Abstract
PD-L1 is overexpressed on the surface of tumor cells and binds to PD-1, resulting in tumor immune escape. Therapeutic strategies to target the PD-1/PD-L1 pathway involve blocking the binding. Immune checkpoint inhibitors have limited efficacy against tumors because PD-L1 is also present in the cytoplasm. PD-L1 of post-translational modifications (PTMs) have uncovered numerous mechanisms contributing to carcinogenesis and have identified potential therapeutic targets. Therefore, small molecule inhibitors can block crucial carcinogenic signaling pathways, making them a potential therapeutic option. To better develop small molecule inhibitors, we have summarized the PTMs of PD-L1. This review discusses the regulatory mechanisms of small molecule inhibitors in carcinogenesis and explore their potential applications, proposing a novel approach for tumor immunotherapy based on PD-L1 PTM.
GRAPHICAL ABSTRACT
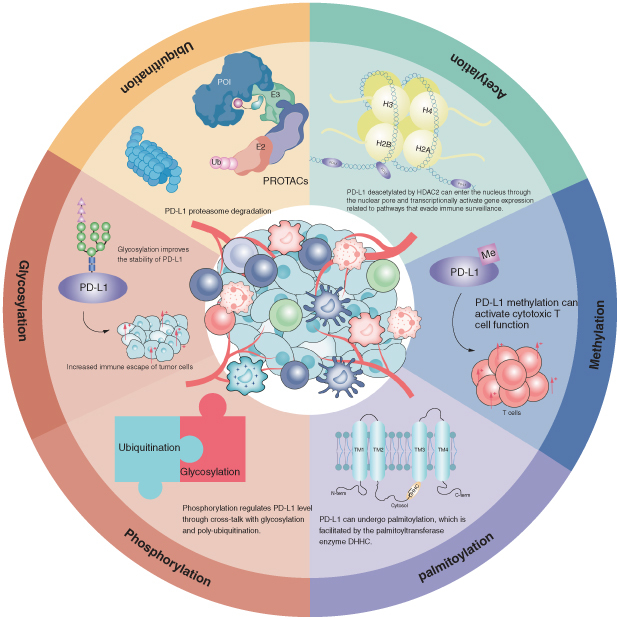
The post-translational modifications of PD-L1, including N-glycosylation, ubiquitination, deubiquitination, palmitoylation, phosphorylation, acetylation and methylation, are summarized.
This review provides a comprehensive overview of small molecule compounds targeting the PD-1/PD-L1 signaling pathway, which either degrade or up-regulate PD-L1 expression by modulating transcription factors or proteins involved in this pathway.
Enhanced potential of small molecule inhibitors targeting post-translational modifications of PD-L1 for cancer therapy.
Financial disclosure
This work was supported by the National Natural Science Foundation of China (82273792, 82003597), Natural Science Foundation of Hebei Province (B2022201092, B2021201026), Natural Science Foundation of Science and Technology Project of Hebei Education Department (QN2022057), Natural Science Interdisciplinary Research Program of Hebei University (DXK202215), Undergraduate Innovation and Entrepreneurship Training Program of Hebei Province (S202310075048) and the Super-Computational-Center of Hebei University, Innovation Team Project of Hebei University (No. IT2023C01). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Competing interests disclosure
The authors have no competing interests or relevant affiliations with any organization or entity with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Writing disclosure
No writing assistance was utilized in the production of this manuscript.
