect of lactose particle size on the properties of DPI formulations using experimental design. Int J Pharm. 2009;380:80–88.
- Kawashima Y, Serigano T, Hino T, et al. Effect of surface morphology of carrier lactose on dry powder inhalation of pranlukast hydrate. Int J Pharm. 1998;172:179–188.
- Ferrari F, Cocconi D, Bettini R, et al. The surface roughness of lactose particles can be modulated by wet-smoothing using a high-shear mixer. AAPS PharmSciTech. 2004;5:69–74.
- Kayali W, Ticehurst MD, Murphy J, et al. Improved aerosolization performance of salbutamol sulfate formulated with lactose crystallized from binary mixtures of ethanol-acetone. J Pharm Sci. 2011;100:2665–2684.
- Arnold K, Grass P, Knecht A, et al. Powders for inhalation. US patent 5478578. 1995.
- Jones MD, Price R. The influence of fine excipient particles on the performance of carrier-based dry powder inhalation formulations. Pharm Res. 2006;23:1665–1674.
- Begat P, Morton DAV, Staniforth JN, et al. The cohesive-adhesive balances in dry powder inhaler formulations I: direct quantification by atomic force microscopy. Pharm Res. 2004;21:1591–1597.
- Tong HY, Shekunov BY, York P, et al. Characterization of two polymorphs of salmeterol xinafoate crystallized from supercritical fluids. Pharm Res. 2001;18:852–858.
- Sitz R. Current innovations in dry powder inhalers. Ondrugdelivery. 2010:10–12. Available from: www.ondrugdelivery.com
- Newman S, Malik S, Hirst P, et al. Lung deposition of salbutamol in healthy human subjects from the MAGhaler dry powder inhaler. Respir Med. 2002;96:1026–1032.
- de Boer AH, Gjaltema D, Hagedoorn P, et al. Comparative in vitro performance evaluation of the novopulmon 200 novolizer and budesonid-ratiopharm jethaler: two novel budesonide dry powder inhalers. Pharmazie. 2004;59:692–699.
- Alhalaweh A, Kaialy W, Buckton G, et al. Theofylline cocrystals prepared by spray drying: physicochemical properties and aerosolization performance. AAPS PharmSciTech. 2013;14:265–276.
- Padrela L, Rodrigues MA, Tiago J, et al. Tuning physiochemical properties of theofylline by cocrystallization using the super critical fluid enhanced atomization technique. J Supercritical Fluids. 2014;86:129–136.
- Velaga SP, Berger R, Carlfors J. Supercritical fluids crystallization of budesonide and flunisolide. Pharm Res. 2002;19:1564–1571.
- Rehman M, Shekunov BY, York P, et al. Optimisation of powders for pulmonary delivery using supercritical fluid technology. Eur J Pharm Sci. 2004;22:1–17.
- Buckton G. Characterisation of small changes in the physical properties of powders of significance for dry powder inhalation formulations. Adv Drug Deliv Rev. 1997;26:17–27.
- Staniforth JN. Carrier particles for use in dry powder inhalers. WO patent 1996023485. 1996.
- Begat P, Morton DA, Shur J, et al. The role of force control agents in high-dose dry powder inhaler formulations. J Pharm Sci. 2009;98:2770–2783.
- Das SC, Zhou Q, Morton DA, et al. Use of surface energy distributions by inverse gas chromatography to understand mechanofusion processing and functionality of lactose coated with magnesium stearate. Eur J Pharm Sci. 2011;43:325–333.
- Grant AC, Walker R, Hamilton M, et al. The `ELLIPTA dry powder inhaler: design, functionality, in vitro dosing performance and critical task compliance by patients and caregivers. J Aerosol Med Pulm Drug Deliv. 2015;28:474–485.
- Pavkov R, Mueller S, Fiebich K, et al. Characteristics of a capsule based dry powder inhaler for the delivery of indacaterol. Curr Med Res Opin. 2010;26:2527–2533.
- Staniforth JN, Morton DA, Brambilla G, et al. Pharmaceutical formulations foer dry powder inhalers in the form of hard-pellets. WO 01/78693. 2001.
- Cocconi D, Alberti MD, Busca A, et al. Use of magnesium stearate in dry powder formulations for inhalation. US patent 20120082727. 2012.
- Corradi M, Chrystyn H, Cosio BG, et al. NEXThaler, an innovative dry powder inhaler delivering an extrafine fixed combination of beclomethasone and formoterol to treat large and small airways in asthma. Exp Opin Drug Deliv. 2014;11:1497–1506.
- Buttini F, Brambilla G, Copelli D, et al. Effect of flow rate on in vitro aerodynamic performance of NEXThaler in comparison with diskus and turbuhaler dry powder inhalers. J Aerosol Med Pulm Drug Deliv. 2015;28:1–12.
- Glover W, Chan H-K, Eberl S, et al. Effect of particle size of dry powder mannitol on the lung deposition in healthy volunteers. Int J Pharm. 2008;349:314–322.
- Bosquillon C, Lombry C, Préat V, et al. Influence of formulation excipients and physical characteristics of inhalation dry powders on their aerosolization performance. J Control Release. 2001;7:329–339.
- Li H-Y, Seville PC, Williamson IJ, et al. The use of amino acids to enhance the aerosolisation of spray-dried powders for pulmonary gene therapy. J Gene Med. 2005;7:343–353.
- Pilcer G, Vanderbist F, Amighi K. Spray-dried carrier free dry powder tobramycin formulations with improved dispersion properties. J Pharm Sci. 2009;98:1463–1475.
- Duddu SP, Sisk SA, Walter YH, et al. Improved lung delivery from a passive dry powder inhaler using an engineered pulmosphere powder. Pharm Res. 2002;19:689–696.
- Newhouse MT, Hirst PH, Duddu SP, et al. Inhalation of a dry powder tobramycin pulmosphere formulation in healthy volunteers. Chest. 2003;124:360–366.
- Vehring R. Pharmaceutical particle engineering by spray drying. Pharm Res. 2008;25:999–1022.
- Schüle S, Schulz-Fademrecht T, Garidel P, et al. Stabilization of IgG1 in spray-dried powders for inhalation. Eur J Pharm Biopharm. 2008;69:793–807.
- Saluja V, Amorij JP, Kapteyn JC, et al. A comparison between spray drying and spray freeze drying to produce influenza subunit vaccine powder for inhalation. J Control Release. 2010;144:127–133.
- Shoyele SA, Sivadas N, Cryan S-A. The effects of excipients and particle engineering on the biophysical stability and aerosol performance of parathyroid hormone (1-34) prepared as dry powder for inhalation. AAPS PharmSciTech. 2011;12:304–311.
- Adouy SA, van der Schaaf G, Hinrichs WL, et al. Development of a dried influenza whole inactivated virus vaccine for pulmonary immunization. Vaccine. 2011;29:4345–4352.
- Bürki K, Jeon I, Arpagaus C, et al. New insights into inspirable protein powder preparation using a nano spray dryer. Int J Pharm. 2011;408:248–256.
- Fiegel J, Fu J, Hanes J. Poly(ether-anhydride) dry powder aerosols for sustained drug delivery in the lungs. J Control Release. 2004;96:411–423.
- Chougule M, Padhi B, Misra A. Development of spray dried liposomal dry powder inhaler of dapsone. AAPS PharmSciTech. 2008;9:47–53.
- Bi R, Shao W, Wang Q, et al. Solid lipid nanoparticles as insulin inhalation carriers for enhanced pulmonary delivery. J Biomed Nanotechnol. 2009;5:84–92.
- Scalia S, Salama R, Young P, et al. Preparation and in vitro evaluation of salbutamol-loaded lipid microparticles for sustained release pulmonary therapy. J Microencapsul. 2012;29:225–233.
- d’Angelo I, Casciaro B, Miro A, et al. Overcoming barriers in Pseudomonas lung infections: engineered nanoparticles for local delivery of a cationic antimicrobial peptide. Colloids Surf B Biointerfaces. 2015;135:717–725.
- Manca ML, Valenti D, Sales OD, et al. Fabrication of polyelectrolyte multilayered vesicles as inhalable dry powder for lung administration of rifampicin. Int J Pharm. 2014;472:102–109.
- Amidi M, Pellikaan HC, de Boer AH, et al. Preparation and physicochemical characterization of supercritically dried insulin-loaded microparticles for pulmonary drug delivery. Eur J Pharm Biopharm. 2008;68:191–200.
- Pilcer G, Sebti T, Amighi K. Formulation and characterization of lipid-coated tobramycin particles for dry powder inhalation. Pharm Res. 2006;23:931–940.
- Colombo G, Terzano C, Colombo P, et al. Methacholine dry powder inhaler as a new tool for bronchial challenge test. Int J Pharm. 2008;352:165–171.
- Edwards DA, Hanes J, Caponetti G, et al. Large porous particles for pulmonary delivery. Science. 1997;276:1868–1871.
- Sharma R, Saxena D, Dwivedi D, et al. Inhalable microparticles containing drug combinations to target alveolar macrophages for treatment of pulmonary tuberculosis. Pham Res. 2001;18:1405–1410.
- Gupta A, Pant G, Mitra K, et al. Inhalable particles containing rapamycin for induction of autophagy in macrophages infected with Mycobacterium tuberculosis. Mol Pharm. 2014;11:1201–1207.
- Patel B, Gupta N, Ahsan F. Particle engineering to enhance or lessen particle uptake by alveolar macrophages and to influence the therapeutic outcome. Eur J Pharm Biopharm. 2015;89:163–174.
- Tian G, Longest WP, Li X, et al. Targeting aerosol deposition to and within the lung airways using excipient enhanced growth. J Aerosol Med Pulm Drug Deliv. 2013;26:248–265.
- Son Y-T, Longest WP, Hindle M. Aerosolization characteristics of dry powder inhaler formulations for the excipient enhanced growth (EEG). Application: effect of spray drying process conditions on aerosol performance. Int J Pharm. 2013;443:137–145.
- Pilcer G, Amighi K. Formulation strategy and use of excipients in pulmonary drug delivery. Int J Pharm. 2010;392:1–19.
- Hoppentocht M, Hagedoorn P, Frijlink HW, et al. Developments and strategies for inhaled antibiotic drugs in tuberculosis therapy: a critical evaluation. Eur J Pharm Biopharm. 2013;86:23–30.
- Hoppentocht M, Hagedoorn P, Frijlink HW, et al. Technological and practical challenges of dry powder inhalers and formulations. Adv Drug Deliv Rev. 2014;75:18–31.
- Claus S, Schoenbrodt T, Weiler C, et al. Novel dry powder inhalation system based on dispersion of lyophilisates. Eur J Pharm Sci. 2011;43:32–40.
- Otake H, Okuda T, Okamoto H. Development of spray-freeze-dried powders for inhalation with high inhalation performance and antihygroscopic property. Chem Pharm Bull. 2016;64:239–245.
- Geller DE, Weers J, Heuerding S. Development of an inhaled dry-powder formulation of tobramycin using PulmoSphere technology. J Aerosol Med Pulm Drug Deliv. 2011;24:175–182.
- Belotti S, Rossi A, Colombo P, et al. Spray-dried amikacin sulphate powder for inhalation in cystic fibrosis patients: a quality by design approach for product construction. Int J Pharm. 2014;471:507–515.
- Borgström L, Olsson B, Thorsson L. Degree of throat deposition can explain the variability in lung deposition of inhaled drugs. J Aerosol Med. 2006;19:473–483.
- Stapleton KW, Guentsch F, Hoskinson MK, et al. On the suitability of k-turbulence modelling for aerosol deposition in the mouth and throat: a comparison with experiment. J Aerosol Sci. 2000;31:739–749.
- Zhou Y, Sun J, Cheng Y-S. Comparison of deposition in the USP and physical mouth-throat models with solid and liquid particles. J Aerosol Med Pulm Drug Deliv. 2011;24:277–284.
- Janssens HM, De Jongste JC, Fokkens WJ, et al. The sophia anatomical infant nose-throat (SAINT) model: A valuable tool to study aerosol deposition in infants. J Aerosol Med. 2001;14:433–441.
- DeHaan WH, Finlay WH. In vitro monodisperse aerosol deposition in mouth and throat with six different inhalation devices. J Aerosol Med. 2001;14:361–367.
- Lindert S, Below A, Breitkreutz J. Performance of dry powder inhalers with single dosed capsules in preschool children and adults with improved upper airway models. Pharmaceutics. 2014;6:36–51.
- Gerrity TR, Lee PS, Hass FJ, et al. Calculated deposition of inhaled particles in the airway generations of normal subjects. J Appl Physiol. 1979;47:867–873.
- Weibel ER, Gomez DM. Architecture of the human lung. Use of quantitative methods establishes fundamental relations between size and number of lung structures. Science. 1962;137:577–585.
- Newman S, Steed K, Hooper G, et al. Comparison of gamma scintigraphy and pharmacokinetic technique for asessing pulmonary deposition of terbutaline sulphate delivered by pressurized metered dose inhaler. Pharm Res. 1995;12:231–236.
- Geller DE, Konstan MW, Smith J, et al. Novel tobramycin inhalation powder in cystic fibrosis subjects: pharmacokinetics and safety. Pediatric Pulmonol. 2007;42:307–313.
- Hoppentocht M, Akkerman OW, Hagedoorn P, et al. Tolerability and pharmacokinetic evaluation of inhaled dry powder tobramycin free base in non-cystic fibrosis bronchiectasis patients. PloS ONE. 2016;11:e0149768. doi:10.1371/journal.pone.0149768.
- Newman SP, Pitcairn GR, Hirst PH, et al. History of gamma scintigraphy. J Aerosol Med. 2001;14:139–145.
- Snell NJ, Ganderton D. Assessing lung deposition of inhaled medications. Respir Med. 1999;93:123–133.
- Conway J. Lung imaging - Two dimensional gamma scintigraphy, SPECT, CT and PET. Adv Drug Deliv Rev. 2012;64:357–368.
- Newman S, Fleming J. Challenges in assessing regional distribution of inhaled drug in the human lungs. Expert Opin Drug Deliv. 2011;8:841–855.
- Scheuch G, Bennett W, Borgström L, et al. Deposition, imaging and clearance: what remains to be done? J Aerosol Med Pulm Drug Deliv. 2010;23:S39–S57.
- Fleming J, Bailey DL, Chan HK, et al. Standardization of techniques for using single-photon emission computed tomography (SPECT) for aerosol deposition assessment of orally inhaled products. J Aerosol Med Pulm Drug Deliv. 2012;25:S29–S51.
- Corcoran TE, Davadason SG, Kuell PJ. Introduction: aerosol delivery of orally inhaled agents. J Aerosol Med Pulm Drug Deliv. 2012;25:S3–S5.
- Laube BL, Corcoran TE, Davadason SG, et al. Editorial: standards for lung imaging techniques. J Aerosol Med Pulm Drug Deliv. 2012;25:S1–S2.
- Conway J, Fleming J, Bennett M, et al. The co-imaging of gamma camera measurements of aerosol deposition and respiratory anatomy. J Aerosol Med Pulm Drug Deliv. 2013;26:123–130.
- De Backer JW, Vos WG, Vinchurkar SC, et al. Validation of computational fluid dynamics in CT-based airway models with SPECT/CT. Radiology. 2010;257:854–862.
- Van Holsbeke CS, Leemans G, Vos WG, et al. Functional respiratory imaging as a tool to personalize respiratory treatment in patients with unilateral diaphragmic paralysis. Resp Care. 2014;59:e127–e131.
- Cohn ML, Davis CL, Middlebrook G. Airborne immunization against tuberculosis. Science. 1967;158:1282–1283.
- Tonnis WF, Kersten GF, Frijlink HW, et al. Pulmonary vaccine delivery: a realistic approach? J Aerosol Med Pulm Drug Deliv. 2012;25:1–12.
- Tonnis WT, Lexmond AJ, Frijlink HW, et al. Devices and formulations for pulmonary vaccination. Expert Opin Drug Deliv. 2013;10:1383–1397.
- Pedersen S. Inhaler use in children with asthma. Dan Med Bull. 1987;34:234–249.
- Lexmond AJ, Kruizinga TJ, Hagedoorn P, et al. Effect of inhaler design variables on pediatric use of dry powder inhalers. PLoS ONE. 2014;19(6). doi:10.1371/journal.pone.0099304.
- Patton JS, Fishburn CS, Weers JG. The lungs as a portal of entry for systemic delivery. Proc Am Thorac Soc. 2004;1:338–344.
- Patton JS, Brain JD, Davies LA, et al. The particle has landed – characterizing the fate of inhaled pharmaceuticals. J Aerosl Med Pulm Drug Deliv. 2010;23:S71–S87.
- Rogueda PG, Traini D. The nanoscale in pulmonary delivery. Part I: deposition, fate, toxicology and effects. Exp Opin Drug Deliv. 2007;4:595–606.
- Ali ME, Lamprecht A. Spray freeze drying for dry powder inhalation of nanoparticles. Eur J Pharm Biopharm. 2014;87:510–517.
- Willis L, Hayes D Jr, Mansour HM. Therapeutic liposomal dry powder inhalation aerosols for targeted lung delivery. Lung. 2012;190:251–262.
- Mack P, Horvath K, Garcia A, et al. Particle Engineering for Inhalation Formulation and Delivery of Biotherapeutics. Inhalation. 2012 Aug. Available from: www/inhalationmag.com
- de Boer AH, Hagedoorn P, Westerman WM, et al. Design and in vitro performance testing of multiple air classifier technology in a new disposable inhaler concept (Twincer) for high powder doses. Eur J Pharm Sci. 2006;28:171–178.
- Hoppentocht M, Akkerman OW, Hagedoorn P, et al. The Cyclops for pulmonary delivery of aminoglycosides: a new member of the twincer family. Eur J Pharm Biopharm. 2015;90:8–15.
- Young PM, Salama RO, Zhu B, et al. Multi-breath dry powder inhaler for delivery of cohesive powders in the treatment of bronchiectasis. Drug Develop Ind Pharm. 2015;41:859–865.
- Young PM, Crapper J, Phillips G, et al. Overcoming dose limitations using the Orbital multi-breath dry powder inhaler. J Aerosol Med Pulm Drud Deliv. 2014;27:138–147.
- Farkas DR, Hindle M, Longest PW. Characterization of a new high-dose dry powder inhaler (DPI) based on fluid bed design. Ann Biomed Eng. 2015;43:2804–2815.
- Dinh K, Myers DJ, Glazer M, et al. In vitro aerosol characterization of staccato loxapine. Int J Pharm. 2011;403:101–108.
- Richardson PC, Boss AH. Technosphere insulin technology. Diabetes Technol Ther. 2007;9:S65–S72.
- Kling J. Dreamboat sinks prospects for fast approval of inhaled insulin. Nat Biotechnol. 2011;29:176–176.
- Lipp MM, Sung JC. Monovalent metal cation dry powders for inhalation. US0231066A1. 2015.
- Lexmond AJ, Hagedoorn P, van der Wiel E, et al. Adenosine dry powder inhalation for bronchial challenge testing, part 1: Inhaler and formulation development and in vitro performance testing. Eur J Pharm Biopharm. 2014;86:105–114.
- Anderson SD, Brannan J, Spring J, et al. A new method for bronchial-provocation testing in asthmatic subjects using a dry powder of mannitol. Am J Respir Crit Care Med. 1997;156:758–765.
- Byron PR, Patton JS. Drug delivery via the respiratory tract. J Aerosol Med. 1994;7:49–75.
- Laube BL. Aerosolized medications for gene and peptide therapy. Resp Care. 2015;60:806–821.
- McElroy MC, Kirton C, Gliddon D, et al. Inhaled pharmaceutical drug development: nonclinical considerations and case studies. Inhal Toxicol. 2013;25:219–232.
- Strack T. The pharmacokinetics of alternative insulin delivery systems. Curr Opin Investig Drugs. 2010;11:394–401.
- de Boer AH, Hagedoorn P. The role of disposable inhalers in pulmonary drug delivery. Exp Opin Drug Deliv. 2015;12:143–157.
- Suwandecha T, Wongpoowarak K, Schrichana T. Computer-aided design of dry powder inhalers using computational fluid dynamics to assess performance. Pharm Dev Technol. 2016;21:54–60.
- de Boer AH, Hagedoorn P, Woolhouse R, et al. Computational fluid dynamics (CFD) assisted performance evaluation of the Twincer disposable high-dose dry powder inhaler. J Pharm Pharmacol. 2012;64:1316–1325.
- Ruzycki CA, Javaheri E, Finlay WH. The use of computational fluid dynamics in inhaler design. Exp Opin Drug Deliv. 2013;10:307–323.
- Behara SRB, Longest PW, Farkas DR, et al. Development and comparison of new high efficiency dry powder inhalers for carrier-free formulations. J Pharm Sci. 2014;103:465–477.
- Wong W, Fletcher DF, Traini D, et al. The use of computational approaches in inhaler development. Adv Drug Deliv Rev. 2012;64:312–322.
- Minocchieri S, Burren JM, Bachmann MA, et al. Development of the premature infant nose and throat-model (PrINT-Model): an upper airway replica of a premature neonate for the study of aerosol delivery. Pediatr Res. 2008;64:141–146.
- Hannemann LA. What is new in asthma: new dry powder inhalers. J Pediatr Health Care. 1999;13:159–165.
- Smith IJ, Parry-Billings M. The inhalers of the future? A review of dry powder inhalers on the market today. Pulm Pharmacol Ther. 2003;16:79–95.
- Prime D, Atkins PJ, Slater A, et al. Review of dry powder inhalers. Adv Drug Deliv Rev. 1997;26:51–58.
- Chan HK. Dry powder aerosol delivery systems: current and future research directions. J Aerosol Med. 2006;19:21–27.
- Newman SP, Busse WW. Evolution of dry powder inhaler design, formulation and performance. Resp Med. 2002;96:292–304.
- Rootmensen GN, Van Keimpema HM, Jansen HM, et al. Predictors for incorrect inhalation technique: a study using a validated videotape scoring method. J Aerosol Med Pulm Drug Deliv. 2010;23:232–328.
- Parlati C, Colombo P, Buttini F, et al. Pulmonary spray dried powders of tobramycin containing sodium stearate to improve aerosolization efficiency. Pharm Res. 2009;26:1084–1092.
- Fenton C, Keating GM, Ploster GL. Novolizer, a multidose dry powder inhaler. Drugs. 2003;63:2437–2445.
