 ?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.ABSTRACT
The aim of this study was to analyse the effect of fat replacement by oat β-glucan concentrates on the physicochemical properties of beef burgers. The goal was to obtain a low-fat product accepted by consumers, having physicochemical parameters similar to the control sample, composed of β-glucan at least by 1%. This evaluation was based on basic composition, β-glucan and cholesterol content, colour and texture analysis, pH value, WHC, volatile compounds profile and sensory properties of burgers. Burgers with β-glucan concentrates had lower cholesterol content and higher WHC and texture (hardness, cohesiveness and springiness increase) parameters than control burgers. The highest overall acceptance scores among the samples with β-glucan were recorded for the sample with β-glucan 30% concentrate in amount of 4%. This modification could allow the substitution of traditional beef burgers with a healthier low-fat meat product with similar customer appeal.
RESUMEN
El presente estudio se propuso analizar el efecto que provoca la sustitución de la grasa por concentrados de β-glucano de avena en las propiedades fisicoquímicas de las hamburguesas de carne de res. El objetivo era obtener un producto de bajo contenido graso, compuesto por β-glucano al menos en un 1%, cuyos parámetros fisicoquímicos fueran similares a los de la muestra de control y que, a la vez, fuera aceptado por los consumidores. En este sentido, la evaluación atendió la composición básica, el contenido de β-glucano y de colesterol, el análisis de color y textura, el valor de pH, WHC, el perfil de compuestos volátiles y las propiedades sensoriales de las hamburguesas. Las hamburguesas con concentrados de β-glucano mostraron un contenido de colesterol más bajo y parámetros de WHC y textura más altos (aumento de dureza, cohesión y elasticidad) que las hamburguesas de control. Entre las muestras de hamburguesas con β-glucano los mayores puntajes de aceptación general se registraron en la muestra que contenía 4% de concentrado de β-glucano al 30%. Esta modificación podría permitir sustituir las tradicionales hamburguesas de carne de res por un producto cárnico de bajo contenido graso, más saludable y con un atractivo similar para el consumidor.
1. Introduction
Beef burgers are one of the most popular meat products in the world. Their popularity among consumers is mainly due to their easy accessibility, low price and the desirability of fast foods across the world (Gordon-Larsen et al., Citation2011). Unfortunately, overweight and obese are a common problem among a wide range of burger consumers. Prevalence of diabetes, heart disease, overweight, obesity and other diet-related diseases is increasing worldwide. Thus, the development of new alternatives for these diseases management is significant (Bozbulut & Sanlier, Citation2019; Ho et al., Citation2016). Luckily, in recent years, consumers have paid more attention to health and nutrition. As a consequence, the meat industry, like other food sectors, has undertaken steps to meet consumer expectations (Jiménez-Colmenero, Citation2007; Soncu et al., Citation2015). Consumers are increasingly turning to functional products with reduced fat content, but reduced fat content in food products is not easy to obtain. Fats are responsible for the physical properties of food (such as texture, colour, rheological parameters and taste), as well as for delivering fat-soluble vitamins (Brewer, Citation2012; Drewnowski & Almiron-Roig, Citation2010; Ognean et al., Citation2006; Ravisankar et al., Citation2015). Hence, reduction of fat content from meat products may have adverse effect on their juiciness, flavour and overall acceptance after consumption (Ktari et al., Citation2016).
One way to reduce total fat intake is to replace the fats in meat products with substances obtained from plants, among which are different types of cereal dietary fibres. One such fibre is oat β-glucan concentrate (Piñero et al., Citation2008). β-glucan (β-(1-3),(1-4)-D-glucan) is a soluble fraction of dietary fiber obtained from oat with special positive nutritional, functional and technological properties. The intake of fiber and especially β-glucans becomes an important alternative for diabetics and for people with high cholesterol levels (Bozbulut & Sanlier, Citation2019; Shanmugam & Priya, Citation2016). In most cases, the addition of β-glucan concentrates is used in the production of cereal and bakery products, but as of recently they can also be found in dairy products, pasta, soups and meat products (for example meat batters, sausages and chopped poultry products) (Ahmad et al., Citation2012; Álvarez & Barbut, Citation2013; Choi et al., Citation2011; Ktari et al., Citation2016; Kurek et al., Citation2016; Piñero et al., Citation2008). In addition to its documented biological activity, oat β-glucan also has hydrocolloid characteristics, which are important with regard to food processing. Hydrocolloids have water-binding abilities, increasing its viscosity, stabilising emulsion and modifying the gelation process (Lee et al., Citation2015; Pintado et al., Citation2016). β-glucan forms porous network held between protein–gel matrix and the fat globules in the meat product system. This process explains the absorption of large water amounts and the formation of dense matrices which play a key role in textural and rheological parameters of products (Morin et al., Citation2004). Consequently, due to its stability and gelation properties and water-binding capacity, β-glucan can be a factor in shaping the textural and rheological features of low-fat food products (Da Oliveira et al., Citation2012; Piñero et al., Citation2008). In addition to its technological properties, oat β-glucan has a positive influence on human health. There are many sources of β-glucan, but only in the case of oat and barley β-glucan EFSA expressed its positive recommendations (EFSA, Citation2010, Citation2011). Among its most important health benefits and nutritional value, oat β-glucan has the ability to lower blood LDL cholesterol levels, provide support in the treatment of obesity, have an impact on the treatment of inflammatory bowel and gastric mucosa, and alleviate the effects of peptic ulcer (Chang et al., Citation2013; Daou & Zhang, Citation2012; Fulgoni et al., Citation2015; Ho et al., Citation2016; O’Neil et al., Citation2015; Othman et al., Citation2011; Shanmugam & Priya, Citation2016).
The aim of this study was to analyse the influence of fat replacement by oat β-glucan concentrates extracted using hydrothermal extraction method (without using chemicals) on the physicochemical properties of beef burgers. Oat grains are used to produce β-glucan concentrate. The first step is to remove the flower husk. The second stage, as in the production of oatmeal porridge, is the process of hydrothermal stabilization. The next stages consist in reducing the amount of carbohydrates, protein and fat in such a way that the final product retains the possibility of their synergic effect. The only medium in direct contact with oat grain is water vapor with enthalpy depending on the production stage and the species of oat. No enzymes, solvents or other active chemicals are used. The goal of the experiment was to obtain a low-fat product acceptable to consumers, with physicochemical parameters similar to the control sample, which are composed of β-glucan by at least 1%. The final product should be able to adhere to the EFSA health claims: ‘β-glucans contribute to the maintenance of normal blood cholesterol levels’. The claim can be used only for food which contains at least 1 g of beta-glucans from oats, oat bran, barley, barley bran, or from mixtures of these sources per quantified portion. In order to bear the claim information shall be given to the consumer that the beneficial effect is obtained with a daily intake of 3 g of beta-glucans from oats, oat bran, barley, barley bran, or from mixtures of these beta-glucans. And ‘the consumption of β-glucans from oats or barley as part of a meal contributes to the reduction of blood glucose rise after a meal’ – The claim can be used only for food which contains at least 4 g of beta-glucans from oats or barley for each 30 g of available carbohydrates in a quantified portion as part of the meal. In order to bear the claim, information shall be given to the consumer that the beneficial effect is obtained by consuming the beta-glucans from oats or barley as part of the meal (Citationn.d.). Consequently, the meat products contain at least 1% β-glucans of beta-glucans from oats meet both of the above conditions.
2. Materials and methods
2.1. Preparation of beef burgers
Beef chucks (source: Meat Plant Stanislawow Ltd, Poland) without connective tissue and excessive fat were chopped and minced in a grinder [Ø 8 mm plate] (Edesa PI-22-TU-T, Spain). Beef burger was prepared for five different recipes. The composition of control and low-fat burgers with added β-glucan concentrates is presented in . Two (15% and 30%) β-glucan concentrates extracted using hydrothermal extraction method (without using chemicals) from dehulled oat grain Avena Sativa were used (Microstructure Inc., Poland). Compositions of both β-glucan concentrates are presented in . β-glucan concentrates were applied in different doses: 8% and 12% of 15% oat β-glucan concentrate and 4% and 6% of 30% oat β-glucan concentrate. During preparation, all ingredients were mixed. Samples were formed using an aluminium-forming press (diameter: 100 mm). Each burger weighed 150 g. Samples were placed on polyethylene terephthalate/polyethylene (PET/PE) trays 187 × 137 × 50 mm with an absorbent pad (absorbency grades 1,700 mL/m2) and 44 μm thick polyethylene terephthalate/cast polypropylene (PET/CPP) with maximum oxygen permeability not exceeding 10 cm3/m2/24 h/bar at 23°C, with a relative humidity of 0% (EC04, Corenso, Finland). An M3 packaging machine (Sealpack, Germany) was used to pack the burgers in a modified atmosphere (MAP – 30/70:CO2/O2). Samples were stored for 0, 4, 7 and 11 days at 2 ± 1°C. Analysis was carried out immediately after packing (0 day – the samples were packed and immediately unpacked and analysed on the same day as the samples were prepared) and after 4, 7 and 11 days of storage.
Table 1. Proportion of ingredients used in prepared beef burgers with different fat replacement levels by oat β-glucan concentrates. Treatments: control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Tabla 1. Proporción de ingredientes utilizados en las hamburguesas de carne de res preparadas con diferentes niveles de sustitución de la grasa por concentrados de β-glucano de avena. Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%
Table 2. Composition of 15% and 30% β-glucan concentrates from dehulled oat grain Avena Sativa was used (Microstructure Inc., Poland).
Tabla 2. Composición de concentrados de β-glucano al 15% y 30% hechos del grano de avena descascarado. Avena Sativa (Microstructure Inc., Polonia)
Prior to the texture profile analysis (TPA), analysis of volatile compounds and consumer acceptance evaluation, samples were heat treated by grill in standard condition (electrical grill Silex S161GR S-Tronic Single Grill, Germany), and pre-heated using top and bottom heating plates respectively 210°C and 190°C. The burgers were grilled to reach 75°C at their centre of the sample (Ellab, TrackSense® Pro, Denmark).
2.2. Near-infrared spectroscopy analysis (NIR)
The basic composition of beef burgers (water, protein, fat and ash content) was determined by accredited FT-NIR near-infrared laboratory (Polish Centre for Accreditation – Accreditation No. AB 1670) according to the method presented in the work of Szpicer et al. (Citation2019). Büchi near-infrared spectrometer NIRFlex N-500 (Büchi Labortechnik AG, Switzerland) with NIRFlex Solids module in the spectral range of 12.500–4.000 cm1 in reflectance mode and application Büchi Art. N. N555-501 was used. Meat portions of 100 g were homogenised and placed on a Petri dish. NIR analysis was taken at the beginning of the experiment (on day 0) (in six repetitions for each sample).
2.3. β-glucan content
The β-glucan content in raw beef burgers on day 0, 4, 7 and 11 of storage was measured using the enzymatic method with the β-glucan Assay Kit (Mixed Linkage). The samples (200 mg) were mixed with aqueous ethanol (50% v/v) and incubated in a boiling water bath for 5 min. Samples were mixed on a vortex stirrer (IKA MS3 basic, Wilmington, USA), after which 5.0 mL of 50% (v/v) aqueous ethanol was added to the meat. The next steps with lichenase, β-glucosidase, GOPOD Reagent (glucose oxidase/peroxidase) were carried out to the manufacturer’s instructions (Megazyme, Bray, Ireland). Absorbance was measured at 510 nm (Tecan Spark™ 10M, Switzerland). β-glucan content is calculated according to EquationEquation (1)(1)
(1)
where:
2.4. Instrumental colour analysis
The surface colour of raw burgers was measured in a CIEL*a*b* system using a Minolta CR-400 chromameter (Konica Minolta Inc., Japan) according to the method presented in the work of Moczkowska et al. (Citation2015). The parameters measured were L* (L* = 0/black and L* = 100/white), a* (–a* = green and +a* = red) and b* (–b* = blue and +b* = yellow). The diameter of the measuring head was 8 mm, a D65 (colour temperature: 6.500 K) illuminant and a standard 2° observer were used. The colourimeter was calibrated using a standard white Minolta calibration plate (L* = 98.45; a* = −0.10; b* = −0.13). Colour parameters were measured in 15 different places on the surface of each burger sample, on each measurement day, immediately after opening the package.
2.5. Texture profile analysis (TPA)
Texture profile analysis (TPA) of the grilled beef burgers was conducted in a double compression cycle using the Instron 5965 Universal Testing Machine (Instron, USA) with a cell capacity of 500 N with Bluehill®2 software, according to the procedure described by Szpicer et al. (Citation2018). Cylinder-shaped samples 2.45 cm in diameter and 2.50 cm in height were cut from the centre of the burgers and doubly compressed to the point of 50% reduction of their initial height, with a 3 s of relaxation time. The test was conducted 6 times at a constant head speed of 200 mm∙min−1 at 4 ± 1°C. Textural parameters, i.e. hardness (N) (maximum force of the first compression), cohesiveness (proportion of the area under the curve from the second compression to the area under the curve from the first compression), gumminess (N) (hardness × cohesiveness) and chewiness (N) (hardness × cohesiveness × springiness), were calculated according to the procedure described by Półtorak et al. (Citation2014).
2.6. pH value
The samples’ pH values were measured at day 0, 4, 7 and 11 of storage using a hand-held pH electrode, TESTO 205 (Germany). The electrode was calibrated using buffers (pH 4.01 and 7.00) prior to measurement. Each sample was measured in six repetitions.
2.7. Water-holding capacity
The water-holding capacity of the raw beef burger samples was evaluated on day 0, 4, 7 and 11 of storage in accordance with the method used in Gheisari et al. (Citation2009), with certain modifications. Samples of meat (0.3 g) were placed on a Whatman filter paper no. 1 between two glass plates and weighed down with a 2 kg weight for exactly 5 min. Press stains were photographed using OImaging MicroPublisher 5.0 RTV (Canada) with a Kaiser system (Germany). Areas of liquid (Al) and meat (Am) were evaluated using the Image-Pro Plus (v.7.0) software. Water-holding capacity is calculated according to EquationEquation (2)(2)
(2) .
2.8. Cholesterol content
The cholesterol content of raw burgers was measured on day 0, 4, 7 and 11 of storage in accordance with a modified Pearsos et al. (Citation1953) method. Homogenised burger samples (Mm) (0.5 g) were extracted with cyclohexane (5 mL) and centrifuged for 10 min at 2,700 g, at 23°C. 0.25 mL of Supernatant (0.25mL) was dispensed into the bottom of the test tube and 0.25 mL distilled water was dispensed to blank. One milliliter of 12% p-toluenesulfonic acid and 3 mL of acetic anhydride were added to all tubes. The samples were left to equilibrate at room temperature, after which 0.5 mL of sulphuric acid was added. After 20 min absorbance was measured at 510 nm (Tecan Spark™ 10 M, Männedorf, Switzerland). Cholesterol standards in acetic acid at 0.2, 0.4, 0.8 and 1.6 mg/mL concentration were used as reference solutions. A standard curve was generated and slope (a) was calculated. Cholesterol content was calculated using the following EquationEquation (3)(3)
(3) :
where:
2.9. Volatile compounds profile
Volatile compounds in both raw and grilled burgers on day 0 of storage were analysed using a Heracles II electronic nose (Alpha M.O.S., Toulouse, France) based on ultrafast gas chromatography, according to the method presented by Wojtasik-Kalinowska et al. (Citation2017). Prepared samples were incubated at 55°C for 900 s under 8.33 Hz agitation with the temperature of injector set to 200°C, injected volume to 3,500 µl and injection speed to 125 mL/s. The samples were analysed in five repetitions. Principal component analysis (PCA) was used for data processing using the Alpha Soft software (v.8.0).
2.10. Consumer evaluation
Consumer evaluation of the beef burgers was performed by 240 untrained panelists recruited among students from the Warsaw University of Life Sciences campus (120 male and 120 female), according to the Meat Standards Australia [MSA] (Citation2008) with modifications. The age of the panelists ranged from 19 to 25 years. Sessions of consumer evaluation were conducted in an isolated room, with white light at 23 ± 1°C room temperature. Burgers were given randomly on coded (3-digit) plastic plates (1 link sample + 5 samples per consumer). Each sample was evaluated by the consumer for 8 min. Before five test samples, first link sample was served to all consumers. Allocation of samples to consumers using Latin square design (5 × 5) completely balanced effects of carryover and position. Data about the degree of likeness of the samples were received through a questionnaire. Panelists were questionnaire to express their opinion on an unstructured scale (100 mm) by placing a vertical line on it. Seven sensory attributes were evaluated: external appearance, colour, aroma, taste, juiciness, texture and overall acceptability. Consumers were given breaks (0.5 min) between testing each sample, during which they were instructed to eat bread and rinse their mouth. Quantification of the results was obtained by measuring the distance from zero to the vertical line. The results from the unstructured scale were converted to numerical values (ranging between 0 and 10), where 0 denoted the lowest and 10 the highest level of likeness. Variance due to session number and panellist was considered for the sensory data.
2.11. Statistical analysis
The statistical analysis of the data was carried out using Statistica 13.3 (StatSoft Inc., Tulsa, USA) based on the variance analysis for a 2-factor experiment (I factor: oat β-glucan concentrates addition, II factor: storage period). The experiment was designed as a full randomized block with three independent biological replications (in each different ingredients were used). Results are presented as the mean±standard error. The influence of the treatment group, the time of storage and the interaction between the two were analysed by the general linear model procedure (GLM) with Tukey’s test at α = 0.05 significance level (Tukey, Citation1977).
3. Results and discussion
3.1. Proximate composition
The mean percent moisture, fat, protein and ash for raw burgers from different treatments are presented in . The moisture values of all treated burgers were significantly higher than in the control group (P < 0.05). This is probably connected with the high water-binding capacity of β-glucan (Ahmad, Muhammad, Zahoor, Nawaz & Ahmed, Citation2010). Significant increase in protein content was observed after β-glucan addition for both concentrates (P < 0.05). The increase of protein content in the final product is probably associated with proteins contained in the β-glucans concentrates (15% β-glucan concentrate powder contained 16.00% of proteins; 30% β-glucan concentrate powder contained 20.36% of proteins). Replacing fats with β-glucan (for both concentrates) caused the significant decrease of fat and increase ash content (P < 0.05).
Table 3. Proximate composition of beef burgers (mean±SE). Treatments: control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Tabla 3. Composición aproximada de las hamburguesas de carne (media ± EE). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%
3.2. β-glucan content
The β-glucan concentration in beef burgers on day 0, 4, 7 and 11 of storage is presented in . Target mass ratio of 1% β-glucan was achieved for the final product. On day 0, no significant difference in β-glucan content was found between the B15%8 and B30%4 groups (P > 0.05). All control samples during all days of storage contained 0.00% of β-glucan content. Changes in β-glucan content caused by storage time were probably related to changes and oxidative degradation of β-glucans (Faure et al., Citation2014; Mäkelä et al., Citation2015).
Figure 1. Effect of fat replacement by oat concentrates on β-glucan content in low-fat beef burgers during storage measured on the 0th, 4th, 7th and 11th day of storage (respectively: D0, D4, D7, D11) (mean ± SE). Treatments: Control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Figura 1. Efecto del remplazo de la grasa por concentrados de β-glucano de avena en las hamburguesas de carne de res de bajo contenido graso durante el almacenamiento, medido al día 0, 4, 7 y 11 de almacenamiento (respectivamente: D0, D4, D7, D11) (media ± error estándar [EE]). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%
![Figure 1. Effect of fat replacement by oat concentrates on β-glucan content in low-fat beef burgers during storage measured on the 0th, 4th, 7th and 11th day of storage (respectively: D0, D4, D7, D11) (mean ± SE). Treatments: Control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.Figura 1. Efecto del remplazo de la grasa por concentrados de β-glucano de avena en las hamburguesas de carne de res de bajo contenido graso durante el almacenamiento, medido al día 0, 4, 7 y 11 de almacenamiento (respectivamente: D0, D4, D7, D11) (media ± error estándar [EE]). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%](/cms/asset/8478327d-0101-4c6a-8ae4-3e05de07343c/tcyt_a_1750095_f0001_b.gif)
3.3. pH, WHC and colour parameters
The pH values of raw burgers are given in . The addition of β-glucan for both concentrates increased pH compared to the control samples in raw burgers at the beginning of the experiment on day 0 of storage (P < 0.05). The highest pH values on day 0 were observed in the B15%12 (pH 5.88 ± 0.00) and B30%6 group (pH 5.87 ± 0.00). During storage, the pH value decreased significantly in all analysed groups (P < 0.05). These results are in agreement with Talukder and Sharma (Citation2010), who studied the influence of wheat bran and oat in chicken meat patties, and Yılmaz (Citation2005), who reported the increase of pH value in low-fat meatballs with added wheat bran. Some authors, however, reported no influence in the pH values of meat products with added wheat and oat fibre (Huang et al., Citation2011).
Table 4. Evaluation of pH and WHC value in beef burgers during storage in modified atmosphere at refrigerated conditions on the 0th, 4th, 7th and 11th day of storage (respectively: D0, D4, D7, D11) (mean±SE). Treatments: Control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Tabla 4. Evaluación del valor de pH y WHC en las hamburguesas de carne de res durante el almacenamiento en atmósfera modificada bajo condiciones de refrigeración en el día 0, 4, 7 y 11 de almacenamiento (respectivamente: D0, D4, D7, D11) (media ± EE). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%
The WHC values of analysed burgers during refrigerated storage in the modified atmosphere are presented in . In all treatment groups with concentrates addition, the analysis has shown a significant effect of the addition of β-glucan concentrates on burger WHC at the beginning of the experiment on day 0 of storage (P < 0.05). The highest increase of WHC for the B15%12 samples was obtained on the first day of analysis (94.14%). Storage time influenced the decrease of WHC in all analysed groups (P < 0.05). Talukder and Sharma (Citation2010) also showed that adding oat and wheat bran increases the WHC of meat products. A similar conclusion can be found in Fernández-Ginés et al. (Citation2005) and Talukder (Citation2013).
The colour values of raw low-fat beef burgers on day 0, 4, 7 and 11 of storage are presented in . The results showed no significant effect of the addition of β-glucan B15% in the amount of 8% and 12% and β-glucan 30% in the amount of 4% on L* values at the beginning of the experiment on day 0 of storage (P > 0.05). These results are consistent with Bastos et al. (Citation2014). Significant decrease of the L* value caused by the addition of β-glucan was noticed only in the B30%4 and B30%6 groups (P < 0.05). In all groups, significant effect of storage time on the L* values of the burgers was observed (P < 0.05). The addition of β-glucan did not have a significant effect on a* values on day 0 for both concentrates (P > 0.05) except B30%6 group. In all groups, a significant decrease of storage time on the a* value of the burgers was observed (P < 0.05). A significant increase of the b* value caused by the addition of β-glucan was obtained in all analysed groups (P < 0.05). During storage, the b* value decreased significantly in all treatment groups (P < 0.05). A similar tendency of the b* value to increase in meat products with hydrated oatmeal addition was also obtained by Yang et al. (Citation2010) and Serdaroglu (Citation2006) on beef patties containing oat flour (Serdaroglu, Citation2006; Yang et al., Citation2010).
Table 5. Evaluation of CIE parameters (L*, a* and b*) in beef burgers during storage in modified atmosphere at refrigerated conditions on the 0th, 4th, 7th and 11th day of storage (respectively: D0, D4, D7, D11) (mean±SE). Treatments: Control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Tabla 5. Evaluación de los parámetros CIE (L*, a* y b*) de las hamburguesas de carne de res durante el almacenamiento en atmósfera modificada bajo condiciones de refrigeración en el día 0, 4, 7 y 11 de almacenamiento (respectivamente: D0, D4, D7, D11) (media ± EE). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%
3.4. Texture parameters
For grilled burgers (), all studied textural parameters were influenced by the addition of both β-glucan concentrates. Hardness values increased significantly in all treatment groups with concentrates addition (P < 0.05). Only in the B15%12 sample did springiness, increase significantly (P < 0.05). No significant differences in the springiness of burgers were found in the B15%8, B30%4 and B30%6 samples compared to the control group (P > 0.05). The cohesiveness and gumminess of burgers increased significantly after adding both β-glucan concentrates (P < 0.05). A significantly higher value of gumminess was obtained in B15%12 and B30%6 burgers (P < 0.05). These results are in agreement with Santhi and Kalaikannan (Citation2014) and Tabarestani and Tehrani (Citation2014).
Table 6. Texture profile analysis of grilled beef burgers on the 0th day of storage (mean±SE). Treatments: Control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Tabla 6. Análisis del perfil de textura de las hamburguesas de carne de res preparadas a la parrilla el día 0 de almacenamiento (media ± EE). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%
3.5. Cholesterol content
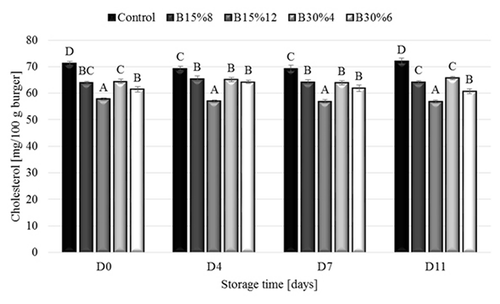
At the start of the experiment and on day 4, 7 and 11 of storage, significantly lower cholesterol content was observed in all samples with added β-glucan concentrates (P > 0.05) (). Similar results were presented in the work of Piñero et al. (Citation2008) The lowest cholesterol content was obtained in B15%12 samples on all days of analysis. No significant changes were observed in cholesterol content during storage (P > 0.05).
Figure 2. Effect of fat replacement by oat β-glucan concentrates on cholesterol content in low-fat beef burgers during storage measured on the 0th, 4th, 7th and 11th day of storage (respectively: D0, D4, D7, D11) (mean ± SE). Treatments: control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Figura 2. Efecto de la sustitución de la grasa por concentrados de β-glucano de avena en el contenido de colesterol de las hamburguesas de carne de res de bajo contenido graso durante el almacenamiento, medido en el día 0, 4, 7 y 11 (respectivamente: D0, D4, D7, D11) (media ± EE). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%; B30%6 – adición de 6% del concentrado de β-glucano de avena al 30%
3.6. Volatile compounds profile
Principal component analysis was used to indicate the differences in the volatile compounds in beef burgers compared to the control group. The results of the PCA analysis are shown in . Values of 97.03% data variance explained by the horizontal axis and 22.79% interpreted by the vertical axis explain the difference among samples caused by adding β-glucan concentrates. The PCA analysis demonstrated that the amount and concentration (15% and 30%) of added β-glucan changed the volatile compounds profile of raw burgers in comparison with the control group. Heat treatment of the burgers reduced the differences between the samples with additional β-glucan, but they still differed from the control samples. The volatile compound profiles of raw and grilled on the 0 day of the experiment are presented in and respectively. The analysis identified the 25 typical volatile compounds in raw and grilled samples. In raw samples, 20 compounds were identified in the control group and 23 compounds in each from the following groups: B15%8, B15%12, B30%4 and B30%6. In grilled samples, 21 compounds were identified in the following burgers: the control samples, B30%4 and B30%6. Twenty-two compounds were identified in samples B15%8 and B15%12. In all raw samples with β-glucan concentrates the presence of 2-methylpropanal and pyrazine was observed. The presence of 2-methylpropanal is related to Strecker degradation of the amino acid valine or presence of microbial metabolism (Purri et al., Citation2010). Whereas pyrazine is a heterocyclic compound created in the interaction of the Maillard reaction and the Strecker degradation (Whitfield, Citation2009). Additionally, heat treatment caused increasing of pyrazine intensity in all samples with β-glucan. Also in grilled burgers, 2,3-dimethylpyrazine was found especially in samples with β-glucan concentrates. 2,3-dimethylpyrazine is a volatile compound representing roasted flavors and is formed in the Maillard reaction (Cha et al., Citation2019). 2,3-dimethylpyrazine is also connected with roasted grains aroma which explains its increased intensity in samples with β-glucan (Puvipirom & Chaiseri, Citation2012). β-glucan concentrates addition changes the volatile compound profile decreasing the meaty aroma (Sánchez-Zapata et al., Citation2010; Troutt et al., Citation1992). Reduction of beef tallow amount decreases the fat aroma intensity which is connected with a presence among others of pyridine 2-pentyl (O’Hare & Grigor, Citation2005). Also, the volatile compound profiles of grilled samples showed that the addition of β-glucan reduces butane-2,3-dione, the diacetyl identified as a buttery flavour (Huchedé et al., Citation2019).
Table 7. Volatile compounds recognized using AroChemBase in raw samples (relative area of peaks [%]).Treatments: Control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Tabla 7. Compuestos volátiles identificados usando AroChemBase en muestras crudas (área relativa de los picos [%]). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%; B30%6 – adición de 6% del concentrado de β-glucano de avena al 30%
Table 8. Volatile compounds recognized using AroChemBase in grilled samples (relative area of peaks [%]).Treatments: Control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Tabla 8. Compuestos volátiles identificados usando AroChemBase en muestras a la parrilla (área relativa de los picos [%]). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%; B30%6 – adición de 6% del concentrado de β-glucano de avena al 30%
Figure 3. PCA analysis of the effect of oat β-glucan addition on the volatile compounds in raw and grilled beef burger treatments: control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Figura 3. Análisis PCA del efecto de añadir β-glucano de avena en los compuestos volátiles de las hamburguesas de carne cruda y asada. Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%; B30%6 – adición de 6% del concentrado de β-glucano de avena al 30%

3.7. Consumer evaluation
The results of consumer evaluation of different burger formulations are presented in . The concentration and amount of β-glucan solution had a significant effect on acceptance decline (P < 0.05) for the majority of the sensory attributes (external appearance, juiciness, taste, aroma, texture, overall acceptance) on day 0 of storage. Only the colour has not changed with the addition of both β-glucan concentrates and during storage time (P > 0.05). On the other hand, consumer colour evaluation was correlated with instrumentally evaluated colour parameters. On day 0 of storage, the authors reported there is an inverse correlation between b* value measured instrumentally and acceptance of colour evaluated by consumers (r = −0.792). Correlation at the level of r = 0.525 was calculated between a* value measured instrumentally and acceptance of colour evaluated by consumers on day 0 of the experiment. Also, correlation values were calculated between results of instrumental texture analysis and sensory texture acceptance on day 0. It has been shown strong inverse correlation between acceptance of texture and springiness (r = −0.836) and between acceptance of texture and cohesiveness (r = −0.862). Correlations at the level of r = −0.805 and r = −0.796 were evaluated between texture acceptance and gumminess and between texture acceptance and chewiness, respectively. These findings agree with those of Piñero et al. (Citation2008) who found that the oat’s soluble fibre external appearance and colour of low-fat beef products are comparable to control samples. Previously, Afshari et al. (Citation2017) showed that the reduction of flavor in burgers with inulin and β-glucan is connected witch starchy flavor of β-glucan. On day 0 the B30%6 and B30%4 samples were similarly evaluated for all descriptors.
Table 9. Influence of different oat β-glucan concentrates addition and storage time on sensory attributes measured on the 0th, 4th, 7th and 11th day of storage (respectively: D0, D4, D7, D11). Treatments: Control; B15%8 – 8% of 15% oat β-glucan concentrate addition; B15%12 – 12% of 15% oat β-glucan concentrate addition; B30%4 – 4% of 30% oat β-glucan concentrate addition; B30%6 – 6% of 30% oat β-glucan concentrate addition.
Tabla 9. Efecto provocado por la adición de diferentes concentrados de β-glucano de avena y de diferentes tiempos de almacenamiento en los atributos sensoriales medidos en el día 0, 4, 7 y 11 de almacenamiento (respectivamente: D0, D4, D7, D11). Tratamientos: Control; B15%8 – adición de 8% del concentrado de β-glucano de avena al 15%; B15%12 – adición de 12% del concentrado de β-glucano de avena al 15%; B30%4 – adición de 4% del concentrado de β-glucano de avena al 30%
4. Conclusion
Fat replacement by oat β-glucan concentrates seems to be technologically feasible for the manufacture of low-fat meat products. According to the experimental results, beef tallow substitution by oat β‐glucan concentrates increases the WHC value and reduces cholesterol content in the final product. Use of oat β‐glucan as a beef fat replacer increases significantly pH value and in consequences has an influence on colour parameters of low‐fat beef burgers. The use of 15% and 30% oat β‐glucan concentrates cause the increase of texture parameters (hardness, springiness, cohesiveness, gumminess, chewiness), and changes the profile of volatile compounds of low‐ fat beef burgers. Most of sensory properties acceptance (external appearance, taste, aroma, texture, juiciness, overall acceptability) was changed by the oat β‐glucan concentrates use. Only acceptance of colour did not change in the samples with 15% and 30% oat β‐glucan concentrates. The target value of 1% of β-glucan for the final product was achieved for all samples with 15% and 30% oat β‐glucan concentrates.
This modification could allow the substitution of casual beef burgers with healthier low-fat meat product with similar customer appeal. It can be a statement the technological and nutritional functions of minced beef products can be improved by substitution animal fat using β-glucan concentrates.
Conflicts of interest
The authors declare no conflict of interest.
Acknowledgments
This research was performed as part of Project no. WND-POIG.01.03.01-00-204/09 Optimizing of Beef Production in Poland in accordance with the “From Farm to Fork” Strategy co-financed by the European Regional Development Fund under the Innovative Economy Operational Program 2007–2013.
Additional information
Funding
References
- Afshari, R., Hosseini, H., Mousavi Khaneghah, A., & Khaksar, R. (2017). Physico-chemical properties of functional low-fat beef burgers: Fatty acid profile modification. LWT - Food Science and Technology, 78(5), 325–331. https://doi.org/10.1016/j.lwt.2016.12.054
- Ahmad, A., Muhammad, F., Zahoor, T., Nawaz, H., & Ahmed, Z. (2010). Extraction and characterization of beta-D-glucan from oat for industrial utilization. International Journal of Biological Macromolecules, 46(3), 304–309. https://doi.org/10.1016/j.ijbiomac.2010.01.002
- Ahmad, A., Munir, B., Abrar, M., Bashir, S., Adnan, M., & Tabassum, T. (2012). Perspective of β-glucan as functional ingredient for food industry. Journal of Nutrition & Food Sciences, 2 (2), 2–10. https://doi.org/10.4172/2155-9600.1000133implications
- Álvarez, D., & Barbut, S. (2013). Effect of inulin, β-glucan and their mixtures on emulsion stability, color and textural parameters of cooked meat batters. Meat Science, 94(3), 320–327. https://doi.org/10.1016/j.meatsci.2013.02.011
- Bastos, S. C., Pimenta, M. E. S. G., Pimenta, C. J., Reis, T. A., Nunes, C. A., Pinheiro, A. C. M., Fabrício, L. F. F., & Leal, R. S. (2014). Alternative fat substitutes for beef burger: Technological and sensory characteristics. Journal of Food Science and Technology, 51(9), 2046–2053. https://doi.org/10.1007/s13197-013-1233-2
- Bozbulut, R., & Sanlier, N. (2019). Promising effects of β-glucans on glyceamic control in diabetes. Trends in Food Science & Technology, 83, 159–166. Retrieved October, 2018, from. https://doi.org/10.1016/j.tifs.2018.11.018
- Brewer, M. S. (2012). Reducing the fat content in ground beef without sacrificing quality: A review. Meat Science, 91(4), 385–395. https://doi.org/10.1016/j.meatsci.2012.02.024
- Cha, J., Debnath, T., & Lee, K. (2019). Analysis of α-dicarbonyl compounds and volatiles formed in Maillard reaction model systems. Scientific Reports, 9(5325), 1–6. https://doi.org/10.1038/s41598-019-41824-8
- Chang, H., Huang, C., Yeh, D., Wang, S., Peng, C., & Wang, C. (2013). Oat prevents obesity and abdominal fat distribution, and improves liver function in humans. Plant Foods for Human Nutrition, 68(1), 18–23. https://doi.org/10.1007/s11130-013-0336-2
- Choi, J. W., Kim, S. H., Mun, S., Lee, S. J., Shim, J. Y., & Kim, Y. R. (2011). Optimizing the replacement of pork fat with fractionated barley flour paste in reduced-fat sausage. Food Science and Biotechnology, 20(3), 687–694. https://doi.org/10.1007/s10068-011-0097-3
- Da Oliveira, L. C., Oliveira, M., Meneghetti, V. L., Mazzutti, S., Colla, L. M., Elias, M. C., & Gutkoski, L. C. (2012). Effect of drying temperature on quality of β-glucan in white oat grains. Food Science and Technology (Campinas), 32(4), 775–783. https://doi.org/10.1590/S0101-20612012005000105
- Daou, C., & Zhang, H. (2012). Oat beta-glucan: Its role in health promotion and prevention of diseases. Comprehensive Reviews in Food Science and Food Safety, 11(4), 355–365. https://doi.org/10.1111/j.1541-4337.2012.00189.x
- Drewnowski, A., & Almiron-Roig, E. (2010). Human perceptions and preferences for fat-rich foods. In J. -P. Montmayeur & J. le Coutre (Eds.), Fat Detection: Taste, Texture, and Post Ingestive Effects (1st ed., pp. 265–294). Boca Raton, FL: CRC Press/Taylor & Francis. https://doi.org/10.1201/9781420067767-c11
- EFSA. (2010). Scientific Opinion on the substantiation of a health claim related to oat beta-glucan and lowering blood cholesterol and reduced risk of (coronary) heart disease pursuant to Article 14 of Regulation (EC) No 1924/2006 (Vol. 8). Parma.
- EFSA. (2011). Scientific Opinion on the substantiation of health claims related to beta-glucans from oats and barley and maintenance of normal blood LDL-cholesterol concentrations (ID 1236, 1299), increase in satiety leading to a reduction in energy intake (ID 851, 850 (Vol. 9)). Parma.
- European Commission. Commission Regulation (EU) No 432/2012. (EU) No 432/2012 of 16 May 2012 establishing a list of permitted health claims made on foods, other than those referring to the reduction of disease risk and to children’s development and health. (n.d.).
- Faure, A. M., Sánchez-ferrer, A., Zabara, A., Andersen, M. L., & Nyström, L. (2014). Modulating the structural properties of β-d-glucan degradation products by alternative reaction pathways. Carbohydrate Polymers, 99, 679–686. https://doi.org/10.1016/j.carbpol.2013.08.022
- Fernández-Ginés, J. M., Fernández-López, J., Sayas-Barberá, E., & Pérez-Álvarez, J. A. (2005). Meat products as functional foods: A review. Concise Reviews and Hypotheses in Food Science, 70(2), 37–43. https://doi.org/10.1111/j.1365-2621.2005.tb07110.x
- Fulgoni, V. L., Chu, Y., Shea, M. O., Slavin, J. L., & Dirienzo, M. A. (2015). Oatmeal consumption is associated with better diet quality and lower body mass index in adults: The National Health and Nutrition Examination Survey (NHANES), 2001-2010. Nutrition Research, 35(12), 1052–1059. https://doi.org/10.1016/j.nutres.2015.09.015
- Gheisari, H. R., Aminlari, M., & Shekarforoush, S. S. (2009). A comparative study of the biochemical and functional properties of camel and cattle meat during frozen storage. Veterinarski Arhiv, 79(1), 51–68. https://hrcak.srce.hr/33128
- Gordon-Larsen, P., Guilkey, D. K., & Popkin, B. M. (2011). An economic analysis of community-level fast food prices and individual-level fast food intake: A longitudinal study. Health & Place, 17(6), 1235–1241. https://doi.org/10.1016/j.healthplace.2011.07.011
- Ho, H. V. T., Sievenpiper, J. L., Zurbau, A., Mejia, S. B., Jovanovski, E., Au-Yeung, F., Jenkins, A. L., & Vuksan, V. (2016). The effect of oat β-glucan on LDL-cholesterol, non-HDL-cholesterol and apoB for CVD risk reduction: A systematic review and meta-analysis of randomised-controlled trials. British Journal of Nutrition, 116(8), 1369–1382. https://doi.org/10.1017/S000711451600341X
- Huang, S. C., Tsai, Y. F., & Chen, C. M. (2011). Effects of wheat fiber, oat fiber, and inulin on sensory and physico-chemical properties of Chinese-style sausages. Asian-Australasian Journal of Animal Sciences, 24(6), 875–880. https://doi.org/10.5713/ajas.2011.10317
- Huchedé, M. Q. G., Gauthier, G., Bellieres-Baca, V., Michel, C., & Millet, J. M. (2019). New process of production of butane-2,3-dione by oxidative dehydrogenation of 3-hydroxybutanone. Reaction Chemistry & Engineering, 4, 932–938. https://doi.org/10.1039/C9RE00045C
- Jiménez-Colmenero, F. (2007). Healthier lipid formulation approaches in meat-based functional foods. Technological options for replacement of meat fats by non-meat fats. Trends in Food Science & Technology, 18(11), 567–578. https://doi.org/10.1016/j.tifs.2007.05.006
- Ktari, N., Trabelsi, I., Bkhairia, I., Triki, M., Taktak, M. A., Moussa, H., Nasri, M., & Salah, R. B. (2016). Using barley beta glucan, citrus, and carrot fibers as a meat substitute in Turkey meat sausages and their effects on sensory characteristics and properties. Journal of Food Processing and Technology, 7(9), 9. https://doi.org/10.4172/2157-7110.1000620
- Kurek, M., Wyrwisz, J., Piwińska, M., & Wierzbicka, A. (2016). Application of the response surface methodology in optimizing oat fiber particle size and flour replacement in wheat bread rolls. CyTA - Journal of Food, 14(1), 18–26. https://doi.org/10.1080/19476337.2015.1036309
- Lee, S. H., Jang, G. Y., Hwang, I. G., Kim, H. Y., Woo, K. S., Kim, K. J., Lee, M. J., Kim, T. J., Lee, J., & Jeong, H. S. (2015). Physicochemical properties of β-glucan from acid hydrolyzed barley. Preventive Nutrition and Food Science, 20(2), 110–118. https://doi.org/10.3746/pnf.2015.20.2.110
- Mäkelä, N., Sontag-Strohm, T., & Maina, N. H. (2015). The oxidative degradation of barley β-glucan in the presence of ascorbic acid or hydrogen peroxide. Carbohydrate Polymers, 123, 390–395. https://doi.org/10.1016/j.carbpol.2015.01.037
- Meat Standards Australia (MSA). (2008). Accessory publication: MSA sensory testing protocols. Australian Journal of Experimental Agriculture, 48(11), 1360–1367. https://doi.org/10.1071/EA07176_AC
- Moczkowska, M., Półtorak, A., & Wierzbicka, A. (2015). Impact of the ageing process on the intensity of post mortem proteolysis and tenderness of beef from crossbreeds. Bulletin of the Veterinary Institute in Pulawy, 59(3), 361–367. https://doi.org/10.1515/bvip-2015-0054
- Morin, L. A., Temelli, F., & McMullen, L. (2004). Interactions between meat proteins and barley (Hordeum spp.) β-glucan within a reduced-fat breakfast sausage system. Meat Science, 68(3), 419–430. https://doi.org/10.1016/j.meatsci.2004.04.009
- O’Hare, L., & Grigor, J. (2005). Flavor generation in food. In D. J. Rowe (Ed.), Chemistry and technology of flavors and fragrances (pp. 35–52). Blackwell Publishing Ltd Editorial.
- O’Neil, C. E., Nicklas, T. A., Fulgoni, V. L., & DiRienzo, M. A. (2015). Cooked oatmeal consumption is associated with better diet quality, better nutrient intakes, and reduced risk for central adiposity and obesity in children 2-18 years: NHANES 2001-2010. Food & Nutrition Research, 59(1), 26673. https://doi.org/10.3402/fnr.v59.26673
- Ognean, C. F., Darie, N., & Ognean, M. (2006). Fat replacers – Review. Journal of Agroalimentary Processes and Technologies, XII(2), 433–442. https://www.journal-of-agroalimentary.ro/admin/articole/74033L66_FAT_REPLACERS_final.pdf
- Othman, R. A., Moghadasian, M. H., & Jones, P. J. H. (2011). Cholesterol-lowering effects of oat β-glucan. Nutrition Reviews, 69(6), 299–309. https://doi.org/10.1111/j.1753-4887.2011.00401.x
- Pearsos, S., Stern, S., & McGavack, T. H. (1953). A rapid, accurate method for the determination of total cholesterol in serum. Analytical Chemistry, 25(5), 813–814. https://doi.org/10.1021/ac60077a041
- Piñero, M. P., Parra, K., Huerta-leidenz, N., Moreno, L. A., De, Ferrer, M., Araujo, S., & Barboza, Y. (2008). Effect of oat’s soluble fibre (β-glucan) as a fat replacer on physical, chemical, microbiological and sensory properties of low-fat beef patties. Meat Science, 80(3), 675–680. https://doi.org/10.1016/j.meatsci.2008.03.006
- Pintado, T., Herrero, A. M., Jiménez-Colmenero, F., & Ruiz-Capillas, C. (2016). Emulsion gels as potential fat replacers delivering β-glucan and healthy lipid content for food applications. Journal of Food Science and Technology, 53(12), 4336–4347. https://doi.org/10.1007/s13197-016-2432-4
- Półtorak, A., Wyrwisz, J., Moczkowska, M., Marcinkowska-Lesiak, M., Stelmasiak, A., Ulanicka, U., Zalewska, M., Wierzbicka, A., & Sun, D.-W. (2014). Correlation between instrumental texture and colour quality attributes with sensory analysis of selected cheeses as affected by fat contents. International Journal of Food Science & Technology, 50(4), 1–10. https://doi.org/10.1111/ijfs.12707
- Purri, L., Berm, R., Franco, D., Carballo, J., & Lorenzo, M. (2010). Development of volatile compounds during the manufacture of dry-cured “Lacon,” a Spanish traditional meat product. Journal of Food Science, 76(1), C89–C97. https://doi.org/10.1111/j.1750-3841.2010.01955.x
- Puvipirom, J., & Chaiseri, S. (2012). Contribution of roasted grains and seeds in aroma of oleang (Thai coffee drink). International Food Research Journal, 19(2), 583–588. http://www.ifrj.upm.edu.my/19%20(02)%202012/(31)IFRJ-2012%20Chaiseri.pdf
- Ravisankar, P., Reddy, A. A., Nagalakshmi, B., Sai, O., Kumar, B. V., & Anvith, P. S. (2015). The comprehensive review on fat soluble vitamins. IOSR Journal of Pharmacy, 5(11), 12–28. http://iosrphr.org/papers/v5i11/D0511012028.pdf
- Sánchez-Zapata, E., Muñoz, C. M., Fuentes, E., Fernández-López, J., Sendra, E., Sayas, E., Navarro, C., & Pérez-Alvarez, J. A. (2010). Effect of tiger nut fibre on quality characteristics of pork burger. Meat Science, 85(1), 70–76. https://doi.org/10.1016/j.meatsci.2009.12.006
- Santhi, D., & Kalaikannan, A. (2014). The effect of the addition of oat flour in low-fat chicken nuggets. Journal of Nutrition & Food Sciences, 4(1), 1–4. https://doi.org/10.4172/2155-9600.1000260
- Serdaroglu, M. (2006). The characteristics of beef patties containing different levels of fat and oat flour. International Journal of Food Science and Technology, 41(2), 147–153. https://doi.org/10.1111/j.1365-2621.2005.01041.x
- Shanmugam, J., & Priya, V. (2016). Effects of intake of oatmeal in serum cholesterol. International Journal of Science and Research, 5(8), 2015–2017. https://lipidworld.biomedcentral.com/articles/10.1186/s12944-015-0138-7
- Soncu, E. D., Kolsarıcı, N., Çiçek, N., Öztürk, G., Akoğlu, I., & Arıcı, Y. (2015). The comparative effect of carrot and lemon fiber as a fat replacer on physico-chemical, textural, and organoleptic quality of low-fat beef hamburger. Korean Journal for Food Science of Animal Resources, 35(3), 370–381. https://doi.org/10.5851/kosfa.2015.35.3.370
- Szpicer, A., Onopiuk, A., Półtorak, A., & Wierzbicka, A. (2018). Influence of oat β-glucan and canola oil addition on the physico-chemical properties of low‐fat beef burgers. Journal of Food Processing and Preservation, 42(11), e13785. https://doi.org/10.1111/jfpp.13785
- Szpicer, A., Onopiuk, A., Półtorak, A., & Wierzbicka, A. (2019). Influence of tallow replacement by oat β -glucan and canola oil on the fatty acid and volatile compound profiles of low-fat beef burgers. CyTA - Journal of Food, 17(1), 926–936. https://doi.org/10.1080/19476337.2019.1674924
- Tabarestani, H. S., & Tehrani, M. M. (2014). Optimization of physicochemical properties of low-fat hamburger formulation using blend of soy flour, split-pea flour and wheat starch as part of fat replacer system. Journal of Food Processing and Preservation, 38(1), 278–288. https://doi.org/10.1111/j.1745-4549.2012.00774.x
- Talukder, S. (2013). Effect of dietary fiber on properties and acceptance of meat products: A review. Critical Reviews in Food Science and Nutrition, 55(7), 1005–1011. https://doi.org/10.1080/10408398.2012.682230
- Talukder, S., & Sharma, D. P. (2010). Development of dietary fiber rich chicken meat patties using wheat and oat bran. Journal of Food Science and Technology, 47(2), 224–229. https://doi.org/10.1007/s13197-010-0027-z
- Troutt, E. S., Hunt, M. C., Johnson, D. E., Claus, J. R., Kastner, C. L., Kropf, D. H., & Stroda, S. (1992). Chemical, physical, and sensory characterization ground beef containing 5 to 30 percent fat of chemical compositions. Journal of Food Science, 57(1), 25–29. https://doi.org/10.1111/j.1365-2621.1992.tb05416.x
- Tukey, J. W. (1977). Some thoughts on clinical trials, especially problems of multiplicity. Science, 198(4318), 679–684. https://doi.org/10.1126/science.333584
- Whitfield, F. B. (2009). Volatiles from interactions of Maillard reactions and lipids. Food Science and Nutrition, 31(1/2), 37–41. https://doi.org/10.1080/10408399209527560
- Wojtasik-Kalinowska, I., Guzek, D., Brodowska, M., Godziszewska, J., Sakowska, A., Gantner, M., & Wierzbicka, A. 2017. The effect of addition of Nigella sativa L. oil on the quality and shelf life of pork patties. Journal of Food Processing and Preservation, 41(6), 1–12. November 2016. https://doi.org/10.1111/jfpp.13294.
- Yang, H., Kim, G., Choi, S., & Joo, S. (2010). Physical and sensory properties of low fat sausage amended with hydrated oatmeal and various meats. Korean Journal for Food Science of Animal Resources, 30(3), 365–372. https://doi.org/10.5851/kosfa.2010.30.3.365
- Yılmaz, I. (2005). Physicochemical and sensory characteristics of low fat meatballs with added wheat bran. Journal of Food Engineering, 69(3), 369–373. https://doi.org/10.1016/j.jfoodeng.2004.08.028
