Abstract
A method based on a hazard analysis critical control point (HACCP)-based standard operating procedure (SOP) has been developed and implemented in community-based (CB) production of complementary foods (CFs) in agrarian regions in rural Ethiopia. The objective of this study was to validate the method in another setup, more specifically in cash crop (coffee and khat) producing communities in Southern Ethiopia. A quasi-experimental study was employed for the method validation. Two villages in the Gedeo zone, Southern Ethiopia were selected and a total of 40 mother-child pairs, 20 in each village, were involved in the study. Sociodemographic data, knowledge, and complementary feeding practices were collected using pretested questionnaires. The training was given to each mother for 1 h about community-based CFs production and implementation of the method. Then, 30 CF samples were collected and the level of aflatoxins (AFB1, AFB2, AFG1, and AFG2) was determined using a validated method. The majority (77.5%) of the mothers understand the importance of complementary feeding and give it to their infants beyond 6 months. Nearly two-thirds (62.5%) of the mothers had knowledge about the health impacts of aflatoxins, but the remaining 37.5% lacked awareness about the health risks of aflatoxins. Mothers were very well-motivated for the training and they promised to implement the knowledge and skill gained from the training for improving the nutritional status of their children. Overall, 23.3% of the CFs were contaminated by aflatoxins. AFB1 and AFG1 were detected in 6.7 and 16.7% of the CF samples, respectively. In cash crop-producing communities around the country, the HACCP-based SOP is easily validated in CB CFs production. Therefore, a scaleup of the method at the national level and beyond is recommended.
Introduction
Child undernutrition is a major public health burden that disproportionally affects children in low- and middle-income countries (LMIC), of which sub-Saharan Africa (SSA) and South Asia contribute the largest share of under-five children (UNICEF et al. Citation2020). In Ethiopia, 38% of children younger than five years are stunted, and the country has the largest burden of undernutrition in the region (CSA Citation2016). Inappropriate complementary foods (CFs) and feeding practices are linked to the high incidences of undernutrition among infants and young children in most LMICs including Ethiopia (Mollay et al. Citation2021).
The presence of aflatoxin in CFs may further contribute to poor child growth (Khlangwiset et al. Citation2011; Turner Citation2013; Makori et al. Citation2019; Anitha et al. Citation2020; Ismail et al. Citation2021). Aflatoxins contaminate staple crops that are potentially used in the CFs preparations in many of the countries where child stunting is also prevalent (Chala et al. Citation2014; Mutiga et al. Citation2015; Watson et al. Citation2017). Thus, minimizing and/or preventing the risks of aflatoxin contamination in CFs should be critical in addressing the burden of child undernutrition (Watson et al. Citation2017).
To prevent the risks of mold spoilage and aflatoxin contamination in the community-based (CB) CFs, food safety, and quality assurance standards need to be implemented. Hazard analysis critical control points (HACCP)-based standard operating procedure (SOP) is one of these guidelines which are being adopted globally to prevent CFs from the risks of mold spoilage and aflatoxin contamination (Udomkun et al. Citation2017; Ackah et al. Citation2018). Since its conception in the late 1950s, the HACCP system has grown to become the universally recognized and accepted method for food safety assurance (CAC Citation2009).
The utilization of HACCP as a food safety management tool has been growing in different countries including Ethiopia. As a result, a HACCP-based SOP was developed and implemented in an agrarian community in rural Ethiopia for community-based CFs production to protect CFs from the risks of aflatoxin contamination. This has improved the safety of the CFs and increased the nutritional status of young children (Unpublished work).
However, for national scale-up, validation of the HACCP-based SOP in another setup more specifically in a cash crop (coffee and khat) producing community is of great significance. Validation is very important to check whether the HACCP-based SOP will improve the safety of CFs when put into practice. More specifically, validation is required to ensure that the control measures put in place are effective to reduce or eliminate the risks of aflatoxin contamination in Cfs.
Therefore, this study aimed to validate the HACCP-based SOP for the production of CB CFs in cash crop-producing communities in Southern Ethiopia.
Materials and methods
Study area and design
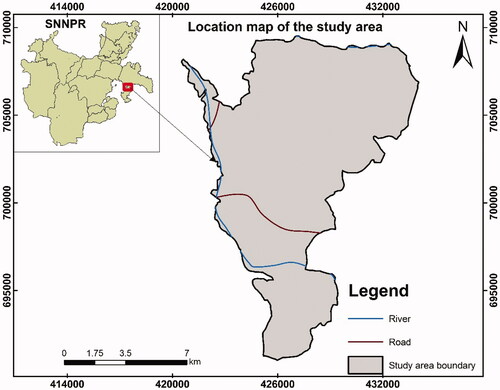
The research was carried out in the Dilla Zuria district, Gedeo zone, Southern Nations Nationalities, and Peoples (SNNP) region, in a cash crop (coffee and khat) producing community (). This study focused on two villages in the district. The research was carried out utilizing a quasi-experimental design. The sample size was calculated using Gpower software (version 3.1) considering; 95% confidence level (α—0.05), 90% power, 0.5 medium effect size and 10% non-response rate. As a result, a total of 40 mothers (with children of 6–23 months), 20 in each village, were included in the study.
Mothers’ sociodemographic characteristics, complementary feeding knowledge, and practices
Pretested questionnaires were used to collect sociodemographic data as well as complementary feeding knowledge and practices for the 40 mother-child pairs. The data were collected in April 2021.
Training on how to use the method
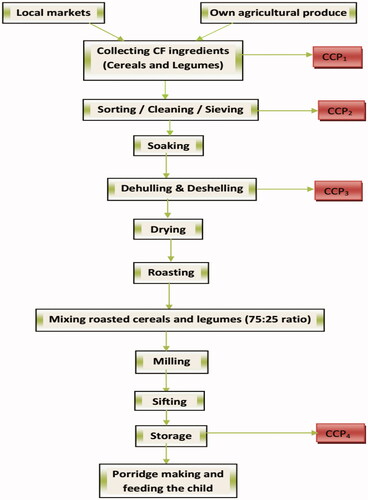
In each village’s health post, mothers, health extension workers (HEWs), and women's development army members (WDA) received 1 h of training. The training was mainly focused on the necessity of breastfeeding, complementary feeding, and home production of CFs using locally available cereals and legumes utilizing HACCP-based SOPs (), as well as hygiene and sanitation. Finally, the mothers were given advice on how to prepare CF ingredients.
CF ingredients collection
Manually cleaned, dehulled, dried, and then roasted (light roasting) CF ingredients (cereals—maize, barley, and legumes—haricot bean, broad bean, chick pea, and pea), about 300 g of each, were collected from 30 mothers’ (households). The samples were kept in polyethylene bags and kept together in a plastic box with lids. This way the samples were transported from the field to the Center for Food Science Nutrition, Addis Ababa University (AAU).
Starting the next day after arrival, the samples were taken out from the polyethylene bags and milled at the Center for Food Science and Nutrition Research Laboratory. The milling machine was cleaned using a blower and rinsed with 70% alcohol (ethanol) between batches of milling, and then sanitized by grinding some amount of the following sample to avoid cross-contamination. After that, CF flour (300 g each) was made with a ratio of 75:25 cereals and legumes using one cereal and one legume. The CF samples were collected in April 2021, a dry season in Ethiopia.
Moisture and aflatoxin analysis
Moisture contents of the CF flour samples were determined according to AOAC (Citation2000), using the official method 925.10. The levels of aflatoxins (AFB1, AFB2, AFG1, and AG2) in the CF flour samples were determined using an LC-MS/MS method (Ediage et al. Citation2015), validated according to the European Commission decision 2002/657/EC. The method consisted of extraction of a 5 g sample with 10 mL ultrapure water and 20 mL of dichloromethane. The extraction tubes were shaken for 45 min using a shaker and then centrifuged for 15 min at 3000 × g. Then 10 mL of filtrate was transferred into a glass tube and evaporated till dry at 40 °C using the TurboVap, followed by defatting in 0.5 mL hexane and analysis on an LC-MS/MS system.
A Quattro XEVO TQ-S LC-MS (Waters) was used for the analysis with a Zorbax Eclipse XDB C18-column (1.8 µm, 100 × 2.1 mm). The instrument settings were: polarity ESI+ (positive electrospray ionization); source and desolvation temperature 130 and 500 °C, respectively; capillary voltage 3.13 kV; cone and desolvation gas flow 150 and 1000 L/h, respectively. For positive identification of the target aflatoxins, the following criteria were met: (1) the deviations of the relative ion intensities of the multiple reaction monitoring (MRM) transitions were not greater than the maximum permitted tolerances (EC Citation2002), (2) the relative retention times with regard to the internal standard were below the maximum permitted deviation of 2.5%, (3) a peak with a signal-to-noise ratio of at least 3 was identified for each MRM transition.
Solvent gradient conditions
Mobile phase A: water/methanol/formic acid (94/5/0.1 v/v/v) + 5 mM ammonium acetate and mobile phase B: water/methanol/formic acid (5/94/0.1 v/v/v) + 5 mM ammonium acetate. The solvent gradient conditions at a flow rate of 0.4 mL/min were: 0–0.4 min, 100% A; 0.4–0.5 min, 100% A; 0.5–20 min, 1.0% A; 20–25 min, 1.0% A; 25–26 min, 100% A; 26–30 min, 100% A.
The injection volume was 10 µL. The limits of detection (LOD) and limit of quantitation (LOQ) are given in .
Table 1. Method validation results for the determination of aflatoxins using LC-MS/MS.
Ethical clearance
The Institutional Review Board (IRB) of the AAU's College of Natural and Computational Sciences granted ethical approval. The regional health bureau review board granted ethical permission, which was subsequently transmitted to the zonal and district health offices, as well as the HEWs for the various villages. The study's goal and methodology were explained to the mothers/caregivers in full, and then a signed agreement was obtained in the presence of local HEWs. Before the study, all questionnaires were translated into the local language, Amharic.
Data analysis
SPSS statistical software version 20 was used to analyse the data from the questionnaires. Using descriptive statistical approaches, the data was analysed, presented, and reported. When p-values were <0.05 (p < 0.05), the result was considered statistically significant.
Results and discussion
Mothers’ sociodemographic characteristics, complementary feeding knowledge, and practices
A total of 40 mothers of infants and young children (18 males and 22 females) 6–23 months of age were enrolled in this study. Of the infants and young children, 10% were aged 6–8 months, 10% were 9–11 months, and 80% were 12–23 months. All infants and young children enrolled in the current study were still being breastfed at the time of the survey. Nearly two-thirds (62.5%) of the mothers were between 25 and 34 years of age. About 60% of the mothers were illiterate, had no formal education, and almost all were married and protestant religion followers. Further, the majority (85%) of the mothers were housewives in occupation. The sociodemographic characteristics of the mothers and children are presented in .
Table 2. Description of sociodemographic characteristics of study participants in Dilla Zuria district, Southern Ethiopia, 2021.
An overview of the mothers’ knowledge and practices about complementary feeding and the risks of aflatoxin contamination is given in . The majority (77.5%) of the mothers knew the importance of complementary feeding and gave CFs to infants after 6 months of age. The most commonly used CFs were cereal-based porridge. Among cereals, maize and barley, and among legumes haricot bean, broad bean, and pea were predominately used for preparing CFs. All the CF ingredients (cereals and legumes) were purchased from the local market. The respondents were asked about the selection criteria of cereals and legumes for CFs preparation. The majority of the respondents (77.4%) use insect infestation, moldiness, color, odor, and type criteria for choosing cereals and legumes for CFs preparation.
Table 3. Respondents knowledge and practice about complementary feeding and the risks of aflatoxin contamination in Dilla Zuria district, Southern Ethiopia, 2021.
From the results displayed in , it can be seen that all the respondents use processing methods, such as cleaning or sorting, dehulling, drying, roasting, and milling while preparing CFs at their homes. Our results are consistent with a previous study by Matumba et al. (Citation2015) reported that hand sorting, dehulling, and combinations thereof assisted in removing naturally occurring aflatoxins in white maize, a commonly used cereal for CFs preparation.
This study shows that nearly half (47.5%) of the respondents observed mold in food or food ingredients used for child or family. Respondents were also asked how they deal with mold-contaminated food stuffs. Nearly all (94.7%) of the respondents reported that they discarded moldy foodstuffs, however, a few respondents consumed moldy foods after roasting. This study is consistent with a previous in Ethiopia by Beyene et al. (Citation2016), who reported that 34% of the respondents discarded material when they observed mold in foods prepared for a child or family.
Responding to knowledge questions related to the health impacts of aflatoxins, nearly two-thirds (62.5%) of the respondents knew about the health impacts of consuming mold-spoiled and aflatoxin-contaminated foods. About 68% of the respondents reported that diarrhea was one of the health impacts of mold-spoiled and aflatoxin-contaminated food. Further, 32% of the respondents reported that mold-spoiled and aflatoxin-contaminated foods are associated with vomiting.
Even though nearly two-thirds of the respondents knew the health impacts of aflatoxins, the other 37.5% lacked an understanding of the possible health risks of aflatoxin-contaminated food consumption. However, our results are consistent with a previous study in Southwest Ethiopia by Bereka et al. (Citation2021) who reported that aflatoxin-contaminated maize was used as human food, indicating that respondents were not aware of the health risks of aflatoxins. A similar study in Ethiopia by Beyene et al. (Citation2016), also reported that mothers lack awareness about the health impacts of aflatoxin-contaminated foods. Another study in Southern Ethiopia also reported that the knowledge towards the health risks of aflatoxin among lactating women was suboptimal (Boshe et al. Citation2020). Another study in Nigeria reported that 85% of consumers lacked awareness of the health risks of eating aflatoxin-contaminated foods (Ezekiel et al. Citation2013). Another study in Malawi also reported that the public is not well-informed of the health implications associated with mold-spoiled foods (Matumba et al. Citation2016).
Training on how to use the method
The training mainly focused on the importance of optimal breastfeeding and complementary feeding practices, which are important contributors to child survival and nutritional status during the first two critical years of life. Complementary feeding occurs between 6 and 23 months of age, which is the largest part of the 1000 days viewed as the key window of opportunity for preventing under nutrition and its long-term adverse consequences. The WHO guiding principles for complementary feeding of the breastfed child were introduced briefly (Dewey Citation2003). Complementary feeding behaviour, such as the amount of food, feeding frequency, food consistency/energy density, food safety and hygiene, responsive feeding, and feeding during and after the illness was also introduced.
The training also highlighted community-based CFs preparation using locally available cereals and legumes by implementing the HACCP-based SOP for reducing the risks of aflatoxin and microbial contamination. During the training, the HACCP concept was described in simple terms and important steps were indicated as critical control points (CCPs) in the CFs flow diagram so that mothers can easily use and prepare CFs without any difficulty.
Further, mothers were also informed about the challenges young children face during CFs introduction. While introducing the child to CFs, the prevalence of malnutrition increases substantially in many LMICs including Ethiopia, because of an increase in infections and poor feeding practices. In Ethiopia, dehydration from diarrhea is a major cause of death in infancy and childhood and 12% of children under five were reported to have had diarrhea according to the 2016 Ethiopian demographic and health survey report (CSA Citation2016). During the training, the health impacts of diarrhea were highly emphasized. As a result, mothers clearly understood the health impacts of diarrhea among young children, and they promised to prevent diarrhea in their children.
Overall, mothers were very well-engaged in the training and they promised to implement the knowledge and skill gained from the training for improving undernutrition, health, and well-being of their children. HEWs and WDA in the respective villages also promised to work together on improving undernutrition among young children in their localities. Our study was supported by a recent study in Ethiopia by Hirvonen et al. (Citation2021), who reported that improving diet quality and safety through improved infant and young child feeding (IYCF) knowledge and implementation of infection prevention and control measures could help prevent child undernutrition.
The occurrence of aflatoxin in complementary foods
The complementary flour samples obtained were contaminated by aflatoxins (AFB1, AFB2, AFG1, and AFG2) at varying occurrences and levels. shows the distribution of AFB1, AFB2, AFG1, and AFG2 in the complementary flour samples. Overall, 23.3% of the complementary flour samples were contaminated by aflatoxins. Among the aflatoxins, AFB1 was detected in 6.7% of the complementary flour with concentrations ranging from 0.22 to 0.52 µg/kg, while AFG1 was detected in 16.7% of the complementary flour samples with concentrations ranging from 0.02 to 0.13 µg/kg.
Table 4. The level of aflatoxins in complementary flours collected from households across two villages in Dilla Zuria district, Southern Ethiopia, 2021.
In this study, 6.7% of the complementary flour samples were contaminated by AFB1 at concentrations exceeding the European Commission's maximum level of 0.1 µg/kg allowed in processed cereal-based foods for infants and young children (EU Citation2006). Even though a few samples were contaminated by AFB1 in this study at concentrations above the maximum level set by the EU, the levels were much lower than in a previous study in Ethiopia by Eshete et al. (Citation2021), who reported that 95% of the complementary flour was contaminated by AFB1 above the EU maximum level. Another study in Ethiopia by Ayelign et al. (Citation2018) also reported that 95% of cereal-based CFs were contaminated by aflatoxins.
In contrast to our study results, high aflatoxin contamination of CFs has been reported in sub-Saharan Africa. A study by Ojuri et al. (Citation2018) in Nigeria reported that 42% of cereal-based CFs were contaminated by aflatoxins. Another study in Nigeria also reported that AFB1 was detected in 67.9 and 34.5% of household processed CFs and industrially produced CFs, respectively (Ojuri et al. Citation2019). A study in Ghana reported that 71% of cereal-based CFs intended for infants and young children were contaminated by aflatoxins (Blankson and Mill-Robertson Citation2016). Another study in Tanzania also reported that AFB1 was detected in 42.5% of the complementary flour with levels ranging from 0.25 to 2128 µg/kg (Makori et al. Citation2019).
As compared to previous studies in Ethiopia and beyond, the lower occurrences of aflatoxins in the CFs may be due to the better agricultural, storage, and hygienic practices implemented in the study villages. Further, it might be due to the fact that mothers have implemented properly the knowledge gained during HACCP-based SOP training while preparing complementary food ingredients. Moreover, it is critical to protect CFs and their ingredients from aflatoxin contamination to protect young children from health risks, such as growth retardation (Turner et al. Citation2007; Makori et al. Citation2019; Andrews-Trevino et al. Citation2021; Ismail et al. Citation2021), immune suppression (Ismail et al. Citation2021) and hepatocellular carcinoma (HCC) (liver cancer) (Wild and Gong Citation2010; Gong et al. Citation2016). Some of the strategies used to control the risks of aflatoxin contamination in CFs and their ingredients include good agricultural practices (preharvest practices, such as proper plant nutrition, avoiding drought stress, and others), good post-harvest practices, good manufacturing practices, awareness creation, and others (Neme and Mohammed Citation2017; Ayelign and De Saeger Citation2020).
Conclusions
This study found that mothers can readily put the HACCP-based SOP method into practice while preparing CFs at home. Furthermore, when properly implemented, the method can reduce and/or eliminate the risks of aflatoxin contamination for CFs. As a result, it is suggested that the method be scaled up on a national level.
Acknowledgements
The authors greatly acknowledge mothers/caregivers of the young children in all villages for giving their time and responding to the questionnaires. The authors’ special gratitude also goes to health extension workers (HEWs) in all villages for their technical support during the sample collection.
Disclosure statement
No potential conflict of interest was reported by the authors.
Correction Statement
This article has been republished with minor changes. These changes do not impact the academic content of the article.
Additional information
Funding
References
- Ackah NB, Baidoo EA, Appiah AHK. 2018. Validating a HACCP system for the production of vegetable Shito. J Food Qual. 2018:1–7. doi:https://doi.org/10.1155/2018/7146040
- Andrews-Trevino JY, Webb P, Shively G, Kablan A, Baral K, Davis D, Paudel K, Shrestha R, Pokharel A, Acharya S, et al. 2021. Aflatoxin exposure and child nutrition: measuring anthropometric and long-bone growth over time in Nepal. Am J Clin Nutr. 113(4):874–883. doi:https://doi.org/10.1093/ajcn/nqaa397
- Anitha S, Muzanila Y, Tsusaka TW, Kachulu L, Kumwenda N, Musoke M, Swai E, Shija J, Siambi M, Monyo ES, et al. 2020. Reducing child undernutrition through dietary diversification, reduced aflatoxin exposure, and improved hygiene practices: the immediate impacts in central Tanzania. Ecol Food Nutr. 59(3):243–262. doi:https://doi.org/10.1080/03670244.2019.1691000
- AOAC. 2000. Association of official analytical chemists. Official methods of analysis. Vol. II 17. Washington (DC): AOAC International.
- Ayelign A, De Saeger S. 2020. Mycotoxins in Ethiopia: current status, implications to food safety and mitigation strategies. Food Control. 113:107163. doi:https://doi.org/10.1016/j.foodcont.2020.107163
- Ayelign A, Woldegiorgis AZ, Adish A, De Saeger S. 2018. Total aflatoxins in complementary foods produced at community levels using locally available ingredients in Ethiopia. Food Addit Contam Part B Surveill. 11(2):111–118. doi:https://doi.org/10.1080/19393210.2018.1437784
- Bereka TY, Kuyu CG, Tolera KD, Addis EM. 2021. Current postharvest practices and aflatoxin contamination awareness amongst maize producers in Jimma Zone, Southwest of Ethiopia. World Mycotoxin J. 115:35–43. doi:https://doi.org/10.3920/WMJ/2020.2642
- Beyene AA, Woldegiorgis AZ, Adish AA, De Saeger S, Tolossa AL. 2016. Assessment of mothers’ knowledge and practice towards aflatoxin contamination in complementary foods in Ethiopia: from pre-harvest to household. World Mycotoxin J. 9(4):535–544. doi:https://doi.org/10.3920/WMJ2016.2088
- Blankson GK, Mill-Robertson FC. 2016. Aflatoxin contamination and exposure in processed cereal-based complementary foods for infants and young children in greater Accra, Ghana. Food Control. 64:212–217. doi:https://doi.org/10.1016/j.foodcont.2015.12.032
- Boshe B, Gebremedhin S, Alemayehu F, Eshete M, Taye M, Stoecker BJ. 2020. Aflatoxin exposure among lactating women in southern Ethiopia. Food Sci Nutr. 8(12):6738–6745. doi:https://doi.org/10.1002/fsn3.1968
- [CSA] Central Statistical Agency. 2016. Ethiopia demographic and health survey 2016. Addis Ababa; Rockville (MD): CSA and ICF.
- Chala A, Taye W, Ayalew A, Krska R, Sulyok M, Logrieco A. 2014. Multimycotoxin analysis of sorghum (Sorghum bicolor L. Moench) and finger millet (Eleusine coracana L. Garten) from Ethiopia. Food Control. 45:29–35. doi:https://doi.org/10.1016/j.foodcont.2014.04.018
- [CAC] Codex Alimentarius Commission. 2009. Food hygiene. Basic text 4th ed. Rome: World Health Organization Food and Agriculture Organization of the United Nations.
- Dewey K. 2003. Guiding principles for complementary feeding of the breastfed child.
- [EC] European Commission. 2002. Commission Decision 2002/657/EC of 12 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. Off J Eur Union. L221:8–36. doi:2002/657/EC
- Ediage EN, Van Poucke C, De Saeger S. 2015. A multi-analyte LC–MS/MS method for the analysis of 23 mycotoxins in different sorghum varieties: the forgotten sample matrix. Food Chem. 177:397–404. doi:https://doi.org/10.1016/j.foodchem.2015.01.060
- Eshete M, Gebremedhin S, Alemayehu FR, Taye M, Boshe B, Stoecker BJ. 2021. Aflatoxin contamination of human breast milk and complementary foods in southern Ethiopia. Matern Child Nutr. 17(1):e13081. doi:https://doi.org/10.1111/mcn.13081
- European Union 2006. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs (Text with EEA relevance).
- Ezekiel CN, Sulyok M, Babalola DA, Warth B, Ezekiel VC, Krska R. 2013. Incidence and consumer awareness of toxigenic Aspergillus section Flavi and aflatoxin B1 in peanut cake from Nigeria. Food Control. 30(2):596–601. doi:https://doi.org/10.1016/j.foodcont.2012.07.048
- Gong YY, Watson S, Routledge MN. 2016. Aflatoxin exposure and associated human health effects, a review of epidemiological studies. Food Saf. 4(1):14–27. doi:https://doi.org/10.14252/foodsafetyfscj.2015026
- Hirvonen K, Wolle A, Laillou A, Vinci V, Chitekwe S, Baye K. 2021. Child growth faltering dynamics in food insecure districts in rural Ethiopia. Matern Child Nutr. e13262. doi:https://doi.org/10.1111/mcn.13262
- Ismail A, Naeem I, Gong YY, Routledge MN, Akhtar S, Riaz M, Ramalho LNZ, de Oliveira CAF, Ismail Z. 2021. Early life exposure to dietary aflatoxins, health impact and control perspectives: a review. Trends Food Sci Technol. 112:212–224. doi:https://doi.org/10.1016/j.tifs.2021.04.002
- Khlangwiset P, Shephard GS, Wu F. 2011. Aflatoxins and growth impairment: a review. Crit Rev Toxicol. 41(9):740–755. doi:https://doi.org/10.3109/10408444.2011.575766
- Makori N, Matemu A, Kimanya M, Kassim N. 2019. Inadequate management of complementary foods contributes to the risk of aflatoxin exposure and low nutrition status among children. World Mycotoxin J. 12(1):67–76. doi:https://doi.org/10.3920/WMJ2018.2354
- Matumba L, Monjerezi M, Kankwamba H, Njoroge SMC, Ndilowe P, Kabuli H, Kambewa D, Njapau H. 2016. Knowledge, attitude, and practices concerning presence of molds in foods among members of the general public in Malawi. Mycotoxin Res. 32(1):27–36.doi:https://doi.org/10.1007/s12550-014-0237-3
- Matumba L, Van Poucke C, Njumbe Ediage E, Jacobs B, De Saeger S. 2015. Effectiveness of hand sorting, flotation/washing, dehulling and combinations thereof on the decontamination of mycotoxin-contaminated white maize. Food Addit Contam Part A. 32(6):960–969. doi:https://doi.org/10.1080/19440049.2015.1029535
- Mollay C, Kassim N, Stoltzfus R, Kimanya M. 2021. Complementary feeding in Kongwa, Tanzania: findings to inform a mycotoxin mitigation trial. Matern Child Nutr. 17(4):e13188. doi:https://doi.org/10.1111/mcn.13188
- Mutiga SK, Hoffmann V, Harvey JW, Milgroom MG, Nelson RJ. 2015. Assessment of aflatoxin and fumonisin contamination of maize in western Kenya. Phytopathology. 105(9):1250–1261. doi:https://doi.org/10.1094/PHYTO-10-14-0269-R
- Neme K, Mohammed A. 2017. Mycotoxin occurrence in grains and the role of postharvest management as a mitigation strategies. A review. Food Control. 78:412–425. doi:https://doi.org/10.1016/j.foodcont.2017.03.012
- Ojuri OT, Ezekiel CN, Eskola MK, Šarkanj B, Babalola AD, Sulyok M, Hajšlová J, Elliott CT, Krska R. 2019. Mycotoxin co-exposures in infants and young children consuming household-and industrially-processed complementary foods in Nigeria and risk management advice. Food Control. 98:312–322. doi:https://doi.org/10.1016/j.foodcont.2018.11.049
- Ojuri OT, Ezekiel CN, Sulyok M, Ezeokoli OT, Oyedele OA, Ayeni KI, Eskola MK, Šarkanj B, Hajšlová J, Adeleke RA, et al. 2018. Assessing the mycotoxicological risk from consumption of complementary foods by infants and young children in Nigeria. Food Chem Toxicol. 121:37–50. doi:https://doi.org/10.1016/j.fct.2018.08.025
- Turner PC. 2013. The molecular epidemiology of chronic aflatoxin driven impaired child growth. Scientifica. 2013:152879–152821. doi:https://doi.org/10.1155/2013/152879
- Turner PC, Collinson AC, Cheung YB, Gong Y, Hall AJ, Prentice AM, Wild CP. 2007. Aflatoxin exposure in utero causes growth faltering in Gambian infants. Int J Epidemiol. 36(5):1119–1125. doi:https://doi.org/10.1093/ije/dym122
- Udomkun P, Wiredu AN, Nagle M, Bandyopadhyay R, Müller J, Vanlauwe B. 2017. Mycotoxins in Sub-Saharan Africa: present situation, socio-economic impact, awareness, and outlook. Food Control. 72:110–122. doi:https://doi.org/10.1016/j.foodcont.2016.07.039
- UNICEF, WHO, World Bank. 2020. Joint child malnutrition estimates. Key findings of the 2020 edition. New York (NY); Geneva; Washington (DC): WHO.
- Watson S, Gong YY, Routledge M. 2017. Interventions targeting child undernutrition in developing countries may be undermined by dietary exposure to aflatoxin. Crit Rev Food Sci Nutr. 57(9):1963–1975. doi:https://doi.org/10.1080/10408398.2015.1040869
- Wild CP, Gong YY. 2010. Mycotoxins and human disease: a largely ignored global health issue. Carcinogenesis. 31(1):71–82. doi:https://doi.org/10.1093/carcin/bgp264