Abstract
Aim: Developing potent medicinal alternates for tuberculosis (TB) is highly desirable due to the advent of drug-resistant lethal TB strains. Methods & results: Novel indole-isoniazid integrates have been synthesized with promising antimycobacterial action against the H37Rv strain, and the nitro analogs 4e and 4j show the highest efficacy with a minimum inhibitory concentration of 1.25 μg/ml. The molecular docking studies against InhA support the experimental findings. Indole conjugates display remarkable radical quenching efficiency, and compounds 4e and 4j demonstrate maximum IC50 values of 50.19 and 52.45 μg/ml, respectively. Pharmacokinetic analysis anticipated appreciable druggability for the title compounds. Conclusion: The notable bioaction of the indole-isoniazid templates projects them as potential lead in developing anti-TB medications with synergetic antioxidant action.
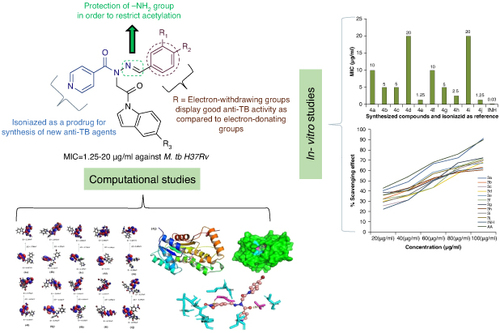
A series of novel N-(2-(1H-indol-1-yl)-2-oxoethyl)-N′-benzylideneisonicotinohydrazides were designed and synthesized.
The indole-isoniazid hybrids display promising in vitro antimycobacterial action. The compounds 4e and 4j demonstrate maximum inhibition potency, with a MIC value of 1.25 μg/ml.
The indole hybrids also demonstrate appreciable radical scavenging ability. The compounds 4e and 4j show maximum radical scavenging action with IC50 values of 50.19 and 52.45 μg/ml, respectively.
Molecular docking simulations validate the in vitro anti-TB findings through remarkable InhA inhibitory competency with a high binding energy. Compound 4j displayed the highest docking affinity in the series with two hydrogen bond interactions.
The DFT-based computational analysis of the synthesized entities revealed their apt biochemical characteristics supporting experimental and theoretical bioaction.
ADMET screening of the investigated compounds projects them as nearly safe anti-TB medication candidates.
With the synergetic antioxidant action, the novel indole-isoniazid analogs, especially 4e and 4j, may open fresh frontiers in developing potent anti-TB drugs.
Supplemental material
Supplemental data for this article can be accessed at https://doi.org/10.1080/17568919.2024.2379240
Acknowledgments
The authors gratefully acknowledge Maharshi Dayanand University, Rohtak, for the necessary facilities and support. The authors are highly thankful to the Department of Microbiology, Vallabhbhai Patel Chest Institute, University of Delhi, for anti-TB assay. The authors would like to facilitate SAIF, Punjab University, Chandigarh, for instrumental support. The authors, R Gavadia, J Rasgania and N Sahu are highly thankful for the research fellowship provided by CSIR and UGC, New Delhi.
Author contributions
K Jakhar designed the study. R Gavadia synthesized the title compounds. M Varma-Basil, V Chauhan and S Kumar performed the biological assays. R Gavadia, J Rasgania and N Sahu performed computational studies. R Gavadia and K Jakhar wrote the research article. S Mor and D Singh performed the review and editing of the research article. All the authors have read and agreed to publish the manuscript.
Financial disclosure
The authors have no financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Competing interests disclosure
The authors have no competing interests or relevant affiliations with any organization or entity with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Writing disclosure
No writing assistance was utilized in the production of this manuscript.