ABSTRACT
Introduction
Nontuberculous mycobacteria (NTM) are a diverse group of mycobacterial species that are ubiquitous in the environment. They are opportunistic pathogens that can cause a range of diseases, especially in individuals with underlying structural lung disease or compromised immune systems.
Areas covered
This paper provides an in-depth analysis of NTM infections, including microbiology, environmental sources and transmission pathways, risk factors for disease, epidemiology, clinical manifestations and diagnostic approaches, guideline-based treatment recommendations, drugs under development, and management challenges.
Expert opinion
Future approaches to the management of NTM pulmonary disease will require therapies that are well tolerated, can be taken for a shorter time period and perhaps less frequently, have few drug–drug interactions, and are active against the various strains of pathogens. As the numbers of infections increase, such therapies will be welcomed by clinicians and patients.
1. Introduction
Mycobacteria are a group of aerobic organisms known for their acid-fastness and thick, lipid-rich cell walls [Citation1]. They are considered one of the oldest bacterial genera, with evidence of their existence dating back millions of years [Citation2]. The most well-known mycobacterial species is Mycobacterium tuberculosis, the causative agent of tuberculosis (TB) that primarily affects the lungs and spreads from person to person through the inhalation of respiratory droplets [Citation3]. While TB has been decreasing around the world, there has been a simultaneous increase in disease caused by nontuberculous mycobacteria (NTM) [Citation4], a diverse group of organisms that are ubiquitous in the environment [Citation5]. With the exception of M. tuberculosis complex, Mycobacterium leprae complex, and Mycobacterium ulcerans, the rest of the species are referred to as NTM [Citation6]. NTM pulmonary disease (NTM-PD) accounts for the majority of NTM-associated diseases, which include lymphadenitis, skin and soft tissue infections, cardiac infection, bone and joint infections, and disseminated disease [Citation7–9].
We conducted a narrative review of the literature to outline the microbiology, environmental sources and transmission pathways, risk factors, epidemiology, diagnosis, treatment recommendations, and management challenges of NTM-PD. This narrative review was based on a synthesis of literature from the PubMed database using the search terms nontuberculous mycobacteria, NTM, nontuberculous mycobacterial pulmonary disease, NTM-PD, Mycobacterium avium, and Mycobacterium abscessus published until July 2023. The authors subjectively reviewed each publication and included those considered most relevant or robust in the review. Information was also retrieved from clinicaltrials.gov when deemed relevant.
2. NTM species
More than 200 NTM species and subspecies have been identified to date [Citation10]. Although these organisms are closely related, they have distinct epidemiologic features and behavior as pathogens. The most relevant groups known to cause NTM-PD worldwide are M. avium complex (MAC) and M. abscessus (MAB) complex [Citation11]. MAC is a group of slowly growing mycobacteria comprising 12 species (M. avium, Mycobacterium intracellulare, Mycobacterium chimaera, Mycobacterium colombiense, Mycobacterium arosiense, Mycobacterium vulneris, Mycobacterium bouchedurhonense, Mycobacterium timonense, Mycobacterium marseillense, Mycobacterium yongonense, Mycobacterium paraintracellulare, and Mycobacterium lepraemurium), whereas MAB organisms are rapidly growing mycobacteria comprising three subspecies (M. abscessus subspecies abscessus, M. abscessus subspecies bolletii, and M. abscessus subspecies massiliense). Following MAC and MAB, Mycobacterium xenopi, Mycobacterium fortuitum, and Mycobacterium kansasii are the next most common causes of NTM-PD [Citation11], and other notable species include Mycobacterium malmoense, Mycobacterium simiae, and Mycobacterium szulgai [Citation12].
3. Environmental sources and transmission pathways
NTM infections are primarily acquired from the environment. NTM organisms can grow in natural as well as artificial water environments (). The soil [Citation23–25], plants [Citation26,Citation27], air [Citation17,Citation28,Citation29], and dust [Citation23,Citation30,Citation31] are also sources of NTM. Inhalation of aerosolized NTM-contaminated water droplets or soil is considered the primary source of transmission of pathogenic NTM species to humans [Citation32]. As such, it should not be surprising that the prevalence of NTM-PD varies regionally, with higher rates in coastal areas compared to continental areas [Citation33–37]. Warm, humid environments with high atmospheric vapor pressure contribute to population risk [Citation11,Citation38], as does exposure to air pollution [Citation39]. Variations in temperature and rainfall may also play a role in environmental exposure to some species of NTM [Citation40]. In fact, there is potential for increased exposure to these pathogens during natural disasters [Citation41] with increased NTM infections linked to hurricanes [Citation42].
Table 1. Potential sources of NTM in natural and artificial water environments.
Prolonged exposure to NTM-contaminated environments, such as certain occupational or recreational settings involving exposure to dust, soil, or water sources, as well as activities like farming, gardening, public bath use, swimming, and hot tub use, can increase the risk of infection [Citation5,Citation43–46]. Healthcare-associated transmission has also been observed in both the inpatient and outpatient settings [Citation21,Citation47–50]. Lastly, there have been rare instances of potential person-to-person transmission reported, particularly in individuals with compromised immune systems [Citation51]. NTM exposure does not always lead to infection or disease. Susceptibility to NTM infections can vary among individuals and populations.
4. Risk factors
NTM-PD can affect any individual, but certain risk factors increase the likelihood of developing infections and disease. Infection often occurs in the setting of underlying structural airway disease as these conditions provide an environment conducive to NTM colonization. Bronchiectasis, chronic obstructive pulmonary disease (COPD), cystic fibrosis (CF), pneumoconiosis, prior TB, pulmonary alveolar proteinosis, α-1-antitrypsin anomalies, and esophageal motility disorders have all been reported to increase the risk of NTM-PD [Citation9,Citation52]. In fact, 80% of the patients with NTM-PD in a large US managed care health plan had at least one underlying lung disease, of which COPD (53%), pneumonia (40%), bronchiectasis (37%), and asthma (23%) were the most common [Citation53]. A recent meta-analysis that included 99 publications affirms the reported association between NTM-PD and various respiratory diseases as the odds ratio (OR) for NTM-PD was significantly increased for bronchiectasis (OR, 21.43), a history of TB (OR, 12.69), interstitial lung disease (OR, 6.39), COPD (OR, 6.63), and asthma (OR, 4.15) [Citation54].
Of particular interest to NTM-PD are the underlying conditions causing structural lung damage such as CF, bronchiectasis, and COPD. The overall prevalence of NTM infection in patients with CF is 7.9% and is increasing over time based on international registry reports [Citation55]. Clinically, NTM-PD increases the rate of CF pulmonary exacerbations three-fold [Citation56] and accelerates deterioration of lung function [Citation57–59]. In COPD patients, the risk of NTM-PD is nine-fold greater compared to those without obstructive lung disease [Citation60]. NTM-PD is also associated with preceding use of inhaled corticosteroids in this population, with evidence for a dose–response relationship [Citation61,Citation62]. Infection doubles the rate of severe exacerbations requiring hospital admissions, leads to a significant deterioration in lung function, and increases the risk of mortality [Citation33,Citation63]. In patients with non-CF bronchiectasis, the global prevalence of NTM from 2006 to 2021 was estimated to be approximately 10% [Citation64,Citation65]. Elderly Caucasian women with a low body mass index seem to be more susceptible to NTM infection in non-CF bronchiectasis [Citation66,Citation67]. In recent years, accumulating evidence has confirmed that patients with an NTM infection or NTM-PD are more likely to have severe bronchiectasis (e.g., diffusely dilated airways, evidence of airflow obstruction, nodular densities often described as tree-in-bud abnormalities, cavities), frequent exacerbations, and higher mortality rates than those without NTM [Citation67–69].
NTM-PD tends to be more prevalent in older individuals, with a higher incidence observed in those ≥65 years [Citation53,Citation60,Citation70,Citation71]. For example, a US study found that the overall annual prevalence of NTM-PD in 2015 was 17 per 100,000 persons but for people ≥65 years, it was 47 per 100,000 persons [Citation53]. The reasons for the increased susceptibility in older age groups are not fully understood but may be due to age-related changes in lung structure and immune function [Citation72]. Furthermore, intensive antimicrobial therapy in chronic disease states may affect the health of the respiratory microbiome, contributing to dysbiosis and potentially making the host more susceptible to NTM infection or disease [Citation73,Citation74].
Some studies have also found that women have a greater risk of NTM-PD compared to men [Citation36,Citation52,Citation53,Citation75–79]. For example, in 2015 the annual prevalence was 17 per 100,000 persons for women compared to the overall prevalence of 12 per 100,000 persons in the US [Citation53]. In addition, Prevots et al. [Citation75] found that the prevalence of NTM-PD was 1.1–1.6-fold greater among women relative to men across four different sites in the US. The reasons for this gender difference are not fully understood and may involve hormonal or genetic factors.
Immune system deficiencies or suppression can increase the risk of NTM-PD. This includes individuals with HIV/AIDS, organ transplant recipients, individuals on immunosuppressive medications, and those with inherited or acquired immunodeficiencies [Citation9,Citation11,Citation52]. Certain genetic conditions that impact the immune response can also predispose individuals to NTM-PD, such as abnormalities of the interleukin-12/interferon-gamma axis, certain human leukocyte antigen alleles, and polymorphisms of solute carrier 11A1 (or natural resistance-associated macrophage protein 1) and vitamin D receptor [Citation80].
The role of genetics in NTM-PD remains scant, but NTM-PD has been reported among families, suggesting there may be a heritable, genetic contribution to this disease [Citation81,Citation82]. Racial or ethnic disparities have also been reported in the literature. For example, Adjemian et al. [Citation36] reported that prevalence among Asians/Pacific Islanders in the US was two-fold greater than among whites. In Hawaii, incidence and risk of NTM-PD are greater among people of Asian ethnicity and lowest among Native Hawaiians and Other Pacific Islanders [Citation83,Citation84], suggesting that biologic and behavioral factors can affect disease susceptibility.
Having one or more risk factors does not guarantee the development of NTM-PD as the interplay between environmental exposure, host susceptibility, and other factors contributes to the overall risk. Furthermore, the long-term outcomes, prognosis, and burden of NTM-PD can vary depending on several factors, including the specific NTM species involved.
5. Epidemiology
NTM infections are not generally subject to mandatory reporting in many countries, including Asia, Europe, and most of the US [Citation85,Citation86]. In fact, it is only notifiable in a few settings, including certain areas of the US, Australia, and Portugal [Citation40,Citation87,Citation88]. This absence of systematic surveillance and reporting mechanisms for NTM-PD limits the availability of comprehensive epidemiological data and makes it difficult to capture the true burden. Moreover, NTM disease is not uniformly defined throughout studies with some reporting prevalence based on one NTM isolate (i.e., NTM infection) and others based on diagnostic codes or guideline criteria for clinically confirmed disease, which may include microbiologic findings, presence of pulmonary or systemic symptoms, and radiologic abnormalities in the lungs [Citation6,Citation85,Citation89].
5.1. Global
NTM-PD is a global health concern as population-based data demonstrate a concerning trend of increasing prevalence worldwide [Citation11]. A recent systematic review of 47 studies that included 285,681 NTM isolates from more than 18 countries found that 82% of the studies reported an increase in NTM infection and 68% reported an increase in NTM disease. The overall annual rate of change for NTM infection and disease per 100,000 persons was 4.0% and 4.1%, respectively, and a majority of studies reported an increasing trend for MAC infection (n = 15/19, 78.9%) and disease (n = 10/12, 83.3%) [Citation90]. Factors influencing this observed increase in NTM infection and disease may include a higher level of physician awareness, improved detection methods, increased NTM exposure, greater host susceptibility including a growing population of immunocompromised and vulnerable individuals, as well as complex medical and surgical procedures [Citation91,Citation92]. The prevalence of NTM is increasing globally albeit with large regional differences as shown in . There are also significant differences in predominant NTM species/groups across geographies (), with MAC being the dominant group of organisms in North America, Europe, Australia/New Zealand, and parts of Asia [Citation11,Citation96] and MAB organisms emerging as the predominant NTM pathogens in parts of Asia, including southern Taiwan [Citation100]. While the reasons for these differences are unclear, climate, cultural practices, and healthcare systems may help account for epidemiological differences among geographies.
Figure 1. Annual prevalence (number of cases per 100,000 persons) of nontuberculous mycobacterial pulmonary disease across different geographies.
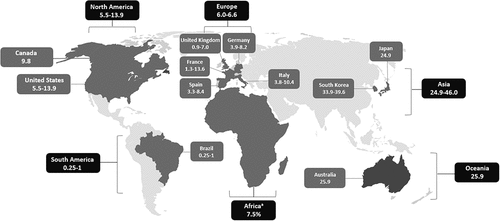
Table 2. Summary of NTM species prevalence across different geographies.
5.2. North America
The prevalence of NTM-PD appears to be increasing in North America. Prevalence data from various US-based studies are shown in , indicating prevalence ranging from 1.4 to 47.5 per 100,000 persons depending on age and geographic location, and annual percent change in prevalence ranging from 2.7% to 9.9% [Citation7,Citation36,Citation53,Citation75,Citation83,Citation101–104]. Canada has shown similar prevalence to the US. Comprehensive population-based studies in Ontario demonstrated an annual disease prevalence of 10 cases per 100,000 persons in 2010. The average annual increase in disease prevalence was 6.3% between 1998 and 2010 [Citation104]. Five-year prevalence data, which may estimate disease prevalence more accurately based on the chronic nature of the disease, revealed a prevalence of 41 per 100,000 persons over the period between 2006 and 2010, an increase from 29 cases per 100,000 persons between 1998 and 2002 [Citation104].
Table 3. Summary of North American studies assessing the prevalence of NTM-PD.
5.3. Europe
Prevalence of NTM-PD in Europe is lower compared to North America and Australia [Citation11], but studies suggest that it is on the rise in several European countries including Spain [Citation105], Portugal [Citation87], Germany [Citation106], the United Kingdom [Citation107–109], and the Netherlands [Citation110]. Schildkraut et al. [Citation85] estimated the annual prevalence of NTM-PD in the European Union (EU)-4 (France, Germany, Italy, Spain) and the United Kingdom (UK) in 2016 using a two-round Delphi NTM-PD-expert consensus-building method at 6.2 cases per 100,000 persons in 2016. Regional prevalence estimates were made using an estimation model that incorporated data obtained from a blinded physician screening survey and a real-world NTM-PD treating-physician/patient-chart observational study. Prevalence per 100,000 persons was found to vary regionally within countries, ranging from 0.9 to 7.0 in the UK, 1.3 to 13.6 in France, 3.9 to 8.2 in Germany, 3.8 to 10.4 in Italy, and 3.3 to 8.4 in Spain.
5.4. Asia
Studies show that the prevalence of NTM-PD in East Asia is increasing, including South Korea, Japan, and Taiwan, especially among large tertiary care centers [Citation77,Citation79,Citation97,Citation111–118]. In fact, a systematic review and meta-analysis that was conducted to estimate the prevalence of NTM in adults with non-CF bronchiectasis reported that the pooled NTM prevalence of six East Asian studies (China, Japan, Korea) was 7.5% greater than that of the eight studies from other countries (Egypt, France, Greece, Israel, Italy, Netherlands, Pakistan, US) in adults with non-CF bronchiectasis [Citation65].
In South Korea, one study estimated that NTM infection prevalence estimates increased five-fold from 2007 to 2016 [Citation113] where the 2016 prevalence estimates were 33.3–39.6 cases of NTM infection per 100,000 persons [Citation113,Citation114]. In Japan, the annual national NTM-PD prevalence was estimated at 24.9 cases per 100,000 persons for the entire country [Citation85] and several studies conducted out of hospitals reported the incidence of NTM-PD in Taiwan to range from 13 to 46 episodes per 100,000 patients [Citation71,Citation115].
5.5. Oceania
Studies conducted in different states of Australia have consistently reported rising rates of NTM infections [Citation40,Citation76,Citation119,Citation120]. In Queensland, Australia, all cases of mycobacterial infection (TB and NTM) are notifiable under the Queensland Public Health Act (2005), which provides an excellent opportunity for large-scale epidemiological research. In 2015, there was a notification rate of 25.9 cases per 100,000 persons and notifications had been increasing approximately 17% per year over the previous four years [Citation120]. The estimated incidence rate increased 2.3-fold from 11.10 per 100,000 in 2001 to 25.88 per 100,000 in 2016 [Citation40].
5.6. Africa
In a systematic review and meta-analysis of 37 articles, the overall prevalence of NTM in pulmonary samples in sub-Saharan Africa was 7.5%, including studies in western (Ghana, Mali, Nigeria), southern (South Africa, Zambia), and eastern (Ethiopia, Kenya, Tanzania, Uganda) Africa [Citation99]. However, the lack of a reliable population denominator makes population prevalence estimates difficult. For example, among patients suspected of having TB, the overall prevalence of NTM isolation ranges from 1.7% in Kenya to 39.1% in Nigeria with Ethiopia, South Africa, and Zambia ranging from 2.7% to 15.1%. In a study conducted out of Kenya, the overall prevalence was 42.4% in patients retreated for TB, whereas mine workers in South Africa had a prevalence of 64.3–68.4%, and patients suspected of having an NTM infection in South Africa and Zambia had a prevalence from 59.3% to 100% [Citation99].
5.7. South America
It is difficult to make conclusions regarding the population frequency of NTM-PD in South America. Based on studies conducted in Brazil, Prevots and Marras [Citation11] suggest an annual prevalence of 1.3–5.3 cases per 100,000 persons for NTM isolation, 0.25–1.0 cases per 100,000 persons for NTM-PD, as well as a six-year period prevalence of NTM-PD of 1.5 cases per 100,000 persons from 2000 to 2005, and a ten-year period prevalence of 10 cases per 100,000 persons for NTM-PD from 1996 to 2005 [Citation11]. More recent studies have assessed annual incidence, with an estimated annual incidence of NTM-PD in French Guiana of 1.07 cases per 100,000 persons [Citation121]. This is similar to a study from Uruguay reporting an incidence of 1.57 cases per 100,000 persons in 2018, which increased from 0.33 cases per 100,000 persons in 2006 [Citation122].
6. Clinical manifestations and diagnostic approaches
NTM-PD can present with a wide range of clinical manifestations that can be nonspecific and mimic any infectious process of the lungs [Citation123]. However, most patients have a chronic, persistent, or recurring cough that may produce sputum, worsen over time, and can be associated with other respiratory symptoms such as dyspnea, hemoptysis, wheezing, chest pain, and systemic manifestations such as fever, fatigue, malaise, and weight loss [Citation9].
The American Thoracic Society (ATS), European Respiratory Society (ERS), European Society of Clinical Microbiology and Infectious Diseases (ESCMID), and Infectious Diseases Society of America (IDSA) have released diagnostic criteria for NTM-PD that include clinical, radiographic, and microbiologic findings [Citation6,Citation124]. Microbiologic confirmation of NTM involves any of the following: (1) positive cultures from at least two separate expectorated sputum samples; (2) positive culture from at least one bronchial wash or lavage; or (3) a transbronchial or other lung biopsy with compatible histopathologic features, such as granulomatous inflammation or acid-fast bacilli, and a positive culture for NTM or ≥1 positive sputum or bronchial wash culture for NTM.
Correct species identification of NTM isolates is clinically important since NTM species differ in their potential to cause clinical disease and in their susceptibility to antimicrobials [Citation125–127]. The most sensitive method to detect NTM from respiratory samples is culture using automated liquid culture systems (e.g. Mycobacteria Growth Indicator Tube (MGIT); BD Biosciences, Sparks, MD, USA) and solid media (e.g., Löwenstein-Jensen or Middlebrook 7H10 and 7H11 media) [Citation91]. Microscopy should be routinely carried out using auramine-phenol staining as it allows the direct evaluation of mycobacteria and provides a useful adjunct to culture [Citation128–131]. All NTM isolates from respiratory samples should be identified using validated molecular or mass spectrometry techniques [Citation91]. Matrix-assisted laser desorption ionization-time of flight (MALDI-TOF) mass spectrometry has shown considerable promise in providing rapid speciation of NTM [Citation132–136]. Unfortunately, some NTM-PD patients do not expectorate sputum [Citation137] and the collection of respiratory specimens for culture and susceptibility testing can take weeks to months to obtain a result in many clinical settings [Citation91]. Therefore, non-sputum, rapid identification and speciation techniques are needed to improve diagnostic and treatment capabilities [Citation138].
The diagnosis of TB can be challenging due to the similarities in presentation and radiological findings to NTM disease. Therefore, differentiation is important to prevent misdiagnosis, and ensure proper care and infection control. Interferon gamma release assays (IGRAs) such as the T-SPOT.TB and QuantiFERON tests measure the amount of IFN-γ produced in response to in vitro stimulation of whole blood with specific M. tuberculosis antigens [Citation139,Citation140]. While they have a moderate positive rate for the diagnosis of NTM [Citation141], they can help discriminate between TB and NTM disease [Citation142,Citation143]. NTM-specific IGRAs may also help to improve the diagnostics of NTM infections in affected patients [Citation144]. The MPT64 test, a rapid immunochromatographic test that targets MPT64 protein in positive cultures by using anti-MPT64 monoclonal antibodies, has high sensitivity and specificity for TB [Citation145,Citation146]. It helps to differentiate between TB and NTM disease [Citation147], especially when combined with MGIT960 cultures [Citation148].
Serological testing has clinical utility in the diagnosis of NTM-PD. For example, in one study from Romania, NTM serodiagnosis correlated well with MAC culture results [Citation149]. Several studies evaluating serum IgA against the glycopeptidolipid core antigen (a cell surface antigen that is found in MAC) have reported a sensitivity of 52–85% and a specificity of 91–100% using the optimal cutoff value of 0.7 U/ml [Citation150–155]; serum IgA against the glycopeptidolipid core antigen also correlated with radiographical changes [Citation156] and has been proposed as a potential biomarker of treatment response [Citation157,Citation158]. In addition, studies assessing assays that measure serum IgG against the mycobacterial antigen A60 have reported both sensitivity (87–95%) and specificity (73–95%) for the diagnosis of MAB infection in patients with cystic fibrosis [Citation159,Citation160]. If validated by larger studies in different cohorts, serological testing could find a potential application in clinical practice.
A real-time polymerase chain reaction (PCR) assay for the detection of MAC that targets the 16S-23S rRNA internal transcribed spacer has been found to be both sensitive (71%) and specific (99.5%) within 24 hours of specimen receipt, with a positive predictive value of 98%, and a negative predictive value of 90% [Citation161]. Other molecular techniques available to detect NTM in respiratory specimens include PCR restriction analysis for the rpoB gene [Citation162] and the GenoType Mycobacteria Direct test [Citation163]. While their utility is somewhat limited at this time [Citation91], two molecular assays based on the DNA/DNA hybridization technique (GenoType CM direct®, Bruker, Billerica, MA, USA and VisionArray Myco®, ZytoVision, Bremerhaven, Germany) may improve rapid identification of mycobacteria from clinical specimens; Schildhaus et al. [Citation164] reported that these two tests had 73.0–89.2% sensitivity, 81.5–96.3% specificity, 86.8–96.4% positive predictive value, and 72.2–84.6% negative predictive value for 37 mycobacterial isolates including 11 TB and 26 NTM isolates.
In addition to developing new, more rapid diagnostic tests, an important research priority is investigating biomarkers associated with disease progression or response to treatment as it can inform clinical decision making [Citation138,Citation165]. Cell-mediated immune responses, rather than antibody responses, could provide an alternate approach for immunologic monitoring of treatment response. Kim et al. [Citation166] reported that declining levels of IL-17 and IL-23 were associated with treatment failure in MAC pulmonary disease (MAC-PD), suggesting that continued impairment of the IL-17 pathway reflects ongoing disease processes. Carbohydrate antigen 19–9 (CA 19–9), a treatment monitoring biomarker for several forms of cancer, may also be a useful marker for monitoring therapeutic responses in NTM-PD. Hong et al. [Citation167] reported that CA 19–9 levels significantly declined in patients with a treatment response within 12 months of therapy, while no difference was observed among patients without a treatment response. Blood-based biomarkers could provide valuable insights into the clinical management of NTM-PD as they are readily available throughout the course of treatment, offer precise measurements that are not reliant on hierarchical scoring systems (such as those used for radiographic findings or microbiology results), and may detect clinical responses that are subtle on physical examination, such as those that may be found with extra-pulmonary forms of NTM disease [Citation165].
7. Guideline-based treatment recommendations
The current guideline-based treatments for NTM-PD are based on recently published recommendations provided by the ATS, ERS, ESCMID, and IDSA [Citation6,Citation124]. They provide susceptibility-based treatment recommendations for NTM-PD in adults (without cystic fibrosis or HIV infection) caused by the most common NTM pathogens including MAC, M. kansasii, M. xenopi, and MAB. An expert panel group has also provided consensus management recommendations for the treatment of less common NTM species including Mycobacterium chelonae, M. fortuitum, Mycobacterium genavense, Mycobacterium gordonae, M. malmoense, M. simiae, and M. szulgai [Citation168]. As the focus of this paper is the most clinically relevant sources of NTM-PD, the treatment recommendations for MAC-PD and MAB pulmonary disease (MAB-PD) are included herein.
Treatment for MAC-PD involves a three-drug regimen that includes a macrolide (azithromycin is preferable to clarithromycin) and ethambutol for a minimum of 12 months after sputum cultures convert from positive to negative. This regimen should be provided three times weekly for noncavitary nodular bronchiectatic disease and daily for cavitary or severe/advanced nodular bronchiectatic disease. Parenteral amikacin or streptomycin is also recommended for patients with cavitary or severe/advanced bronchiectatic or macrolide-resistant MAC-PD for 2–3 months, and amikacin liposome inhalation suspension (ALIS), a polycationic, semisynthetic, broad-spectrum aminoglycoside, is recommended when treatment response is not achieved after six months [Citation6,Citation124].
Treatment of MAB-PD involves a combination of three or more antimicrobials, including macrolides, amikacin, and a β-lactam or imipenem. Disease caused by macrolide-susceptible strains should be treated with a macrolide-containing multidrug-treatment regimen. Disease caused by macrolide-resistant strains may still be treated with a macrolide as it can be used for its immunomodulatory properties, but it is not considered a member of the recommended three-drug regimen. Expert consultation is recommended to guide treatment length [Citation6,Citation124].
Adjunctive surgery is also an effective therapeutic option [Citation169] for select patients with failure of medical management, cavitary disease, drug resistant isolates, or complications such as hemoptysis or severe bronchiectasis [Citation6,Citation124].
7.1. Other drugs under development for NTM-PD
Various other drugs for NTM-PD have reached clinical trials. In addition to being approved for refractory MAC-PD, ALIS is also being assessed as a first-line treatment for MAC-PD when provided with a background regimen in a Phase 3 trial (NCT04677569). Clofazimine, an antimicrobial phenazine dye indicated for leprosy, has in vitro activity against MAC and MAB [Citation170]. Clinical trials report that it is a safe and reasonably tolerated therapeutic that achieves culture conversion and improved treatment outcomes in MAC-PD and MAB-PD [Citation171–173]. As such, it is now recommended as a first-line oral treatment for MAB and an alternative treatment for MAC-PD in patients who are intolerant of first-line drugs [Citation6,Citation124]. Omadacycline (Paratek Pharmaceuticals Inc., Boston, MA, USA), a semisynthetic tetracycline antibiotic approved by the US FDA for the treatment of community-acquired bacterial pneumonia and skin infections [Citation174], has shown in vitro activity against MAB, M. chelonae, M. fortuitum, and M. massiliense [Citation175–177]; safety, efficacy, and tolerability are currently being assessed for MAB-PD in a Phase 2 trial (NCT04922554). Epetraborole (AN2 Therapeutics, Menlo Park, CA, USA), a boron-containing, small molecule inhibitor of bacterial leucyl-tRNA synthetase, has shown in vitro activity against MAC and MAB [Citation178,Citation179]; efficacy, safety, and pharmacokinetics in patients with treatment-refractory MAC-PD are being explored in a Phase 2/3 trial (NCT05327803). Finally, SPR720 (Spero Therapeutics, Inc., Cambridge, MA, USA), a novel aminobenzimidazole antibiotic that inhibits in mycobacteria the ATPase activity of DNA gyrase B (GyrB) [Citation180–183], has shown in vitro activity against some of the major NTM organisms including MAC and MAB [Citation180,Citation184,Citation185]; efficacy, safety, tolerability, and pharmacokinetics in MAC-PD are being explored in a Phase 2 trial (NCT05496374) along with pharmacokinetics with azithromycin and ethambutol co-administration and intrapulmonary pharmacokinetics in healthy volunteers in two Phase 1 trials (NCT05966688 and NCT05955586, respectively).
8. NTM-PD can be challenging to treat
Despite the increasing prevalence of NTM-PD, there is a relative scarcity of high-quality clinical trials evaluating treatment strategies. In fact, current treatment guidelines for NTM-PD are based on available clinical trial data for existing antibiotics [Citation6] that have not been specifically developed for NTM infections [Citation186]. Treatment generally involves multiple drugs for a prolonged duration [Citation6,Citation9,Citation91,Citation124], but even in the setting of prolonged combination therapy, outcomes are poor and mortality rates are high [Citation171,Citation187–193].
The exact number of people treated with guideline-based therapy for NTM-PD is challenging to determine as it depends on various factors; however, some studies show that once diagnosed, only 13–51% of individuals are treated with guideline‐based therapy [Citation118,Citation194–196], of whom only 41% continue at six months, 18% at 12 months, and 11% at 18 months [Citation196]. Furthermore, in many cases, treatment is often not initiated for months to years after a diagnosis [Citation118,Citation192,Citation196,Citation197].
Sputum culture conversion occurs in 34–67% of patients receiving a susceptibility-based treatment regimen [Citation187,Citation190,Citation192,Citation198–201] and in only 11–21% of patients with macrolide-resistant strains [Citation193,Citation202]. Predictors of less favorable outcomes include older age, comorbidities, severe or cavitary disease, MAB infection, poor treatment adherence, drug intolerance, and adverse effects [Citation199,Citation203–210]. Even where culture conversion is achieved, recurrence rates have been found to occur in 25–49% [Citation171,Citation205,Citation211–213], of which 25–54% are true relapses and 46–75% are reinfections [Citation205,Citation212,Citation213].
Adverse drug reactions are very common during the treatment of NTM infections and are associated with treatment interruption or early treatment cessation [Citation190,Citation202,Citation214,Citation215]. For example, among patients enrolled in the US Bronchiectasis and NTM Research Registry, adverse effects occurred in 21% of therapy recipients, of whom 33% discontinued therapy [Citation196]. This is somewhat lower than adverse event reporting in clinical trials [Citation200,Citation216], potentially due to underreporting [Citation217,Citation218].
A wide variety of adverse effects have been reported in the literature, especially among older patients [Citation207]. Macrolides are often associated with gastrointestinal disturbance (e.g. nausea, vomiting, bloating, abdominal pain), decreased auditory acuity, tinnitus, QT prolongation, and hepatitis [Citation186,Citation190,Citation219,Citation220]. The most common rifamycin-induced adverse effects include hepatotoxicity, gastrointestinal disturbance, dermatitis, and immunologic reactions, including acute renal failure and thrombocytopenia [Citation9,Citation186,Citation220,Citation221]. The most well-documented adverse effects linked to ethambutol include ocular toxicity, optic neuritis, vision changes (e.g., blurred vision, color vision disturbance), increases in serum creatinine, and numbness and tingling in the extremities [Citation186,Citation220,Citation221]. Finally, the primary clinical concerns with the aminoglycosides, such as ALIS, are respiratory in nature (e.g., dysphonia, cough, and dyspnea), along with ototoxicity, vestibular toxicity, and nephrotoxicity [Citation186,Citation222].
Various health-related quality of life side effects have also been reported including difficulty walking or interacting with others, fatigue or lack of energy, feelings of sadness or depression, and shortness of breath, wheezing, or other difficulties [Citation223]. The potential for adverse effects and drug–drug interactions limits the tolerability and safety of treatment options, particularly in older individuals [Citation202,Citation203,Citation207].
9. NTM resistance to antimicrobials
NTM species exhibit variability in drug susceptibility, and resistance to commonly used antimicrobial agents can occur. Resistance limits the therapeutic utility of current guideline-based treatments, which can lead to poor clinical outcomes [Citation202,Citation224–227]. NTM either naturally possess or develop high resistance against conventional antibiotics through a variety of mechanisms (). They have several features that restrict drug uptake into the cell including a thick impermeable cell wall, as well as the formation of biofilms and granulomas [Citation229]. They also utilize efflux pumps to remove unwanted molecules that may get inside the cell [Citation230]; this efflux-mediated resistance has been reported for a variety of current guideline-recommended treatments such as macrolides [Citation231–234], fluoroquinolones [Citation235], rifamycins [Citation231], and aminoglycosides [Citation236], as well as other antimicrobials including tetracyclines [Citation236–238], spectinomycin [Citation239], and clofazimine and bedaquiline [Citation240].
Figure 2. Nontuberculous mycobacterial antimicrobial targets and resistance mechanisms. Adapted from van der Laan, Snabilie et al. 2022 [Citation228].
![Figure 2. Nontuberculous mycobacterial antimicrobial targets and resistance mechanisms. Adapted from van der Laan, Snabilie et al. 2022 [Citation228].](/cms/asset/ef2053f6-ca1d-4398-b3d7-0c906bea32fb/ierx_a_2283135_f0002_oc.jpg)
The enzyme catalyzed modification of antibiotics is a major mechanism of antibiotic resistance with β-lactams [Citation241], fluoroquinolones [Citation242], and aminoglycosides [Citation243,Citation244]. Further, functional Erm methyltransferases [Citation245,Citation246] and ADP-ribosylation [Citation247,Citation248] mediate intrinsic macrolide and rifamycin resistance, respectively. Some NTM species such as MAB pathogens are formidable due to inherent resistance mechanisms and the formation of biofilms [Citation228,Citation249]. Their exceptional multidrug resistance to standard antibiotics, including aminoglycosides, rifamycins, tetracyclines, and β-lactams [Citation250], along with their ubiquitous environmental presence make them persistent pathogens that are extremely difficult to treat [Citation251]. Other antibiotic factors, which may have a bearing on outcomes include whether the drug penetrates the pulmonary tissues and macrophages. Tissue penetration can be affected by the route of administration. Additionally, the environment within infected tissues and macrophages is acidic, with a pH range of typically 4 to 5 in macrophages and thus activity at this pH is essential.
Addressing these limitations requires ongoing research and development efforts to improve treatment outcomes for NTM-PD. This includes the development of novel oral antimicrobial agents with improved safety, tolerability, spectrum of activity, efficacy over shorter time periods, and minimal drug–drug interactions [Citation252,Citation253].
10. Conclusion
NTM infections are challenging and affect an increasing number of patients globally. The epidemiology is not uniformly reported, so absolute incidence is unknown. However, the ubiquitous nature makes them a constant threat among patients with structural lung damage. Diagnosis, both clinical and microbiologic, is essential to guide initial therapy which is usually at least 12 months of triple-drug therapy (MAC-PD) or ≥ 3 antimicrobials (MAB-PD) after sputum culture conversion to negative. Such complex therapy can be associated with adverse drug reactions, potential for drug–drug interactions, and development of antimicrobial resistance. New oral therapies active against MAC and MAB, including strains that are resistant to current agents, are urgently needed. This review highlights many of the ongoing challenges of treating patients with NTM-PD.
11. Expert opinion
NTM-PD is a global health concern as population-based data demonstrate increasing prevalence worldwide. A recent systematic review of 47 studies that included 285,681 NTM isolates from more than 18 countries found that 82% of the studies reported an increase in NTM infection and 68% reported an increase in NTM disease. NTM infections can present in a myriad of manifestations including chronic, persistent, or recurring cough that may produce sputum. This may worsen over time and can be associated with other respiratory symptoms such as dyspnea, hemoptysis, wheezing, chest pain, and systemic manifestations such as fever, fatigue, malaise, and weight loss.
Correct species identification of NTM isolates is clinically important since NTM species differ in their potential to cause clinical disease and in their susceptibility to antimicrobials. All NTM isolates from respiratory samples should be identified using validated molecular or mass spectrometry techniques.
Management of NTM infections has been recommended by various societies; treatment for MAC-PD involves triple-drug therapy including a macrolide and treatment for MAB-PD involves a combination of ≥3 antimicrobials including macrolides, amikacin, and a β-lactam or imipenem. The length of therapy, at least 12 months after sputum culture conversion to negative, is a major hurdle for adherence. The lack of well-controlled clinical studies further complicates the selection of appropriate therapy.
These challenges illustrate the requirement for new antimicrobials that are oral, well tolerated, and efficacious. NTM-PD is an increasing clinical and epidemiological challenge as the number of patients with structural lung disease and immunocompromised states increase globally.
Article highlights
The microbiology of the NTM group of organisms is discussed to highlight the complexity of the pathogens.
In addition to the widespread environmental presence of the organisms, the various risk factors for disease are presented.
Global epidemiology is described from a wide range of countries, although the infections are generally not notifiable or routinely monitored.
Laboratory diagnosis of NTM infections is a constant challenge, with new methods being developed to improve the accuracy and efficiency of identification of these infections, also potentially improving the evaluation of microbiologic response to therapy.
NTM-PD is a growing clinical problem and as such, several key societies have produced clinical guidelines to assist clinicians with managing this difficult condition.
Hurdles in treating NTM-PD are discussed, and new therapeutic agents under development are highlighted.
Declaration of interest
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Reviewer disclosures
Peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
Additional information
Funding
References
- Cook GM, Berney M, Gebhard S, et al. Physiology of mycobacteria. Adv Microb Physiol. 2009;55:81–182, 318–9. doi: 10.1016/S0065-2911(09)05502-7
- Hayman J. Mycobacterium ulcerans: an infection from Jurassic time? Lancet. 1984 Nov 3;2(8410):1015–1016. doi: 10.1016/S0140-6736(84)91110-3
- Smith I. Mycobacterium tuberculosis pathogenesis and molecular determinants of virulence. Clin Microbiol Rev. 2003 Jul;16(3):463–496. doi: 10.1128/CMR.16.3.463-496.2003
- Brode SK, Daley CL, Marras TK. The epidemiologic relationship between tuberculosis and non-tuberculous mycobacterial disease: a systematic review. Int J Tuberc Lung Dis. 2014 Nov;18(11):1370–1377. doi: 10.5588/ijtld.14.0120
- Falkinham JO. 3rd. Environmental sources of nontuberculous mycobacteria. Clin Chest Med. 2015 Mar;36(1):35–41. doi: 10.1016/j.ccm.2014.10.003
- Daley CL, Iaccarino JM, Lange C, et al. Treatment of nontuberculous mycobacterial pulmonary disease: an official ATS/ERS/ESCMID/IDSA clinical practice guideline. Clin Infect Dis. 2020 Aug 14;71(4):905–913. doi: 10.1093/cid/ciaa1125
- Winthrop KL, McNelley E, Kendall B, et al. Pulmonary nontuberculous mycobacterial disease prevalence and clinical features: an emerging public health disease. Am J Respir Crit Care Med. 2010 Oct 1;182(7):977–982. doi: 10.1164/rccm.201003-0503OC
- Johnson MM, Odell JA. Nontuberculous mycobacterial pulmonary infections. J Thorac Dis. 2014 Mar;6(3):210–220. doi: 10.3978/j.issn.2072-1439.2013.12.24
- Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007 Feb 15;175(4):367–416. doi: 10.1164/rccm.200604-571ST
- LPSN. Genus Mycobacterium Sudbury, Massachusetts2023. [cited 2023 Jun 6]. Available from: https://lpsn.dsmz.de/genus/mycobacterium
- Prevots DR, Marras TK. Epidemiology of human pulmonary infection with nontuberculous mycobacteria: a review. Clin Chest Med. 2015 Mar;36(1):13–34. doi: 10.1016/j.ccm.2014.10.002
- Yan M, Brode SK, Marras TK. The other nontuberculous mycobacteria: clinical aspects of lung disease caused by less common slowly growing nontuberculous mycobacteria species. Chest. 2023 Feb;163(2):281–291. doi: 10.1016/j.chest.2022.09.025
- Jacobs JM, Stine CB, Baya AM, et al. A review of mycobacteriosis in marine fish. J Fish Dis. 2009 Feb;32(2):119–130. doi: 10.1111/j.1365-2761.2008.01016.x
- Iivanainen EK, Martikainen PJ, Vaananen PK, et al. Environmental factors affecting the occurrence of mycobacteria in brook waters. Appl Environ Microbiol. 1993 Feb;59(2):398–404. doi: 10.1128/aem.59.2.398-404.1993
- Amha YM, Anwar MZ, Kumaraswamy R, et al. Mycobacteria in municipal wastewater treatment and reuse: microbial diversity for screening the occurrence of clinically and environmentally relevant species in arid regions. Environ Sci Technol. 2017 Mar 7;51(5):3048–3056. doi: 10.1021/acs.est.6b05580
- Thomson R, Tolson C, Sidjabat H, et al. Mycobacterium abscessus isolated from municipal water - a potential source of human infection. BMC Infect Dis. 2013 May 25;13(1):241. doi: 10.1186/1471-2334-13-241
- Thomson R, Tolson C, Carter R, et al. Isolation of nontuberculous mycobacteria (NTM) from household water and shower aerosols in patients with pulmonary disease caused by NTM. J Clin Microbiol. 2013 Sep;51(9):3006–3011. doi: 10.1128/JCM.00899-13
- Cooksey RC, Jhung MA, Yakrus MA, et al. Multiphasic approach reveals genetic diversity of environmental and patient isolates of Mycobacterium mucogenicum and Mycobacterium phocaicum associated with an outbreak of bacteremias at a Texas hospital. Appl Environ Microbiol. 2008 Apr;74(8):2480–2487. doi: 10.1128/AEM.02476-07
- Falkinham JO 3rd, Iseman MD, de Haas P, et al. Mycobacterium avium in a shower linked to pulmonary disease. J Water Health. 2008 Jun;6(2):209–213. doi: 10.2166/wh.2008.232
- Lumb R, Stapledon R, Scroop A, et al. Investigation of spa pools associated with lung disorders caused by Mycobacterium avium complex in immunocompetent adults. Appl Environ Microbiol. 2004 Aug;70(8):4906–4910. doi: 10.1128/AEM.70.8.4906-4910.2004
- Millar BC, Moore JE. Hospital ice, ice machines, and water as sources of nontuberculous mycobacteria: description of qualitative risk assessment models to determine host-nontuberculous mycobacteria interplay. Int J Mycobacteriol. 2020 Oct;9(4):347–362. doi: 10.4103/ijmy.ijmy_179_20
- Gira AK, Reisenauer AH, Hammock L, et al. Furunculosis due to Mycobacterium mageritense associated with footbaths at a nail salon. J Clin Microbiol. 2004 Apr;42(4):1813–1817. doi: 10.1128/JCM.42.4.1813-1817.2004
- Lahiri A, Kneisel J, Kloster I, et al. Abundance of Mycobacterium avium ssp. hominissuis in soil and dust in Germany - implications for the infection route. Lett Appl Microbiol. 2014 Jul;59(1):65–70. doi: 10.1111/lam.12243
- De Groote MA, Pace NR, Fulton K, et al. Relationships between Mycobacterium isolates from patients with pulmonary mycobacterial infection and potting soils. Appl Environ Microbiol. 2006 Dec;72(12):7602–7606. doi: 10.1128/AEM.00930-06
- Iivanainen EK, Martikainen PJ, Räisänen ML, et al. Mycobacteria in boreal coniferous forest soils. FEMS Microbiol Ecol. 1997;23(4):325–332. doi: 10.1016/S0168-6496(97)00040-8
- Bouam A, Armstrong N, Levasseur A, et al. Mycobacterium terramassiliense, Mycobacterium rhizamassiliense and Mycobacterium numidiamassiliense sp. nov., three new Mycobacterium simiae complex species cultured from plant roots. Sci Rep. 2018 Jun 18;8(1):9309. doi: 10.1038/s41598-018-27629-1
- Tran PM, Dahl JL. Mycobacterium sarraceniae sp. nov. And Mycobacterium helvum sp. nov., isolated from the pitcher plant Sarracenia purpurea. Int J Syst Evol Microbiol. 2016 Nov;66(11):4480–4485. doi: 10.1099/ijsem.0.001377
- Primm TP, Lucero CA, Falkinham JO. 3rd. Health impacts of environmental mycobacteria. Clin Microbiol Rev. 2004 Jan;17(1):98–106. doi: 10.1128/CMR.17.1.98-106.2004
- Mangione EJ, Huitt G, Lenaway D, et al. Nontuberculous mycobacterial disease following hot tub exposure. Emerg Infect Dis. 2001 Nov;7(6):1039–1042. doi: 10.3201/eid0706.010623
- Choi S, Choi MI. Solation of nontuberculous mycobacteria (NTM) from air conditioner dust. Korean J Clin Lab Sci. 2017;49:435–438. doi: 10.15324/kjcls.2017.49.4.435
- Leski TA, Malanoski AP, Gregory MJ, et al. Application of a broad-range resequencing array for detection of pathogens in desert dust samples from Kuwait and Iraq. Appl Environ Microbiol. 2011 Jul;77(13):4285–4292. doi: 10.1128/AEM.00021-11
- Parikh A, Vinnard C, Fahrenfeld N, et al. Revisiting John snow to meet the challenge of nontuberculous mycobacterial lung disease. Int J Environ Res Public Health. 2019 Nov 1;16(21):4250. doi: 10.3390/ijerph16214250
- Pyarali FF, Schweitzer M, Bagley V, et al. Increasing non-tuberculous mycobacteria infections in veterans with COPD and association with increased risk of mortality. Front Med. 2018;5:311. doi: 10.3389/fmed.2018.00311
- Jankovic M, Samarzija M, Sabol I, et al. Geographical distribution and clinical relevance of non-tuberculous mycobacteria in Croatia. Int J Tuberc Lung Dis. 2013 Jun;17(6):836–841. doi: 10.5588/ijtld.12.0843
- Liu CF, Song YM, He WC, et al. Nontuberculous mycobacteria in China: incidence and antimicrobial resistance spectrum from a nationwide survey. Infect Dis Poverty. 2021 Apr 29;10(1):59. doi: 10.1186/s40249-021-00844-1
- Adjemian J, Olivier KN, Seitz AE, et al. Prevalence of nontuberculous mycobacterial lung disease in U.S. Medicare beneficiaries. Am J Respir Crit Care Med. 2012 Apr 15;185(8):881–886. doi: 10.1164/rccm.201111-2016OC
- Strollo SE, Adjemian J, Adjemian MK, et al. The burden of pulmonary nontuberculous mycobacterial disease in the United states. Ann Am Thorac Soc. 2015 Oct;12(10):1458–1464. doi: 10.1513/AnnalsATS.201503-173OC
- Adjemian J, Olivier KN, Seitz AE, et al. Spatial clusters of nontuberculous mycobacterial lung disease in the United states. Am J Respir Crit Care Med. 2012 Sep 15;186(6):553–558. doi: 10.1164/rccm.201205-0913OC
- Maki T, Noda J, Morimoto K, et al. Long-range transport of airborne bacteria over East Asia: Asian dust events carry potentially nontuberculous Mycobacterium populations. Environ Int. 2022 Oct;168:107471
- Thomson RM, Furuya-Kanamori L, Coffey C, et al. Influence of climate variables on the rising incidence of nontuberculous mycobacterial (NTM) infections in Queensland, Australia 2001-2016. Sci Total Environ. 2020 Oct 20;740:139796. doi: 10.1016/j.scitotenv.2020.139796
- Honda JR, Bernhard JN, Chan ED. Natural disasters and nontuberculous mycobacteria: a recipe for increased disease? Chest. 2015 Feb;147(2):304–308. doi: 10.1378/chest.14-0974
- Kambali S, Quinonez E, Sharifi A, et al. Pulmonary nontuberculous mycobacterial disease in Florida and association with large-scale natural disasters. BMC Public Health. 2021 Nov 10;21(1):2058. doi: 10.1186/s12889-021-12115-7
- Nishiuchi Y, Iwamoto T, Maruyama F. Infection sources of a common non-tuberculous mycobacterial Pathogen, Mycobacterium avium complex. Front Med. 2017;4:27. doi: 10.3389/fmed.2017.00027
- Jeon D. Infection source and epidemiology of nontuberculous mycobacterial lung disease. Tuberc Respir Dis (Seoul). 2019 Apr;82(2):94–101. doi: 10.4046/trd.2018.0026
- Park Y, Kwak SH, Yong SH, et al. The association between behavioral risk factors and nontuberculous mycobacterial pulmonary disease. Yonsei Med J. 2021 Aug;62(8):702–707. doi: 10.3349/ymj.2021.62.8.702
- Gundacker ND, Gonzalez JA, Sheinin YM, et al. Hot tub lung: case report and review of the literature. WMJ. 2022 Jul;121(2):E31–E33.
- Campos-Gutierrez S, Ramos-Real MJ, Abreu R, et al. Pseudo-outbreak of Mycobacterium fortuitum in a hospital bronchoscopy unit. Am J Infect Control. 2020 Jul;48(7):765–769. doi: 10.1016/j.ajic.2019.11.019
- Falkinham JO. Growth in catheter biofilms and antibiotic resistance of Mycobacterium avium. J Med Microbiol. 2007 Feb;56(Pt 2):250–254. doi: 10.1099/jmm.0.46935-0
- Motawea KR, Rabea RK, Elhalag RH, et al. Cosmetic operative care abroad leads to a multidrug-resistant Mycobacterium abscessus infection in a patient: a case report. J Med Case Rep. 2022 Nov 29;16(1):448. doi: 10.1186/s13256-022-03678-z
- Bolcato M, Rodriguez D, Aprile A. Risk management in the New frontier of professional liability for nosocomial infection: review of the literature on mycobacterium chimaera. Int J Environ Res Public Health. 2020 Oct 7;17(19):7328. doi: 10.3390/ijerph17197328
- Aitken ML, Limaye A, Pottinger P, et al. Respiratory outbreak of Mycobacterium abscessus subspecies massiliense in a lung transplant and cystic fibrosis center. Am J Respir Crit Care Med. 2012 Jan 15;185(2):231–232. doi: 10.1164/ajrccm.185.2.231
- Chan ED, Iseman MD. Underlying host risk factors for nontuberculous mycobacterial lung disease. Semin Respir Crit Care Med. 2013 Feb;34(1):110–123. doi: 10.1055/s-0033-1333573
- Winthrop KL, Marras TK, Adjemian J, et al. Incidence and prevalence of nontuberculous mycobacterial lung disease in a large U.S. Managed Care health plan, 2008-2015. Ann Am Thorac Soc. 2020 Feb;17(2):178–185. doi: 10.1513/AnnalsATS.201804-236OC
- Loebinger MR, Quint JK, van der Laan R, et al. Risk factors for nontuberculous mycobacterial pulmonary disease: a systematic literature review and meta-analysis. Chest. 2023 Jun 17;164(5):1115–1124. doi: 10.1016/j.chest.2023.06.014
- Prieto MD, Alam ME, Franciosi AN, et al. Global burden of nontuberculous mycobacteria in the cystic fibrosis population: a systematic review and meta-analysis. ERJ Open Res. 2023 Jan;9(1):00336–2022. doi: 10.1183/23120541.00336-2022
- Low D, Wilson DA, Flume PA. Screening practices for nontuberculous mycobacteria at US cystic fibrosis centers. J Cyst Fibros. 2020 Jul;19(4):569–574. doi: 10.1016/j.jcf.2020.02.013
- Esther CR Jr., Esserman DA, Gilligan P, et al. Chronic Mycobacterium abscessus infection and lung function decline in cystic fibrosis. J Cyst Fibros. 2010 Mar;9(2):117–123. doi: 10.1016/j.jcf.2009.12.001
- Qvist T, Taylor-Robinson D, Waldmann E, et al. Comparing the harmful effects of nontuberculous mycobacteria and gram negative bacteria on lung function in patients with cystic fibrosis. J Cyst Fibros. 2016 May;15(3):380–385. doi: 10.1016/j.jcf.2015.09.007
- Eikani MS, Nugent M, Poursina A, et al. Clinical course and significance of nontuberculous mycobacteria and its subtypes in cystic fibrosis. BMC Infect Dis. 2018 Jul 6;18(1):311. doi: 10.1186/s12879-018-3200-z
- Marras TK, Campitelli MA, Kwong JC, et al. Risk of nontuberculous mycobacterial pulmonary disease with obstructive lung disease. Eur Respir J. 2016 Sep;48(3):928–931. doi: 10.1183/13993003.00033-2016
- Andrejak C, Nielsen R, Thomsen VO, et al. Chronic respiratory disease, inhaled corticosteroids and risk of non-tuberculous mycobacteriosis. Thorax. 2013 Mar;68(3):256–262. doi: 10.1136/thoraxjnl-2012-201772
- Shu CC, Wei YF, Chen KH, et al. Inhaled corticosteroids increase risk of nontuberculous mycobacterial lung disease: a nested case-control study and meta-analysis. J Infect Dis. 2022 Feb 15;225(4):627–636. doi: 10.1093/infdis/jiab428
- Huang CT, Tsai YJ, Wu HD, et al. Impact of non-tuberculous mycobacteria on pulmonary function decline in chronic obstructive pulmonary disease. Int J Tuberc Lung Dis. 2012 Apr;16(4):539–545. doi: 10.5588/ijtld.11.0412
- Chu H, Zhao L, Xiao H, et al. Prevalence of nontuberculous mycobacteria in patients with bronchiectasis: a meta-analysis. Arch Med Sci. 2014 Aug 29;10(4):661–668. doi: 10.5114/aoms.2014.44857
- Zhou Y, Mu W, Zhang J, et al. Global prevalence of non-tuberculous mycobacteria in adults with non-cystic fibrosis bronchiectasis 2006-2021: a systematic review and meta-analysis. BMJ Open. 2022 Aug 1;12(8):e055672. doi: 10.1136/bmjopen-2021-055672
- Mirsaeidi M, Hadid W, Ericsoussi B, et al. Non-tuberculous mycobacterial disease is common in patients with non-cystic fibrosis bronchiectasis. Int J Infect Dis. 2013 Nov;17(11):e1000–4. doi: 10.1016/j.ijid.2013.03.018
- Aksamit TR, O’Donnell AE, Barker A, et al. Adult patients with bronchiectasis: a first look at the US bronchiectasis research registry. Chest. 2017 May;151(5):982–992. doi: 10.1016/j.chest.2016.10.055
- Yin H, Gu X, Wang Y, et al. Clinical characteristics of patients with bronchiectasis with nontuberculous mycobacterial disease in Mainland China: a single center cross-sectional study. BMC Infect Dis. 2021 Dec 6;21(1):1216. doi: 10.1186/s12879-021-06917-8
- Lin CY, Huang HY, Hsieh MH, et al. Impacts of Nontuberculous Mycobacteria Isolates in Non-cystic Fibrosis Bronchiectasis: A 16-Year Cohort Study in Taiwan. Front Microbiol. 2022;13:868435. doi: 10.3389/fmicb.2022.868435
- Wang PH, Pan SW, Wang SM, et al. The Impact of nontuberculous mycobacteria species on mortality in patients with nontuberculous mycobacterial lung disease. Front Microbiol. 2022;13:909274. doi: 10.3389/fmicb.2022.909274
- Huang HL, Cheng MH, Lu PL, et al. Epidemiology and predictors of NTM pulmonary infection in Taiwan - a retrospective, five-year multicenter study. Sci Rep. 2017 Nov 24;7(1):16300. doi: 10.1038/s41598-017-16559-z
- Fifor A, Krukowski K, Honda JR. Sex, ancestry, senescence, and aging (SAnSA) are stark drivers of nontuberculous mycobacterial pulmonary disease. J Clin Tuberc Other Mycobact Dis. 2022 Feb;26:100297. doi: 10.1016/j.jctube.2022.100297
- Namasivayam S, Sher A, Glickman MS, et al. The microbiome and tuberculosis: early evidence for cross talk. MBio. 2018 Sep 18;9(5). doi: 10.1128/mBio.01420-18
- Thornton CS, Mellett M, Jarand J, et al. The respiratory microbiome and nontuberculous mycobacteria: an emerging concern in human health. Eur Respir Rev. 2021 Jun 30;30(160):200299. doi: 10.1183/16000617.0299-2020
- Prevots DR, Shaw PA, Strickland D, et al. Nontuberculous mycobacterial lung disease prevalence at four integrated health care delivery systems. Am J Respir Crit Care Med. 2010 Oct 1;182(7):970–976. doi: 10.1164/rccm.201002-0310OC
- Thomson RM. Centre NTMwgaQTC, Queensland mycobacterial reference L. Changing epidemiology of pulmonary nontuberculous mycobacteria infections. Emerg Infect Dis. 2010 Oct;16(10):1576–1583. doi: 10.3201/eid1610.091201
- Park Y, Kim CY, Park MS, et al. Age- and sex-related characteristics of the increasing trend of nontuberculous mycobacteria pulmonary disease in a tertiary hospital in South Korea from 2006 to 2016. Korean J Intern Med. 2020 Nov;35(6):1424–1431. doi: 10.3904/kjim.2019.395
- Cassidy PM, Hedberg K, Saulson A, et al. Nontuberculous mycobacterial disease prevalence and risk factors: a changing epidemiology. Clin Infect Dis. 2009 Dec 15;49(12):e124–9. doi: 10.1086/648443
- Izumi K, Morimoto K, Hasegawa N, et al. Epidemiology of adults and children treated for nontuberculous mycobacterial pulmonary disease in Japan. Ann Am Thorac Soc. 2019 Mar;16(3):341–347. doi: 10.1513/AnnalsATS.201806-366OC
- Sexton P, Harrison AC. Susceptibility to nontuberculous mycobacterial lung disease. Eur Respir J. 2008 Jun;31(6):1322–1333. doi: 10.1183/09031936.00140007
- Colombo RE, Hill SC, Claypool RJ, et al. Familial clustering of pulmonary nontuberculous mycobacterial disease. Chest. 2010 Mar;137(3):629–634. doi: 10.1378/chest.09-1173
- Kobashi Y, Yoshida K, Niki Y, et al. Sibling cases of Mycobacterium avium complex disease associated with hematological disease. J Infect Chemother. 2006 Oct;12(5):331–334. doi: 10.1007/s10156-006-0461-Z
- Adjemian J, Frankland TB, Daida YG, et al. Epidemiology of Nontuberculous Mycobacterial Lung Disease and Tuberculosis, Hawaii, USA. Emerg Infect Dis. 2017 Mar;23(3):439–447. doi: 10.3201/eid2303.161827
- Blakney RA, Ricotta EE, Frankland TB, et al. Incidence of Nontuberculous Mycobacterial Pulmonary Infection, by Ethnic Group, Hawaii, USA, 2005-2019. Emerg Infect Dis. 2022 Aug;28(8):1543–1550. doi: 10.3201/eid2808.212375
- Schildkraut JA, Gallagher J, Morimoto K, et al. Epidemiology of nontuberculous mycobacterial pulmonary disease in Europe and Japan by Delphi estimation. Respir med. 2020 Nov;173:106164
- van Ingen J, Obradovic M, Hassan M, et al. Nontuberculous mycobacterial lung disease caused by Mycobacterium avium complex - disease burden, unmet needs, and advances in treatment developments. Expert Rev Respir Med. 2021 Nov;15(11):1387–1401. doi: 10.1080/17476348.2021.1987891
- Oliveira MJ, Gaio AR, Gomes M, et al. Mycobacterium avium infection in Portugal. Int J Tuberc Lung Dis. 2017 Feb 1;21(2):218–222. doi: 10.5588/ijtld.16.0002
- Winthrop KL, Henkle E, Walker A, et al. On the reportability of nontuberculous mycobacterial disease to public health authorities. Ann Am Thorac Soc. 2017 Mar;14(3):314–317. doi: 10.1513/AnnalsATS.201610-802PS
- Adjemian J, Daniel-Wayman S, Ricotta E, et al. Epidemiology of nontuberculous mycobacteriosis. Semin Respir Crit Care Med. 2018 Jun;39(3):325–335. doi: 10.1055/s-0038-1651491
- Dahl VN, Molhave M, Floe A, et al. Global trends of pulmonary infections with nontuberculous mycobacteria: a systematic review. Int J Infect Dis. 2022 Dec;125:120–131. doi: 10.1016/j.ijid.2022.10.013
- Haworth CS, Banks J, Capstick T, et al. British Thoracic Society guidelines for the management of non-tuberculous mycobacterial pulmonary disease (NTM-PD). Thorax. 2017 Nov;72(Suppl 2):ii1–ii64. doi: 10.1136/thoraxjnl-2017-210927
- Ahmed I, Tiberi S, Farooqi J, et al. Non-tuberculous mycobacterial infections-A neglected and emerging problem. Int J Infect Dis. 2020 Mar;92S:S46–S50
- Hoefsloot W, van Ingen J, Andrejak C, et al. The geographic diversity of nontuberculous mycobacteria isolated from pulmonary samples: an NTM-NET collaborative study. Eur Respir J. 2013 Dec;42(6):1604–1613. doi: 10.1183/09031936.00149212
- Tsai YV, Derrick C, Yunusa I, et al. Epidemiology, outcomes and tolerability of protracted treatment of nontuberculous mycobacterial infections at a community teaching hospital in the southeastern United states. Antibiotics. 2022 Nov 29;11(12):1720. doi: 10.3390/antibiotics11121720
- Zweijpfenning SMH, van Ingen J, Hoefsloot W. Geographic distribution of nontuberculous mycobacteria isolated from clinical specimens: asystematic review. Semin Respir Crit Care Med. 2018 Jun;39(3):336–342. doi: 10.1055/s-0038-1660864
- Dahl VN, Laursen LL, He Y, et al. Species distribution among patients with nontuberculous mycobacteria pulmonary disease in Europe. J Infect. 2023 Nov;87(5):469–472. doi: 10.1016/j.jinf.2023.03.010
- Lee YM, Kim MJ, Kim YJ. Increasing trend of nontuberculous mycobacteria isolation in a referral clinical laboratory in South Korea. Medicina (Kaunas). 2021 Jul 16;57(7):720. doi: 10.3390/medicina57070720
- Ryoo SW, Shin S, Shim MS, et al. Spread of nontuberculous mycobacteria from 1993 to 2006 in Koreans. J Clin Lab Anal. 2008;22(6):415–420. doi: 10.1002/jcla.20278
- Okoi C, Anderson STB, Antonio M, et al. Non-tuberculous mycobacteria isolated from pulmonary samples in sub-Saharan Africa - a systematic review and meta analyses. Sci Rep. 2017 Sep 20;7(1):12002. doi: 10.1038/s41598-017-12175-z
- Cheng A, Sun HY, Tsai YT, et al. Longitudinal non-cystic fibrosis trends of pulmonary Mycobacterium abscessus disease from 2010 to 2017: spread of the “globally successful clone” in Asia. ERJ Open Res. 2021 Jan;7(1):00191–2020. doi: 10.1183/23120541.00191-2020
- Donohue MJ, Wymer L. Increasing prevalence rate of nontuberculous mycobacteria infections in Five states, 2008-2013. Ann Am Thorac Soc. 2016 Dec;13(12):2143–2150. doi: 10.1513/AnnalsATS.201605-353OC
- Boyle DP, Zembower TR, Reddy S, et al. Comparison of Clinical Features, Virulence, and Relapse among Mycobacterium avium Complex Species. Am J Respir Crit Care Med. 2015 Jun 1;191(11):1310–1317. doi: 10.1164/rccm.201501-0067OC
- Dean SG, Ricotta EE, Fintzi J, et al. Mycobacterial testing trends, United states, 2009-2015(1). Emerg Infect Dis. 2020 Sep;26(9):2243–2246. doi: 10.3201/eid2609.200749
- Marras TK, Mendelson D, Marchand-Austin A, et al. Pulmonary nontuberculous mycobacterial disease, Ontario, Canada, 1998-2010. Emerg Infect Dis. 2013 Nov;19(11):1889–1891. doi: 10.3201/eid1911.130737
- Santin M, Barrabeig I, Malchair P, et al. Pulmonary infections with nontuberculous mycobacteria, Catalonia, Spain, 1994-2014. Emerg Infect Dis. 2018 Jun;24(6):1091–1094. doi: 10.3201/eid2406.172095
- Ringshausen FC, Wagner D, de Roux A, et al. Prevalence of nontuberculous mycobacterial pulmonary disease, Germany, 2009-2014. Emerg Infect Dis. 2016 Jun;22(6):1102–1105. doi: 10.3201/eid2206.151642
- Henry MT, Inamdar L, O’Riordain D, et al. Nontuberculous mycobacteria in non-HIV patients: epidemiology, treatment and response. Eur Respir J. 2004 May;23(5):741–746. doi: 10.1183/09031936.04.00114004
- Moore JE, Kruijshaar ME, Ormerod LP, et al. Increasing reports of non-tuberculous mycobacteria in England, Wales and Northern Ireland, 1995-2006. BMC Public Health. 2010 Oct 15;10(1):612. doi: 10.1186/1471-2458-10-612
- Cowman S, Burns K, Benson S, et al. The antimicrobial susceptibility of non-tuberculous mycobacteria. J Infect. 2016 Mar;72(3):324–331. doi: 10.1016/j.jinf.2015.12.007
- van Ingen J, Hoefsloot W, Dekhuijzen PN, et al. The changing pattern of clinical mycobacterium avium isolation in the Netherlands. Int J Tuberc Lung Dis. 2010 Sep;14(9):1176–1180.
- Koh WJ, Chang B, Jeong BH, et al. Increasing recovery of nontuberculous mycobacteria from respiratory specimens over a 10-year period in a tertiary referral hospital in South Korea. Tuberc Respir Dis (Seoul). 2013 Nov;75(5):199–204. doi: 10.4046/trd.2013.75.5.199
- Ko RE, Moon SM, Ahn S, et al. Changing epidemiology of nontuberculous mycobacterial lung diseases in a tertiary referral hospital in Korea between 2001 and 2015. J Korean Med Sci. 2018 Feb 19;33(8):e65. doi: 10.3346/jkms.2018.33.e65
- Lee H, Myung W, Koh WJ, et al. Epidemiology of nontuberculous mycobacterial infection, South Korea, 2007-2016. Emerg Infect Dis. 2019 Mar;25(3):569–572. doi: 10.3201/eid2503.181597
- Park SC, Kang MJ, Han CH, et al. Prevalence, incidence, and mortality of nontuberculous mycobacterial infection in Korea: a nationwide population-based study. BMC Pulm Med. 2019 Aug 1;19(1):140. doi: 10.1186/s12890-019-0901-z
- Chien JY, Lai CC, Sheng WH, et al. Pulmonary infection and colonization with nontuberculous mycobacteria, Taiwan, 2000-2012. Emerg Infect Dis. 2014 Aug;20(8):1382–1385. doi: 10.3201/eid2008.131673
- Ding LW, Lai CC, Lee LN, et al. Disease caused by non-tuberculous mycobacteria in a university hospital in Taiwan, 1997-2003. Epidemiol Infect. 2006 Oct;134(5):1060–1067. doi: 10.1017/S0950268805005698
- Namkoong H, Kurashima A, Morimoto K, et al. Epidemiology of pulmonary nontuberculous mycobacterial disease, Japan. Emerg Infect Dis. 2016 Jun;22(6):1116–1117. doi: 10.3201/eid2206.151086
- Kim HO, Lee K, Choi HK, et al. Incidence, comorbidities, and treatment patterns of nontuberculous mycobacterial infection in South Korea. Medicine (Baltimore). 2019 Nov;98(45):e17869. doi: 10.1097/MD.0000000000017869
- O’Brien DP, Currie BJ, Krause VL. Nontuberculous mycobacterial disease in northern Australia: a case series and review of the literature. Clin Infect Dis. 2000 Oct;31(4):958–967. doi: 10.1086/318136
- Thomson R, Donnan E, Konstantinos A. Notification of nontuberculous mycobacteria: an Australian perspective. Ann Am Thorac Soc. 2017 Mar;14(3):318–323. doi: 10.1513/AnnalsATS.201612-994OI
- Chaptal M, Andrejak C, Bonifay T, et al. Epidemiology of infection by pulmonary non-tuberculous mycobacteria in French Guiana 2008-2018. PLoS Negl Trop Dis. 2022 Sep;16(9):e0010693. doi: 10.1371/journal.pntd.0010693
- Greif G, Coitinho C, van Ingen J, et al. Species distribution and isolation Frequency of nontuberculous mycobacteria, Uruguay. Emerg Infect Dis. 2020 May;26(5):1014–1018. doi: 10.3201/eid2605.191631
- Bethencourt Mirabal A, Ferrer G. Lung Nontuberculous Mycobacterial Infections. In StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan.
- Daley CL, Iaccarino JM, Lange C, et al. Treatment of nontuberculous mycobacterial pulmonary disease: an official ATS/ERS/ESCMID/IDSA clinical practice guideline. Eur Respir J. 2020 Jul;56(1):2000535. doi: 10.1183/13993003.00535-2020
- Brown-Elliott BA, Wallace RJ Jr. Clinical and taxonomic status of pathogenic nonpigmented or late-pigmenting rapidly growing mycobacteria. Clin Microbiol Rev. 2002 Oct;15(4):716–746. doi: 10.1128/CMR.15.4.716-746.2002
- Koh WJ, Jeon K, Lee NY, et al. Clinical significance of differentiation of Mycobacterium massiliense from Mycobacterium abscessus. Am J Respir Crit Care Med. 2011 Feb 1;183(3):405–410. doi: 10.1164/rccm.201003-0395OC
- Choi GE, Shin SJ, Won CJ, et al. Macrolide treatment for Mycobacterium abscessus and Mycobacterium massiliense infection and inducible resistance. Am J Respir Crit Care Med. 2012 Nov 1;186(9):917–925. doi: 10.1164/rccm.201111-2005OC
- Steingart KR, Henry M, Ng V, et al. Fluorescence versus conventional sputum smear microscopy for tuberculosis: a systematic review. Lancet Infect Dis. 2006 Sep;6(9):570–581. doi: 10.1016/S1473-3099(06)70578-3
- Ulukanligil M, Aslan G, Tasci S. A comparative study on the different staining methods and number of specimens for the detection of acid fast bacilli. Mem Inst Oswaldo Cruz. 2000 Nov;95(6):855–858. doi: 10.1590/S0074-02762000000600019
- Wright PW, Wallace RJ Jr., Wright NW, et al. Sensitivity of fluorochrome microscopy for detection of Mycobacterium tuberculosis versus nontuberculous mycobacteria. J Clin Microbiol. 1998 Apr;36(4):1046–1049. doi: 10.1128/JCM.36.4.1046-1049.1998
- Murray SJ, Barrett A, Magee JG, et al. Optimisation of acid fast smears for the direct detection of mycobacteria in clinical samples. J Clin Pathol. 2003 Aug;56(8):613–615. doi: 10.1136/jcp.56.8.613
- Saleeb PG, Drake SK, Murray PR, et al. Identification of mycobacteria in solid-culture media by matrix-assisted laser desorption ionization-time of flight mass spectrometry. J Clin Microbiol. 2011 May;49(5):1790–1794. doi: 10.1128/JCM.02135-10
- Lotz A, Ferroni A, Beretti JL, et al. Rapid identification of mycobacterial whole cells in solid and liquid culture media by matrix-assisted laser desorption ionization-time of flight mass spectrometry. J Clin Microbiol. 2010 Dec;48(12):4481–4486. doi: 10.1128/JCM.01397-10
- El Khechine A, Couderc C, Flaudrops C, et al. Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry identification of mycobacteria in routine clinical practice. PLoS One. 2011;6(9):e24720. doi: 10.1371/journal.pone.0024720
- Fangous MS, Mougari F, Gouriou S, et al. Classification algorithm for subspecies identification within the Mycobacterium abscessus species, based on matrix-assisted laser desorption ionization-time of flight mass spectrometry. J Clin Microbiol. 2014 Sep;52(9):3362–3369. doi: 10.1128/JCM.00788-14
- Rodriguez-Sanchez B, Ruiz-Serrano MJ, Marin M, et al. Evaluation of matrix-assisted laser desorption ionization-time of flight mass spectrometry for identification of nontuberculous mycobacteria from clinical isolates. J Clin Microbiol. 2015 Aug;53(8):2737–2740. doi: 10.1128/JCM.01380-15
- Huang JH, Kao PN, Adi V, et al. Mycobacterium avium-intracellulare pulmonary infection in HIV-negative patients without preexisting lung disease: diagnostic and management limitations. Chest. 1999 Apr;115(4):1033–1040. doi: 10.1378/chest.115.4.1033
- Daley CL, Winthrop KL. Mycobacterium avium complex: addressing gaps in diagnosis and management. J Infect Dis. 2020 Aug 20;222(Suppl 4):S199–S211. doi: 10.1093/infdis/jiaa354
- Herrera V, Perry S, Parsonnet J, et al. Clinical application and limitations of interferon-gamma release assays for the diagnosis of latent tuberculosis infection. Clin Infect Dis. 2011 Apr 15;52(8):1031–1037. doi: 10.1093/cid/cir068
- Hamada Y, Gupta RK, Quartagno M, et al. Predictive performance of interferon-gamma release assays and the tuberculin skin test for incident tuberculosis: an individual participant data meta-analysis. EClinicalMedicine. 2023 Feb;56:101815.
- Guan CP, Wu YH, Wang XF, et al. The performance of interferon gamma release assays in patients with nontuberculous mycobacterial infection: a systematic review and meta-analysis. Eur J Clin Microbiol Infect Dis. 2023 Oct;42(10):1251–1262. doi: 10.1007/s10096-023-04662-1
- Martinez-Planas A, Baquero-Artigao F, Santiago B, et al. Interferon-gamma release assays differentiate between Mycobacterium avium complex and tuberculous Lymphadenitis in children. J Pediatr. 2021 Sep;236:211–218 e2 doi: 10.1016/j.peds.2021.05.008
- Yang C, Luo X, Fan L, et al. Performance of interferon-gamma release assays in the diagnosis of nontuberculous mycobacterial diseases-A retrospective survey from 2011 to 2019. Front Cell Infect Microbiol. 2020;10:571230. doi: 10.3389/fcimb.2020.571230
- Steindor M, Stehling F, Olivier M, et al. Species-specific interferon-gamma release assay for the diagnosis of Mycobacterium abscessus complex infection. Front Microbiol. 2021;12:692395. doi: 10.3389/fmicb.2021.692395
- Cao XJ, Li YP, Wang JY, et al. MPT64 assays for the rapid detection of Mycobacterium tuberculosis. BMC Infect Dis. 2021 Apr 10;21(1):336. doi: 10.1186/s12879-021-06022-w
- Yin X, Zheng L, Lin L, et al. Commercial MPT64-based tests for rapid identification of Mycobacterium tuberculosis complex: a meta-analysis. J Infect. 2013 Nov;67(5):369–377. doi: 10.1016/j.jinf.2013.06.009
- Dadheech M, Malhotra AG, Patel S, et al. Molecular identification of non-tuberculous mycobacteria in suspected tuberculosis cases in central India. Cureus. 2023 Jun;15(6):e39992. doi: 10.7759/cureus.39992
- Chen S, Wang F, Xue Y, et al. Doubled nontuberculous mycobacteria isolation as a consequence of changes in the diagnosis algorithm. Infect Drug Resist. 2022;15:3347–3355. doi: 10.2147/IDR.S368671
- Stavri H, Ulea I, Radu DL, et al. Serodiagnosis of environmental mycobacterial infections. J Microbiol Methods. 2011 Sep;86(3):283–290. doi: 10.1016/j.mimet.2011.05.010
- Kitada S, Kobayashi K, Ichiyama S, et al. Serodiagnosis of Mycobacterium avium-complex pulmonary disease using an enzyme immunoassay kit. Am J Respir Crit Care Med. 2008 Apr 1;177(7):793–797. doi: 10.1164/rccm.200705-771OC
- Jeong BH, Kim SY, Jeon K, et al. Serodiagnosis of Mycobacterium avium complex and Mycobacterium abscessus complex pulmonary disease by use of IgA antibodies to glycopeptidolipid core antigen. J Clin Microbiol. 2013 Aug;51(8):2747–2749. doi: 10.1128/JCM.00702-13
- Kitada S, Levin A, Hiserote M, et al. Serodiagnosis of Mycobacterium avium complex pulmonary disease in the USA. Eur Respir J. 2013 Aug;42(2):454–460. doi: 10.1183/09031936.00098212
- Shu CC, Ato M, Wang JT, et al. Sero-diagnosis of Mycobacterium avium complex lung disease using serum immunoglobulin A antibody against glycopeptidolipid antigen in Taiwan. PLoS One. 2013;8(11):e80473. doi: 10.1371/journal.pone.0080473
- Kobayashi K. Serodiagnosis of Mycobacterium avium complex disease in humans: translational research from basic mycobacteriology to clinical medicine. Jpn J Infect Dis. 2014;67(5):329–332. doi: 10.7883/yoken.67.329
- Shibata Y, Horita N, Yamamoto M, et al. Diagnostic test accuracy of anti-glycopeptidolipid-core IgA antibodies for Mycobacterium avium complex pulmonary disease: systematic review and meta-analysis. Sci Rep. 2016 Jul 4;6(1):29325. doi: 10.1038/srep29325
- Kitada S, Nishiuchi Y, Hiraga T, et al. Serological test and chest computed tomography findings in patients with Mycobacterium avium complex lung disease. Eur Respir J. 2007 Jun;29(6):1217–1223. doi: 10.1183/09031936.00061806
- Jhun BW, Kim SY, Park HY, et al. Changes in serum IgA antibody levels against the glycopeptidolipid core antigen during antibiotic treatment of Mycobacterium avium complex lung disease. Jpn J Infect Dis. 2017 Sep 25;70(5):582–585. doi: 10.7883/yoken.JJID.2016.523
- Kitada S, Maekura R, Yoshimura K, et al. Levels of Antibody against Glycopeptidolipid Core as a Marker for Monitoring Treatment Response in Mycobacterium avium Complex Pulmonary Disease: a Prospective Cohort Study. J Clin Microbiol. 2017 Mar;55(3):884–892. doi: 10.1128/JCM.02010-16
- Ferroni A, Sermet-Gaudelus I, Le Bourgeois M, et al. Measurement of immunoglobulin G against Mycobacterial antigen A60 in patients with cystic fibrosis and lung infection due to Mycobacterium abscessus. Clin Infect Dis. 2005 Jan 1;40(1):58–66. doi: 10.1086/426442
- Qvist T, Pressler T, Taylor-Robinson D, et al. Serodiagnosis of Mycobacterium abscessus complex infection in cystic fibrosis. Eur Respir J. 2015 Sep;46(3):707–716. doi: 10.1183/09031936.00011815
- Tran AC, Halse TA, Escuyer VE, et al. Detection of Mycobacterium avium complex DNA directly in clinical respiratory specimens: opportunities for improved turn-around time and cost savings. Diagn Microbiol Infect Dis. 2014 May;79(1):43–48. doi: 10.1016/j.diagmicrobio.2014.01.019
- Kim S, Park EM, Kwon OJ, et al. Direct application of the PCR restriction analysis method for identifying NTM species in AFB smear-positive respiratory specimens. Int J Tuberc Lung Dis. 2008 Nov;12(11):1344–1346.
- Seagar AL, Prendergast C, Emmanuel FX, et al. Evaluation of the GenoType mycobacteria direct assay for the simultaneous detection of the Mycobacterium tuberculosis complex and four atypical mycobacterial species in smear-positive respiratory specimens. J Med Microbiol. 2008 May;57(Pt 5):605–611. doi: 10.1099/jmm.0.47484-0
- Schildhaus HU, Steindor M, Kolsch B, et al. GenoType CM direct® and VisionArray Myco® for the rapid identification of mycobacteria from clinical specimens. J Clin Med. 2022 Apr 25;11(9):2404. doi: 10.3390/jcm11092404
- Vinnard C, Mezochow A, Oakland H, et al. Assessing response to therapy for nontuberculous mycobacterial lung disease: Quo Vadis? Front Microbiol. 2018;9:2813. doi: 10.3389/fmicb.2018.02813
- Kim SY, Koh WJ, Park HY, et al. Changes in serum immunomolecules during antibiotic therapy for Mycobacterium avium complex lung disease. Clin Exp Immunol. 2014 Apr;176(1):93–101. doi: 10.1111/cei.12253
- Hong JY, Jang SH, Kim SY, et al. Elevated serum CA 19-9 levels in patients with pulmonary nontuberculous mycobacterial disease. Braz J Infect Dis. 2016 Jan;20(1):26–32. doi: 10.1016/j.bjid.2015.09.005
- Lange C, Bottger EC, Cambau E, et al. Consensus management recommendations for less common non-tuberculous mycobacterial pulmonary diseases. Lancet Infect Dis. 2022 Jul;22(7):e178–e190. doi: 10.1016/S1473-3099(21)00586-7
- Kim JY, Lee HW, Yim JJ, et al. Outcomes of adjunctive surgery in patients with nontuberculous mycobacterial pulmonary disease: a systematic review and meta-analysis. Chest. 2023 Apr;163(4):763–777. doi: 10.1016/j.chest.2022.09.037
- Kwak N, Whang J, Yang JS, et al. Minimal Inhibitory Concentration of Clofazimine among clinical isolates of nontuberculous mycobacteria and its Impact on treatment outcome. Chest. 2021 Feb;159(2):517–523. doi: 10.1016/j.chest.2020.07.040
- Jarand J, Davis JP, Cowie RL, et al. Long-term follow-up of Mycobacterium avium complex lung disease in patients treated with regimens including clofazimine and/or rifampin. Chest. 2016 May;149(5):1285–1293. doi: 10.1378/chest.15-0543
- Yang B, Jhun BW, Moon SM, et al. Clofazimine-containing regimen for the treatment of Mycobacterium abscessus lung disease. Antimicrob Agents Chemother. 2017 Jun;61(6). doi: 10.1128/AAC.02052-16
- Pfaeffle HOI, Alameer RM, Marshall MH, et al. Clofazimine for treatment of multidrug-resistant non-tuberculous mycobacteria. Pulm Pharmacol Ther. 2021 Oct;70:102058
- Opal S, File TM, van der Poll T, et al. An integrated safety Summary of Omadacycline, a novel aminomethylcycline antibiotic. Clin Infect Dis. 2019 Aug 1;69(Suppl 1):S40–S47. doi: 10.1093/cid/ciz398
- Shoen C, Benaroch D, Sklaney M, et al. In vitro activities of Omadacycline against rapidly growing mycobacteria. Antimicrob Agents Chemother. 2019 May;63(5). doi: 10.1128/AAC.02522-18
- Kaushik A, Ammerman NC, Martins O, et al. In vitro activity of New tetracycline analogs omadacycline and eravacycline against drug-resistant clinical isolates of Mycobacterium abscessus. Antimicrob Agents Chemother. 2019 Jun;63(6). doi: 10.1128/AAC.00470-19
- Brown-Elliott BA, Wallace RJ Jr. In Vitro Susceptibility Testing of Omadacycline against Nontuberculous Mycobacteria. Antimicrob Agents Chemother. 2021 Feb 17;65(3). doi: 10.1128/AAC.01947-20
- DeStefano MS, Shoen CM, Alley MRK, et al. In vitro Activities of Epetraborole, a novel bacterial Leucyl-tRNA Synthetase inhibitor, against Mycobacterium avium complex isolates [poster 1713]. (WA) DC: IDWeek; 2022.
- Ganapathy US, Gengenbacher M, Dick T. Epetraborole Is Active against Mycobacterium abscessus. Antimicrob Agents Chemother. 2021 Sep 17;65(10):e0115621. doi: 10.1128/AAC.01156-21
- Locher CP, Jones SM, Hanzelka BL, et al. A novel inhibitor of gyrase B is a potent drug candidate for treatment of tuberculosis and nontuberculosis mycobacterial infections. Antimicrob Agents Chemother. 2015 Mar;59(3):1455–1465. doi: 10.1128/AAC.04347-14
- Grossman TH, Bartels DJ, Mullin S, et al. Dual targeting of GyrB and ParE by a novel aminobenzimidazole class of antibacterial compounds. Antimicrob Agents Chemother. 2007 Feb;51(2):657–666. doi: 10.1128/AAC.00596-06
- Durcik M, Tomasic T, Zidar N, et al. ATP-competitive DNA gyrase and topoisomerase IV inhibitors as antibacterial agents. Expert Opin Ther Pat. 2019 Mar;29(3):171–180. doi: 10.1080/13543776.2019.1575362
- Henderson SR, Stevenson CEM, Malone B, et al. Structural and mechanistic analysis of ATPase inhibitors targeting mycobacterial DNA gyrase. J Antimicrob Chemother. 2020 Oct 1;75(10):2835–2842. doi: 10.1093/jac/dkaa286
- Cotroneo N, Stokes SS, Pucci MJ, et al. Efficacy of SPR720 in murine models of nontuberculous mycobacterial pulmonary infection. J Antimicrob Chemother. 2023.
- Rubio A, Stapleton M, Verma D, et al. Potent activity of a novel gyrase inhibitor (SPR719/SPR720) in vitro and in a prolonged acute Mycobacterium abscessus mouse model of infection [poster SUN-539]. (WA) DC2018: ASM Microbe/American Society for Microbiology; 2018.
- Egelund EF, Fennelly KP, Peloquin CA. Medications and monitoring in nontuberculous mycobacteria infections. Clin Chest Med. 2015 Mar;36(1):55–66. doi: 10.1016/j.ccm.2014.11.001
- Abate G, Stapleton JT, Rouphael N, et al. Variability in the management of adults with pulmonary nontuberculous mycobacterial disease. Clin Infect Dis. 2021 Apr 8;72(7):1127–1137. doi: 10.1093/cid/ciaa252
- Marras TK, Vinnard C, Zhang Q, et al. Relative risk of all-cause mortality in patients with nontuberculous mycobacterial lung disease in a US managed care population. Respir med. 2018 Dec;145:80–88. doi: 10.1016/j.rmed.2018.10.022
- Mirsaeidi M, Machado RF, Garcia JG, et al. Nontuberculous mycobacterial disease mortality in the United states, 1999-2010: a population-based comparative study. PLoS One. 2014;9(3):e91879. doi: 10.1371/journal.pone.0091879
- Kwak N, Park J, Kim E, et al. Treatment outcomes of Mycobacterium avium complex lung disease: a systematic review and meta-analysis. Clin Infect Dis. 2017 Oct 1;65(7):1077–1084. doi: 10.1093/cid/cix517
- Diel R, Lipman M, Hoefsloot W. High mortality in patients with Mycobacterium avium complex lung disease: a systematic review. BMC Infect Dis. 2018 May 3;18(1):206. doi: 10.1186/s12879-018-3113-x
- Im Y, Hwang NY, Kim K, et al. Impact of time between diagnosis and treatment for nontuberculous mycobacterial pulmonary disease on culture conversion and all-cause mortality. Chest. 2022 May;161(5):1192–1200. doi: 10.1016/j.chest.2021.10.048
- Park Y, Lee EH, Jung I, et al. Clinical characteristics and treatment outcomes of patients with macrolide-resistant Mycobacterium avium complex pulmonary disease: a systematic review and meta-analysis. Respir Res. 2019 Dec 18;20(1):286. doi: 10.1186/s12931-019-1258-9
- Adjemian J, Prevots DR, Gallagher J, et al. Lack of adherence to evidence-based treatment guidelines for nontuberculous mycobacterial lung disease. Ann Am Thorac Soc. 2014 Jan;11(1):9–16. doi: 10.1513/AnnalsATS.201304-085OC
- Izumi K, Morimoto K, Uchimura K, et al. Population-based survey of antimycobacterial drug use among patients with non-tuberculosis mycobacterial pulmonary disease. ERJ Open Res. 2020;6(1):00097–2019. doi: 10.1183/23120541.00097-2019
- Ku JH, Henkle E, Aksamit TR, et al. Treatment of nontuberculous mycobacterial (NTM) pulmonary infection in the US bronchiectasis and NTM registry: treatment patterns, adverse events, and adherence to american thoracic Society/Infectious disease Society of America treatment guidelines. Clin Infect Dis. 2023 Jan 13;76(2):338–341. doi: 10.1093/cid/ciac788
- Kwak N, Lee JH, Kim HJ, et al. New-onset nontuberculous mycobacterial pulmonary disease in bronchiectasis: tracking the clinical and radiographic changes. BMC Pulm Med. 2020 Nov 10;20(1):293. doi: 10.1186/s12890-020-01331-3
- Kwak N, Kim SA, Choi SM, et al. Longitudinal changes in health-related quality of life according to clinical course among patients with non-tuberculous mycobacterial pulmonary disease: a prospective cohort study. BMC Pulm Med. 2020 May 7;20(1):126. doi: 10.1186/s12890-020-1165-3
- Pasipanodya JG, Ogbonna D, Ferro BE, et al. Systematic review and meta-analyses of the effect of chemotherapy on pulmonary Mycobacterium abscessus outcomes and disease recurrence. Antimicrob Agents Chemother. 2017 Nov;61(11). doi: 10.1128/AAC.01206-17
- Miwa S, Shirai M, Toyoshima M, et al. Efficacy of clarithromycin and ethambutol for Mycobacterium avium complex pulmonary disease. A preliminary study. Ann Am Thorac Soc. 2014 Jan;11(1):23–29. doi: 10.1513/AnnalsATS.201308-266OC
- Diel R, Nienhaus A, Ringshausen FC, et al. Microbiologic outcome of interventions against mycobacterium avium complex pulmonary disease: a systematic review. Chest. 2018 Apr;153(4):888–921. doi: 10.1016/j.chest.2018.01.024
- Morimoto K, Namkoong H, Hasegawa N, et al. Macrolide-Resistant Mycobacterium avium Complex Lung Disease: Analysis of 102 Consecutive Cases. Ann Am Thorac Soc. 2016 Nov;13(11):1904–1911. doi: 10.1513/AnnalsATS.201604-246OC
- Cheng LP, Chen SH, Lou H, et al. Factors associated with treatment outcome in patients with nontuberculous mycobacterial pulmonary disease: a large population-based retrospective cohort study in Shanghai. Trop Med Infect Dis. 2022 Feb 15;7(2):27. doi: 10.3390/tropicalmed7020027
- Kim BG, Jhun BW, Kim H, et al. Treatment outcomes of Mycobacterium avium complex pulmonary disease according to disease severity. Sci Rep. 2022 Feb 4;12(1):1970. doi: 10.1038/s41598-022-06022-z
- Koh WJ, Moon SM, Kim SY, et al. Outcomes of Mycobacterium avium complex lung disease based on clinical phenotype. Eur Respir J. 2017 Sep;50(3):1602503. doi: 10.1183/13993003.02503-2016
- Zweijpfenning S, Kops S, Magis-Escurra C, et al. Treatment and outcome of non-tuberculous mycobacterial pulmonary disease in a predominantly fibro-cavitary disease cohort. Respir med. 2017 Oct;131:220–224
- Kim JY, Kim NY, Jung HW, et al. Old age is associated with worse treatment outcome and frequent adverse drug reaction in Mycobacterium avium complex pulmonary disease. BMC Pulm Med. 2022 Jul 14;22(1):269. doi: 10.1186/s12890-022-02063-2
- Aliberti S, Sotgiu G, Castellotti P, et al. Real-life evaluation of clinical outcomes in patients undergoing treatment for non-tuberculous mycobacteria lung disease: a ten-year cohort study. Respir med. 2020 Apr;164:105899. doi: 10.1016/j.rmed.2020.105899
- Kwon YS, Han M, Kwon BS, et al. Discontinuation rates attributed to adverse events and treatment outcomes between clarithromycin and azithromycin in Mycobacterium avium complex lung disease: a propensity score analysis. J Glob Antimicrob Resist. 2020 Sep;22:106–112. doi: 10.1016/j.jgar.2020.01.004
- Diel R, Ringshausen F, Richter E, et al. Microbiological and clinical outcomes of treating non-Mycobacterium avium complex nontuberculous mycobacterial pulmonary disease: a systematic review and meta-analysis. Chest. 2017 Jul;152(1):120–142. doi: 10.1016/j.chest.2017.04.166
- Lee BY, Kim S, Hong Y, et al. Risk factors for recurrence after successful treatment of Mycobacterium avium complex lung disease. Antimicrob Agents Chemother. 2015;59(6):2972–2977. doi: 10.1128/AAC.04577-14
- Boyle DP, Zembower TR, Qi C. Relapse versus Reinfection of Mycobacterium avium Complex Pulmonary Disease. Patient Characteristics and Macrolide Susceptibility. Ann Am Thorac Soc. 2016 Nov;13(11):1956–1961. doi: 10.1513/AnnalsATS.201605-344BC
- Wallace RJ Jr., Brown-Elliott BA, McNulty S, et al. Macrolide/Azalide therapy for nodular/bronchiectatic mycobacterium avium complex lung disease. Chest. 2014 Aug;146(2):276–282. doi: 10.1378/chest.13-2538
- Kwon YS, Kwon BS, Kim OH, et al. Treatment outcomes after discontinuation of ethambutol due to adverse events in Mycobacterium avium complex lung disease. J Korean Med Sci. 2020 Mar 9;35(9):e59. doi: 10.3346/jkms.2020.35.e59
- Balavoine C, Blanc F-X, Lanotte P, et al. Adverse events during treatment of nontuberculous mycobacterial lung disease: do they really matter? Eur Respir J. 2018;52(suppl 62):PA2664. doi: 10.1183/13993003.congress-2018.PA2664
- Griffith DE, Brown BA, Girard WM, et al. Adverse events associated with high-dose rifabutin in macrolide-containing regimens for the treatment of Mycobacterium avium complex lung disease. Clin Infect Dis. 1995 Sep;21(3):594–598. doi: 10.1093/clinids/21.3.594
- Costa C, Abeijon P, Rodrigues DA, et al. Factors associated with underreporting of adverse drug reactions by patients: a systematic review. Int J Clin Pharm. 2023 May 29. doi: 10.1007/s11096-023-01592-y
- Garcia-Abeijon P, Costa C, Taracido M, et al. Factors associated with underreporting of adverse drug reactions by health Care professionals: a systematic review update. Drug Saf. 2023 Jul;46(7):625–636. doi: 10.1007/s40264-023-01302-7
- Brown BA, Griffith DE, Girard W, et al. Relationship of adverse events to serum drug levels in patients receiving high-dose azithromycin for mycobacterial lung disease. Clin Infect Dis. 1997 May;24(5):958–964. doi: 10.1093/clinids/24.5.958
- van Ingen J, Aliberti S, Andrejak C, et al. Management of Drug Toxicity in Mycobacterium avium Complex Pulmonary Disease: An Expert Panel Survey. Clin Infect Dis. 2021 Jul 1;73(1):e256–e259. doi: 10.1093/cid/ciaa1361
- Kamii Y, Nagai H, Kawashima M, et al. Adverse reactions associated with long-term drug administration in Mycobacterium avium complex lung disease. Int J Tuberc Lung Dis. 2018 Dec 1;22(12):1505–1510. doi: 10.5588/ijtld.18.0171
- Griffith DE, Eagle G, Thomson R, et al. Amikacin Liposome Inhalation Suspension for Treatment-Refractory Lung Disease Caused by Mycobacterium avium Complex (CONVERT). A Prospective, Open-Label, Randomized Study. Am J Respir Crit Care Med. 2018 Dec 15;198(12):1559–1569. doi: 10.1164/rccm.201807-1318OC
- Pravosud V, Mannino DM, Prieto D, et al. Symptom burden and medication use among patients with nontuberculous mycobacterial lung disease. Chronic Obstr Pulm Dis. 2021 Apr 27;8(2):243–254. doi: 10.15326/jcopdf.2020.0184
- Kim SY, Jhun BW, Moon SM, et al. Mutations in gyrA and gyrB in Moxifloxacin-Resistant Mycobacterium avium Complex and Mycobacterium abscessus Complex Clinical Isolates. Antimicrob Agents Chemother. 2018 Sep;62(9). doi: 10.1128/AAC.00527-18
- Griffith DE, Brown-Elliott BA, Langsjoen B, et al. Clinical and molecular analysis of macrolide resistance in Mycobacterium avium complex lung disease. Am J Respir Crit Care Med. 2006 Oct 15;174(8):928–934. doi: 10.1164/rccm.200603-450OC
- Kadota T, Matsui H, Hirose T, et al. Analysis of drug treatment outcome in clarithromycin-resistant Mycobacterium avium complex lung disease. BMC Infect Dis. 2016 Jan 27;16(1):31. doi: 10.1186/s12879-016-1384-7
- Moon SM, Park HY, Kim SY, et al. Clinical Characteristics, Treatment Outcomes, and Resistance Mutations Associated with Macrolide-Resistant Mycobacterium avium Complex Lung Disease. Antimicrob Agents Chemother. 2016 Nov;60(11):6758–6765. doi: 10.1128/AAC.01240-16
- van der Laan R, Snabilie A, Obradovic M. Meeting the challenges of NTM-PD from the perspective of the organism and the disease process: innovations in drug development and delivery. Respir Res. 2022 Dec 24;23(1):376. doi: 10.1186/s12931-022-02299-w
- Saxena S, Spaink HP, Forn-Cuni G. Drug Resistance in Nontuberculous Mycobacteria: Mechanisms and Models. Biology. 2021 Jan 29;10(2):96. doi: 10.3390/biology10020096
- Nasiri MJ, Haeili M, Ghazi M, et al. New insights in to the intrinsic and acquired drug resistance mechanisms in mycobacteria. Front Microbiol. 2017;8:681. doi: 10.3389/fmicb.2017.00681
- Li XZ, Zhang L, Nikaido H. Efflux pump-mediated intrinsic drug resistance in Mycobacterium smegmatis. Antimicrob Agents Chemother. 2004 Jul;48(7):2415–2423. doi: 10.1128/AAC.48.7.2415-2423.2004
- Schmalstieg AM, Srivastava S, Belkaya S, et al. The antibiotic resistance arrow of time: efflux pump induction is a general first step in the evolution of mycobacterial drug resistance. Antimicrob Agents Chemother. 2012 Sep;56(9):4806–4815. doi: 10.1128/AAC.05546-11
- Machado D, Cannalire R, Santos Costa S, et al. Boosting effect of 2-phenylquinoline efflux inhibitors in combination with macrolides against Mycobacterium smegmatis and Mycobacterium avium. ACS Infect Dis. 2015 Dec 11;1(12):593–603. doi: 10.1021/acsinfecdis.5b00052
- Duan W, Li X, Ge Y, et al. Mycobacterium tuberculosis Rv1473 is a novel macrolides ABC Efflux Pump regulated by WhiB7. Future Microbiol. 2019 Jan;14(1):47–59. doi: 10.2217/fmb-2018-0207
- Liu J, Takiff HE, Nikaido H. Active efflux of fluoroquinolones in Mycobacterium smegmatis mediated by LfrA, a multidrug efflux pump. J Bacteriol. 1996 Jul;178(13):3791–3795. doi: 10.1128/jb.178.13.3791-3795.1996
- Silva PE, Bigi F, Santangelo MP, et al. Characterization of P55, a multidrug efflux pump in Mycobacterium bovis and Mycobacterium tuberculosis. Antimicrob Agents Chemother. 2001 Mar;45(3):800–804. doi: 10.1128/AAC.45.3.800-804.2001
- De Rossi E, Blokpoel MC, Cantoni R, et al. Molecular cloning and functional analysis of a novel tetracycline resistance determinant, tet(V), from Mycobacterium smegmatis. Antimicrob Agents Chemother. 1998 Aug;42(8):1931–1937. doi: 10.1128/AAC.42.8.1931
- Ramon-Garcia S, Martin C, Ainsa JA, et al. Characterization of tetracycline resistance mediated by the efflux pump Tap from Mycobacterium fortuitum. J Antimicrob Chemother. 2006 Feb;57(2):252–259. doi: 10.1093/jac/dki436
- Hurst-Hess KR, Phelps GA, Wilt LA, et al. Mab2780c, a TetV-like efflux pump, confers high-level spectinomycin resistance in mycobacterium abscessus. Tuberculosis (Edinb). 2023 Jan;138:102295. doi: 10.1016/j.tube.2022.102295
- Gutierrez AV, Richard M, Roquet-Baneres F, et al. The TetR family transcription factor MAB_2299c regulates the expression of two distinct mmps-mmpl efflux pumps involved in cross-resistance to clofazimine and bedaquiline in Mycobacterium abscessus. Antimicrob Agents Chemother. 2019 Oct;63(10). doi: 10.1128/AAC.01000-19
- Lavollay M, Dubee V, Heym B, et al. In vitro activity of cefoxitin and imipenem against Mycobacterium abscessus complex. Clin Microbiol Infect. 2014 May;20(5):O297–300. doi: 10.1111/1469-0691.12405
- Adjei MD, Heinze TM, Deck J, et al. Acetylation and nitrosation of ciprofloxacin by environmental strains of mycobacteria. Can J Microbiol. 2007 Jan;53(1):144–147. doi: 10.1139/w06-101
- Ainsa JA, Perez E, Pelicic V, et al. Aminoglycoside 2’-N-acetyltransferase genes are universally present in mycobacteria: characterization of the aac(2’)-Ic gene from Mycobacterium tuberculosis and the aac(2’)-id gene from Mycobacterium smegmatis. Mol Microbiol. 1997 Apr;24(2):431–441. doi: 10.1046/j.1365-2958.1997.3471717.x
- Ripoll F, Pasek S, Schenowitz C, et al. Non mycobacterial virulence genes in the genome of the emerging pathogen Mycobacterium abscessus. PLoS One. 2009 Jun 19;4(6):e5660. doi: 10.1371/journal.pone.0005660
- Wetzstein N, Kohl TA, Schultze TG, et al. Antimicrobial susceptibility and phylogenetic relations in a german cohort infected with Mycobacterium abscessus. J Clin Microbiol. 2020 Nov 18;58(12). doi: 10.1128/JCM.01813-20
- Lipworth S, Hough N, Leach L, et al. Whole-genome sequencing for predicting clarithromycin resistance in Mycobacterium abscessus. Antimicrob Agents Chemother. 2019 Jan;63(1). doi: 10.1128/AAC.01204-18
- Rominski A, Roditscheff A, Selchow P, et al. Intrinsic rifamycin resistance of Mycobacterium abscessus is mediated by ADP-ribosyltransferase MAB_0591. J Antimicrob Chemother. 2017 Feb;72(2):376–384. doi: 10.1093/jac/dkw466
- Baysarowich J, Koteva K, Hughes DW, et al. Rifamycin antibiotic resistance by ADP-ribosylation: structure and diversity of arr. Proc Natl Acad Sci U S A. 2008 Mar 25;105(12):4886–4891. doi: 10.1073/pnas.0711939105
- Griffith DE, Daley CL. Treatment of Mycobacterium abscessus pulmonary disease. Chest. 2022 Jan;161(1):64–75. doi: 10.1016/j.chest.2021.07.035
- Luthra S, Rominski A, Sander P. The role of antibiotic-target-modifying and antibiotic-modifying enzymes in Mycobacterium abscessus drug resistance. Front Microbiol. 2018;9:2179. doi: 10.3389/fmicb.2018.02179
- Nessar R, Cambau E, Reyrat JM, et al. Mycobacterium abscessus: a new antibiotic nightmare. J Antimicrob Chemother. 2012 Apr;67(4):810–818. doi: 10.1093/jac/dkr578
- Wu ML, Aziz DB, Dartois V, et al. NTM drug discovery: status, gaps and the way forward. Drug Discov Today. 2018 Aug;23(8):1502–1519. doi: 10.1016/j.drudis.2018.04.001
- Ruth MM, van Ingen J. New insights in the treatment of nontuberculous mycobacterial pulmonary disease. Future Microbiol. 2017 Oct;12(13):1109–1112. doi: 10.2217/fmb-2017-0144
