Abstract
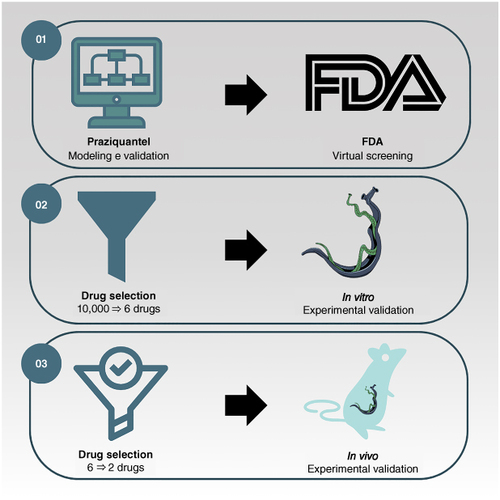
Aim: To identify potential antischistosomal agents through 3D pharmacophore-based virtual screening of US FDA approved drugs. Materials & methods: A comprehensive virtual screening was conducted on a dataset of 10,000 FDA approved drugs, employing praziquantel as a template. Promising candidates were selected and assessed for their impact on Schistosoma mansoni viability in vitro and in vivo using S. mansoni infected mice. Results & conclusion: Among the selected drugs, betamethasone and doxazosin demonstrated in vitro efficacy, with effective concentration 50% (EC50) values ranging from 35 to 60 μM. In vivo studies revealed significant (>50%) reductions in worm burden for both drugs. These findings suggest that betamethasone and doxazosin hold promise for repurposing in treating schistosomiasis. Additionally, the study showcases a useful approach for identifying new antischistosomal drugs.
TWEETABLE ABSTRACT
Discovering new treatments for #schistosomiasis is crucial![]() . Our study used virtual screening to identify potential antischistosomal drugs from US FDA approved compounds
. Our study used virtual screening to identify potential antischistosomal drugs from US FDA approved compounds ![]() . Promising results in vitro and in vivo.
. Promising results in vitro and in vivo. ![]() #drugdiscovery #tropicaldiseases
#drugdiscovery #tropicaldiseases
Models & molecules
Employed a 3D pharmacophore-based virtual screening strategy using praziquantel (PZQ) as a template.
Identified several pharmacophoric models and ranked database molecules based on QFIT values.
In vitro antiparasitic assays
Six drugs were selected for in vitro studies, with betamethasone and doxazosin showing notable activity against Schistosoma mansoni.
Sex-specific susceptibility observed, with male parasites more sensitive to betamethasone and doxazosin.
Betamethasone and doxazosin demonstrated delayed onset of action compared with PZQ.
Identified drugs exhibited activity at higher concentrations compared with PZQ, highlighting the need for refinement in virtual screening models.
In vivo studies in S. mansoni infected mice
Betamethasone and doxazosin tested in mice, resulting in dose-dependent reductions in worm burden.
Betamethasone and doxazosin at 400 mg/kg showed significant reduction in worm burden, comparable to untreated animals.
Potential of betamethasone and doxazosin as promising candidates for drug repositioning in schistosomiasis treatment.
Challenges in discovering potent antischistosomal agents within the current drug repertoire underscore the need for a targeted approach.
Introduces a relevant methodology for discovering novel antischistosomal agents among FDA approved drugs.
Acknowledgments
We are grateful for the invaluable support provided by MB Silva and RA Cajas for their dedicated efforts in maintaining the S. mansoni life cycle at the Research Center on Neglected Diseases, Guarulhos University (São Paulo, Brazil).
Author contributions
L Dobrachinsk, LLG Ferreira, AC Mafud, YP Mascarenhas, AD Andricopulo and J de Moraes were responsible for study conception and design; L obrachinski, LLG Ferreira, AC Mafud, ME Cirino, and AI Andrade-de-Siqueiraa were responsible for investigation and data analysis. L Dobrachinski, LLGF Ferreira, YP Mascarenhas, AD Andricopulo and Jd de Moraes were responsible for drafting and revision of the manuscript. J de Moraes was responsible for project administration and funding acquisition.
Financial disclosure
The work was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Brazil (Grants 13/07600-3, 13/25658-9, 14/02282-6, 16/18023-5, 16/22488-3) and partially financed by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, Finance Code 001) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Competing interests disclosure
The authors have no competing interests or relevant affiliations with any organization or entity with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Writing disclosure
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
The authors confirm obtaining institutional review board approval and ethical clearance from the Committee for Ethical Use of Animals in Experimentation of Guarulhos University (protocol ID 47/20), adhering to Brazilian legislation (Law 11.794/2008).