ABSTRACT
Over the last two decades our understanding of the gut microbiota and its contribution to health and disease has been transformed. Among a new ‘generation’ of potentially beneficial microbes to have been recognized are members of the genus Eubacterium, who form a part of the core human gut microbiome. The genus consists of phylogenetically, and quite frequently phenotypically, diverse species, making Eubacterium a taxonomically unique and challenging genus. Several members of the genus produce butyrate, which plays a critical role in energy homeostasis, colonic motility, immunomodulation and suppression of inflammation in the gut. Eubacterium spp. also carry out bile acid and cholesterol transformations in the gut, thereby contributing to their homeostasis. Gut dysbiosis and a consequently modified representation of Eubacterium spp. in the gut, have been linked with various human disease states. This review provides an overview of Eubacterium species from a phylogenetic perspective, describes how they alter with diet and age and summarizes its association with the human gut and various health conditions.
Introduction
The importance of the gut microbiota in human health is now well establishedCitation1. Components of the microbiota can facilitate the extraction of energy from nutrients, the deposition of fat in adipose tissues and provide for other resident microbes of the gut, besides eliminating pathogens through exclusion and other means.Citation2 Depending on a plethora of factors that may be personal or environmental, the composition and function of the gut microbiota can vary significantly. However, distinguishing between a healthy or an unhealthy gut microbiome is difficult due to this large variability. Community composition alone, therefore, is not a reliable indicator of an aberrant or unhealthy stateCitation1 and, thus, a nuanced understanding of the microbiota, encompassing how specific taxa contribute to gut homeostasis and interact with their human host, is required for the development of evidence-based microbial therapeutics.Citation3,Citation4
Here, we focus on the genus Eubacterium, which was first proposed by Prévot in 1938 to describe a group of beneficial bacteria isolated from human feces.Citation5 Eubacterium spp. are frequently encountered in the oral cavity and intestinal tract of mammals, including in the rumen of ruminants, as well as in the environment. The genus forms one of the core genera of the human gut microbiota and shows widespread colonization of the human gut across various human populations in Africa,Citation6,Citation7 Australia,Citation8 Europe,Citation9 India,Citation10 South America,Citation11,Citation12 AsiaCitation13 and North America.Citation14,Citation15 Indeed, extensive human gut metagenome studies have reported the recovery of a large complement of metagenome-assembled Eubacterium rectale genomes irrespective of geographical location, age, lifestyle and clinical status.Citation16,Citation17 Interestingly, while Eubacterium spp. are routinely recovered from animal gut, an absence of E. rectale have been reported in primate gut; coupled with its omnipresence in the human gut this suggests a high degree of specificity and adaptation for the latter.Citation17
Multiple species of the genus are currently regarded as promising targets for microbial therapeutics. Indeed, recent consensus among gut microbiologists suggests that specific strains of butyrate-producing microbes belonging to the genera Eubacterium, Roseburia and Faecalibacterium, among others, may ultimately be considered as beneficial to human health in the same manner as strains of Lactobacillus and Bifidobacterium.Citation18 The genus Eubacterium is challenging to define, as discussed further below, and several species initially assigned to the genus have been subsequently reassigned to an existing or novel genus. Even now, the genus continues to be phylogenetically diverse and members can be assigned to several lineages. In recognition of this taxonomic flux, we will include some former Eubacterium species that have been recently reassigned to other genera for the purpose of this review. Additionally, we will largely restrict our discussion to Eubacterium species that are most relevant to the gut. Notably, even though much is known about the genus in general, our understanding of its function in the gut continues to evolve. Ultimately, here we review the literature to date relating to the phylogeny, characteristics and contributions of the members of the genus in relation to the human gut health and microbial ecology.
The genus Eubacterium is phylogenetically diverse
The genus Eubacterium consists of Gram positive, uniform or pleomorphic non-spore forming, obligately anaerobic, and chemoorganotrophic bacterial rods. Species in this genus can be saccharoclastic or nonsaccharoclastic and motile or immotile in nature.Citation19 Bacteria from this genus produce mixtures of organic acids from carbohydrates or peptone, which may include copious amounts of butyric, acetic and formic acids but do not produce: (a) only lactic acid, (b) propionic acid as the major acid, (c) greater quantities of acetic acid than lactic acid with or without the formation of formic acid and (d) lactic and succinic acid with small quantities of acetic or formic acid.Citation5 This definition is rather loose and leads to the incorporation of species in the genus by default; historically resulting in the inclusion of species with a variety of phenotypes and genotypes in the genus and, ultimately, making it highly heterogeneous. According to the latest iteration of the Bergey’s Manual of Systematics of Bacteria and ArchaeaCitation19 as well as NCBI Taxonomy, the genus Eubacterium belongs to the bacterial phylum Firmicutes, order Clostridiales and family Eubacteriaceae. However, according to the Genome Taxonomy Database (GTDB), which uses whole/draft genome information for classification of taxa, the genus should be assigned to the family Lachnospiraceae.Citation20 The genus currently consists of 42–44 species depending on the taxonomy being followed, and the major species of interest in relation to the human gut include Eubacterium rectale, E. hallii, E. ventriosum, E. eligens, E. coprostanoligens, and E. limosum. The DNA G + C content (mol%) of the genus varies from 30 to 57% and the type strain is Eubacterium limosum.
Due to the rather loose definition of the genus, many of the species currently in the genus do not belong in the genus sensu stricto and are likely be moved to novel or existing genera in time. Indeed, 16S rRNA analysis of the species in genus Eubacterium has highlighted their wide distribution across phylogenetic trees.Citation5 Here, we provide an update of this tree to display the phylogenetic relationship between some members of the genus and other closely related species using a representative maximum-likelihood tree constructed with 16 ribosomal protein markers (). The majority of members of Eubacterium that have undergone taxonomic reassignment are assigned to phylum Firmicutes and are widely distributed therein. Examples include E. formicigenerans and E. timidum, which were reassigned to the genera Dorea and Mogibacterium respectively.Citation24,Citation25 However, several other members have been reassigned to other phyla; instances include the reassignment of Eubacterium species to genera such as Slackia, Cryptobacterium and Eggerthela, all of which belong to phylum Actinobacteria. Notably, certain Eubacterium species such as E. cylindroides may exhibit both Gram-positive and Gram-negative characteristics, thereby creating ambiguity in a fundamental phenotypic characteristic that is frequently implemented in taxonomic assignment; this contributes further to the considerable confusion in classification of Eubacterium species.Citation26 It has been proposed that the core genotype of the genus Eubacterium sensu stricto be restricted to the type species of the genus, Eubacterium limnosum, along with Eubacterium callanderi, Eubacterium barkeri and Eubacterium aggregans,Citation19,Citation27 with the remaining species potentially assimilated into/reclassified as existing or novel genera when ample genomic and phylogenetic evidence supporting the same is available. For practicality, members have to date been grouped into subcategories based on phylogenetic characteristics. One of the loose phylogenetic subcategories proposed contain E. rectale, E. oxidoreducens, E. ramulus, Roseburia cecicola and R. intestinalis, where all species included except E. oxidoreducens produce butyrate and are saccharolytic.Citation19 The taxonomic relationships of the members in this subcategory are discussed in detail by Duncan et al,Citation28 and presents a strong case for reclassification of some of these species. E. eligens, an important gut Eubacterium, has been found to share considerable phylogenetic and phenotypic similarity with Lachnospira pectinoschiza and merits possible reclassification with availability of further evidence.Citation19
Figure 1. Phylogenetic relationship of Eubacterium spp. Complete genomes for Eubacterium species (current and recently reassigned) were obtained from NCBI along with other closely related gut microbes. 16 ribosomal marker proteins (including rpL14, rpL15, rpL16, rpL18, rpL22, rpL24, rpL2, rpL3, rpL4, rpL5, rpL6, rpS10, rpS17, rpS19, rpS3 and rpS8) were extracted from each genome, aligned with MAFFT v7.271Citation21 and concatenated to create a RP16 protein alignment. Phylogenetic reconstruction using maximum likelihood was carried out in IQ-TREECitation22 with the following settings: -mset WAG,LG,JTT,Dayhoff -mrate E,I,G,I + G -mfreq FU -wbtl. Only genomes with at least 4 ribosomal marker proteins were included in the tree. The resulting tree was visualized using iTOL.Citation23 Possible misclassifications are denoted by filled, inverted triangles in the phylogram. Tree nodes are depicted by filled circles.
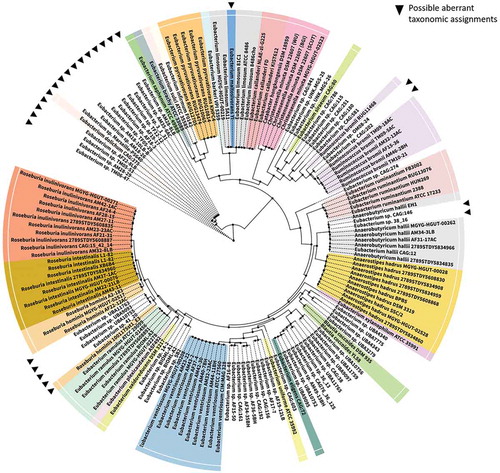
Certain Eubacterium species that are important in relation to gut health have already undergone, or are proposed to undergo, reclassification in view of their divergent phenotypic and phylogenetic characteristics. For example, Shetty et al. proposed the reclassification of E. hallii as Anaerobutyricum hallii Comb. Nov., when reporting a similar novel butyrate and propionate-producing species Anaerobutyricum soehngenii.Citation29 Indeed, E. hallii, along with E. indolis, E. cellulosolvens, E. plexicaudatum, E. ruminantium, E. saburreum, E. xylanophilum, E. uniforme, and E. ventriosum form a subcategory of interest in the genus Eubacterium. Notably, members of this group are not phylogenetically or phenotypically related to other species in the genus and exhibit distinct characteristics that warrants the creation of a novel genus for each.Citation19 Another common intestinal inhabitant, Eubacterium hadrum, was also assigned to the genus Anaerostipes, based on both genotypic and biochemical features.Citation30 Additionally, it has recently been proposed that one of the most important gut microbes, E. rectale, be reclassified as Agathobacter rectalis.Citation31 This reclassification was however challenged by Sheridan et al.Citation32 who argued that the evidence presented by Rosero et al.Citation31 did not justify reclassification. The need for reclassification of E. rectale was however acknowledged by Sheridan et al., but urged exercising caution with this important member of the human gut, noting that any change in its taxonomic or phylogenetic affiliations will have a major impact on human microbiota research.
Understandably, taxonomic reassignments proposed have not been universally accepted yet and indeed, as noted, care must be taken while considering taxonomic classification and reporting of any member of genus Eubacterium. Further efforts relating to the classification of the genus should have a primarily genotypic focus with an emphasis on genomic characteristics. The prokaryotic taxonomy devised by Parks et al.Citation33 in the GTDB, where a battery of universal, single copy marker genes derived from whole/draft genomes, are used to classify microorganisms, can be used as a model. Such an approach standardizes taxonomic assignments through normalization of taxonomic ranks on the basis of relative evolutionary divergence and has been shown to be capable of deconvoluting polyphyletic groups. Combined with rapidly declining sequencing prices, the increasing and ample availability of prokaryotic genomes can contribute greatly to such an exercise. With assembly of high-resolution draft genomes from metagenomes also now routine, microbiologists can glean information from truly uncultivable organisms and a definitive reclassification of the genus Eubacterium should be possible in the near future. Until then, there is likely to continue to be those who will view Eubacterium as a combined group – Eubacterium et rel. – when discussing human health, especially in relation to the gut. We will adopt this approach for the remainder of this review.
Modulation of Eubacterium spp. in the gut by diet and age
Diet is one of the most important factors that dictates the composition and diversity of the gut microbiota. In case of Eubacterium spp., their presence in the gut have been largely associated with increased intake of dietary fibers and have been shown to decrease with an increasing protein/fat percent in diet.Citation34 These observations are supported by recent studies outlining the utilization of digestion resistant complex carbohydrates by Eubacterium species.Citation35–Citation37 In a recent study by Scott et al., E. hallii and E. rectale were shown to be capable of utilizing media supplemented with resistant carbohydrates, i.e., fructans of increasing chain lengths such as P95 short-chain fructo-oligosaccharides, high-performance inulin, and Synergy-1; Dahlia inulin was metabolized exclusively by E. rectale.Citation35 Several studies have reported that a Western diet, which includes increased proportions of animal protein and fat and is low in fiber, leads to a marked decrease in bacterial abundance in the gut including desirable taxa such as Bifidobacterium and Eubacterium.Citation38–Citation40 The corollary all appears to be true in that research involving the Mediterranean diet, which is well established as a diet that can contribute to health, has been shown to increase Eubacterium spp. populations in the gut.Citation41,Citation42 Other studies with diverging diets have also contributed to our understanding of how Eubacterium spp. is modulated in the gut. For example, Noriega et al. investigated changes in the gut microbiota when a polyunsaturated omega-3 fatty acid-rich diet was fed to a 45-year-old male.Citation43 After the feeding phase, the fecal samples collected showed a drastic increase in abundances of several butyrate producers including Eubacterium spp. indicating a positive modulation of Eubacterium by polyunsaturated fatty acids. Further investigations, however, must be performed, to better understand the changes in Eubacterium spp. population in the gut with diet.
Through the process of aging, the gastrointestinal tract undergoes changes, including degeneration of the mucosal barrier and enteric nervous system along with an alteration of intestinal motility and an increase in gastrointestinal pathologies. As a general trend, microbiota diversity in the elderly is decreased with fewer butyrate producers and an increase in the number of potential pathogens.Citation44,Citation45 Among other things, a decrease in short-chain fatty acid (SCFA) production in the gut can result in an impaired secretion of mucins by the intestinal epithelial cells, providing enhanced access for pathogens to the intestinal mucosa and, potentially, gut inflammation.Citation46 In elderly individuals, gut inflammation can be exacerbated by impairment of the gut-associated lymphoid tissue (GALT), leading to inefficient control of the resident microbiota and release of pro-inflammatory cytokines and chemokines by enterocytes; with the latter driving the differentiation of effector TH1, TH2 and TH17 cells.Citation47–Citation49 Consistent with these observations, a decrease in the relative proportion of E. hallii, E. rectale, and E. ventriosum has been noted in centenarians, whereas potentially pathogenic bacteria from the phylum Proteobacteria were increased.Citation50 The beneficial effects of Eubacterium spp. were highlighted in an extensive study carried out by Ghosh et al., where a large cohort (n = 612) of elderly individuals were investigated to assess the modulatory effects of the Mediterranean diet on their gut microbiota.Citation42 The authors reported that Eubacterium species such as E. rectale and E. eligens were positively associated with several markers of lower frailty and improved cognitive ability as well as increased short/branched chain fatty acid production. Eubacterium spp. also showed negative correlations with inflammatory markers such as IL-2 and C-reactive protein. Furthermore, network analysis revealed Eubacterium spp. to be a keystone species in the elderly gut microbial ecosystem, with frailty-associated taxa on the fringe. However, in contrast to these general observations, other studies have inferred a positive association between Eubacterium spp. and age.Citation51,Citation52 Clearly, inconsistent observations, albeit from studies involving diverse experimental designs with presumably quite variable diets, highlight the need for additional age-related studies in which other variables, especially diet, are as consistent as possible.
Short-chain fatty acids produced by Eubacterium spp. contribute to gut health
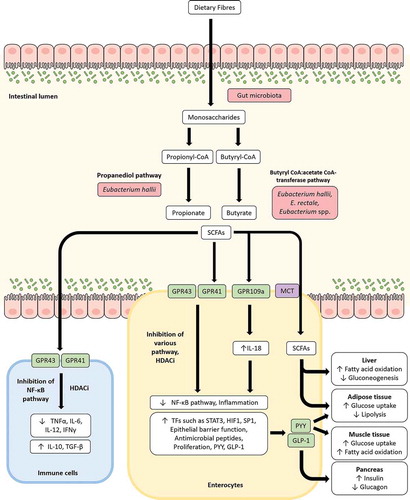
During the process of digestion, most available nutrients undergo absorption in the duodenum. However, a fraction of ingested carbohydrates that are resistant to digestion, including dietary fibers, remain intact until they reach the colon. Here, these microbiota accessible carbohydrates (MACs) are fermented and metabolized by specialized enzymes produced by the resident gut microbiotaCitation53 (). Microbial degradation of these complex carbohydrates, and of host mucin, results in the production of hexoses and pentoses before subsequent conversion to lactate and SCFAs such as acetate, propionate, butyrate, formate, and succinate by several other gut microbes.Citation54 These SCFAs can, in turn, be taken up by the host. Unsurprisingly, ingestion of dietary fibers have been directly correlated to SCFA concentration and abundance of butyrate producers including Eubacterium spp., whereas high-fat diets have been associated with reduced formation of SCFAs.Citation34,Citation55,Citation56 Additionally, the abundance of Eubacterium spp. in the gut is strongly correlated with SCFA levels and the beneficial effects of SCFAs in a variety of clinical conditions such as inflammatory bowel diseases, metabolic syndromes, and colorectal cancer, as discussed below.
Figure 2. Modulation of various processes through short-chain fatty acids (SCFAs) produced by Eubacterium spp. Upon reaching the gut, carbohydrates resistant to digestion (commonly derived from dietary fibers) are degraded by gut microbiota to produce monosaccharides. These monosaccharides can be utilized by certain bacteria, including Eubacterium spp., in the gut to produce SCFAs such as butyrate, propionate, and acetate. SCFAs interact with G-protein-coupled receptors such as GPR43, GPR41, and GPR109a to modulate inflammation, intestinal barrier integrity, glycemic response, energy homeostasis and other host responses. Inflammation is suppressed by SCFAs primarily through inhibition of the NF-κB pathway and/or histone deacetylase function (HDACi) to downregulate pro-inflammatory cytokines such as TNFα, IL-6, IL-12, IFNγ and upregulate anti-inflammatory cytokines such as IL-10, TGF-β in a variety of cells including immune cells such as macrophages in lamina propria. IL-18 expression upregulated by GPR109a contributes to the enhancement of intestinal barrier integrity. SCFAs can also be taken up by enterocytes through the monocarboxylate transporter (MCT) and along with peptide YY (PYY) and glucagon-like peptide-1 (GLP-1) variably stimulates the liver, muscles, pancreas and adipose tissues to influence glycemic response, lipolysis, fatty acid oxidation and gluconeogenesis.
Production of butyrate and propionate by Eubacterium spp. in the gut
Among the SCFAs, propionate, and butyrate are most often considered to benefit human health and are produced by distinct cohorts of the colonic microbiota including several species from the genus EubacteriumCitation57 (). One of the most extensively studied Eubacterium species, E. rectale, was first isolated from the feces of healthy Japanese-Hawaiian males and identified as a major butyrate producer capable of utilizing complex carbohydrates such as cellobiose and starch for growth and proliferation.Citation58 E. hallii, on the other hand, was first reported as a butyrate producer in the human gut by Barcenilla et al. in a 16S rRNA gene-based RFLP study.Citation59 In the gut, butyrate can be produced from carbohydrates via glycolysis where two molecules of acetyl-CoA are combined to form acetoacetyl-CoA and undergo stepwise reduction to produce butyryl-CoA. Two distinct pathways are currently known for the final transformation of butyryl-CoA to butyrate; this proceeds either through the butyryl-CoA:acetate CoA-transferase pathway or the phosphotransbutyrylase and butyrate kinase pathway.Citation60 The butyryl CoA:acetate CoA-transferase route for the final production of butyryl-CoA from acetyl-CoA is shared by E. rectale and the closely related Roseburia species, along with genomic organization of the butyrate synthetic genes. The same pathway is also employed by other Eubacterium species such as E. hallii and E. biforme (now reclassified as Holdemanella biformis) for the production of butyrate.Citation60 Both E. rectale and E. hallii have been subsequently identified as prolific butyrate producers in the gut. Indeed, they were found to be among the 10 most abundant members of the human fecal microbiota, contributing more than 44% of butyryl-CoA:acetate CoA-transferase sequences retrieved from fecal samples of 10 healthy volunteers.Citation61,Citation62 A recent Swiss cohort study has also shown E. hallii to be one of the first producers of butyric acid in the infant gut.Citation63
Propionate can be formed via two pathways from sugar fermentation by gut microbes. While the succinate pathway processes most pentose and hexose sugars to produce propionate, deoxy sugars such as fucose and rhamnose are metabolized by the propanediol pathway. The latter are readily available in the gut environment as dietary (e.g. human milk oligosaccharides) or host-derived (mucin) glycans and upon utilization by a variety of gut microbes can produce 1,2-propanediol as an end product. Although unable to degrade deoxy sugars, 1,2-propanediol can be metabolized by E. hallii, which has been shown to carry the glycerol/diol dehydratase PduCDE, a key enzyme in the transformation of 1,2-propanediol to produce propionate and propanol with the generation of one ATP.Citation64,Citation65 The conversion of 1,2-propanediol to propionate is dependent on the availability of vitamin B12 and occurs within microcompartments called polyhedral bodies to sequester the toxic intermediate, propionaldehyde.Citation66
Although Eubacterium spp. can degrade complex carbohydrates, certain strains of Eubacterium spp. strains may lack the ability to degrade specific complex carbohydrates and rely on metabolites produced by other gut microbes for doing so; fermented products produced by these other gut microbes can then be utilized by Eubacterium spp.Citation57 The importance of cross-feeding mechanisms in SCFA production by Eubacterium spp. have been demonstrated in a number of instances.Citation67–Citation69 In these studies, Eubacterium spp. were co-cultured with Bifidobacterium spp. in the presence of complex carbohydrates. The Bifidobacterium strains, which are capable of degrading complex carbohydrates such as arabinoxylan oligosaccharides and fucosyllactose, were shown to produce acetate, lactate and 1,2-propanediol, all of which were in turn taken up and used by Eubacterium spp. to produce butyrate and propionate. Evidence of such cross-feeding by Eubacterium spp. not only highlights the synergistic interactions between gut microbes and butyrogenic effects of resistant carbohydrates, but also underlines the ecological roles of Eubacterium spp. in the gut environment.
Eubacterium spp. modulate gut inflammation through SCFAs
SCFAs, and especially butyrate has been reported to impart varied beneficial effects on human health.Citation70 Although, the least abundant SCFA produced, butyrate constitutes the primary energy source of colonocytes, promoting their proliferation, maturation, and a healthy colon.Citation71 Indeed, E. rectale has been reported to preferentially colonize the mucus layer, thereby increasing the bioavailability of butyrate for epithelial colonocytes.Citation72 Additionally, SCFAs, have been shown to play a major role in modulation of inflammation in the gut through promotion of intestinal integrity and regulation of immune response. SCFAs can improve transepithelial resistance through upregulation of tight junction proteins such as claudin-1 and occludin as well as the intestinal mucin protein, mucin 2.Citation73,Citation74 Modification of various signaling pathways have been also described to highlight regulation of immune response by SCFAs including activation of G-protein-coupled receptors (GPCRs) and inhibition of histone deacetylases (HDACs).Citation75 SCFAs can bind to at least four discrete GPCRs – FFAR2 (Free fatty acid receptor), FFAR3, GPR109a, and Olfr78 as ligands, albeit with varying specificity.Citation76 For example, butyrate binds preferentially to FFAR3 over FFAR2, which exhibits higher affinities for acetate and propionate.Citation76 FFAR2 is widely expressed in diverse tissues with highest expression in immune cells. Several studies have shown that SCFAs can act as an anti-inflammatory agent through inhibition of pro-inflammatory cytokines such as IFN-γ, IL-1β, IL-6, IL-8, and TNF-α, while upregulating anti-inflammatory cytokines such as IL-10 and TGF-β in a FFAR2/FFAR3 dependent manner.Citation75,Citation77 GPR109a activates the inflammasome pathway in colonic macrophages and dendritic cells, thereby inducing differentiation of regulatory T cells, and anti-inflammatory IL-10 producing T-cells.Citation78 GPR109a activation by SCFAs in intestinal epithelial cells (IECs) can also increase production of IL-18, a key cytokine for repair and maintenance of intestinal epithelial integrity.Citation79 Inhibition of HDAC activity by propionate and butyrate have been associated with the downregulation of expression for pro-inflammatory cytokines and chemokines such as CXCL8 and CCL20 in IECs.Citation80 HDAC inhibition by SCFAs have also been associated with the increase in expression of β-defensins and cathelicidins such as LL-37.Citation81 Given the extensive involvement of SCFAs in modulation of gut health as described, especially butyrate, a dysbiosis of the gut microbiota involving SCFA producers has major implications due to alteration of the SCFA profile in the intestine.Citation82
Inflammatory bowel diseases (IBDs) are severe and chronic inflammations of the gastrointestinal tract and are characterized by two major clinical phenotypes: Crohn’s disease (CD) and ulcerative colitis (UC). CD involves the transmural inflammation of all layers of the epithelial wall, whereas UC only affects the superficial mucosal layer. In general, IBDs recurrently exhibit dysbiosis of the gut microbiota that is characterized by a decrease in the diversity and temporal stability of the microbiota. While the exact role of microbial disturbances in the pathogenesis or causation of IBDs is still being elucidated, the proportion of butyrate producers including Eubacterium spp. in the gut are consistently reduced in IBD subjectsCitation83-Citation85 (). Indeed, a decreased abundance of clostridial clusters IV and XIVa in IBD patients compared to non-IBD, healthy individuals along with a concomitant increase in proteobacterial pathobionts constitute a signature for microbial dysbiosis in IBDs and can be considered as biomarkers.Citation86,Citation95-Citation97 Consequently, a decrease in gut butyrate levels is commonly observed in patients suffering from IBD, leading to improper modulation of the host immune system.Citation98 Decreased levels of SCFA in the gut in IBD and experimental colitis have also been correlated with reduced regulatory T cell functionality and increased inflammation.Citation99,Citation100
Table 1. Case-control studies showing association of Eubacterium spp. with inflammatory bowel diseases (IBDs).
A butyrate-mediated protective effect provided by Eubacterium spp. in IBDs has been demonstrated in several recent studies. In vitro studies using fecal microbiota from UC and CD patients, represented by fewer butyrate producers, exhibited a decreased capacity for colonization and butyrate production; supplementation of the IBD microbiota with known butyrate producers including Eubacterium spp. restored butyrate production and improved epithelial barrier integrity and colonization capacity.Citation87,Citation101 The role of the Eubacterium spp.-butyrate-anti-inflammation axis in gut health was further demonstrated in children suffering from IBD who underwent an anti-TNF-α treatment; patients harboring a higher baseline abundance of E. rectale were more responsive to treatment with the presence of E. rectale being predictive of successful attenuation of inflammation.Citation90 The protective effects of E. limosum, and the SCFAs it produces, in gut inflammation have been demonstrated in in vitro and murine models.Citation102 SCFAs produced by E. limosum induced T84 colonocyte growth and reduced IL-6 and TLR4 expression by the colonocytes when stimulated by TNF-α treatment, with butyrate being the most prominent effector. Additionally, when provided with a 5% E. limosum chow, mice showed significant retention of body weight and colon length compared to the control group upon induction of colitis. These observations exhibit a butyrate-mediated anti-inflammatory effect of Eubacterium spp. on gut health and presents it as an attractive biotherapeutic in inflammatory gut ailments.
Effects of SCFA production by Eubacterium spp. in Type II diabetes mellitus (T2DM) and obesity
The association of Eubacterium spp. with obesity remains controversial so far, with several reports suggesting a positive correlation of Eubacterium spp. with obesity.Citation103–105 BMI is often considered a proxy for adiposity; some BMI-based studies have also reported greater abundances of Eubacterium spp. in obese subjects.Citation106,Citation107 Interestingly, higher levels of total butyrate have been reported in obese individuals with reduced fecal SCFAs in treated obese subjects, which suggests an enhanced assimilation of carbohydrates and lipids that can contribute to the obese phenotype.Citation108–Citation110 Such an observation can explain the higher abundance of butyrate producers including Eubacterium spp. in obese individuals. A closer look at dietary intervention studies indicates that the proportions of Eubacterium spp. and other butyrate producers in obese subjects may be influenced primarily by diet. A significant reduction of Eubacterium spp. is consistently reported in several studies where availability of complex carbohydrates to gut microorganisms have been restricted in obese individuals.Citation15,Citation111,Citation112 A study by Balamurugan et al. in obese and non-obese Indian children exposed to similar diets also did not find any difference in E. rectale abundance between groups.Citation113 Taken together, current evidence indicates that Eubacterium spp. along with other butyrate producers, when maintained in the gut through a consistent availability of reasonable amount of complex carbohydrates, increases in obese individuals in proportion, thereby facilitating energy extraction in the gut. It is also possible that diet rather than altered metabolic parameters in obese individuals, may drive the growth and proliferation of butyrate producers including Eubacterium spp. The exact mechanisms through which the gut microbiota may modulate obesity are still being elucidated. Instances where butyrate have been shown to alleviate diet-induced obesity and improve glucose homeostasis make it difficult to make linear conclusions and provides an incentive for further investigations.Citation114,Citation115 Ultimately, care must be taken while inferring direct associations between taxa and obesity, as such conclusions may be misleadingly oversimplistic for a metabolic syndrome with multifactorial influences.
Eubacterium spp. and butyrate producers have been positively associated with insulin sensitivity in several studies.Citation116,Citation117 Recent independent studies which compared metagenomes from healthy and T2D individuals, have clearly indicated a potential correlation between gut microbiota and T2D pathophysiology.Citation118,Citation119 The studies, carried out in Chinese and European populations, both reported a significant reduction of butyrate producers including Eubacterium spp. in T2D subjects.109,Citation119 Additional studies have demonstrated the restorative effect of butyrate producers, including Eubacterium spp., transplanted from lean individuals, in both human and murine insulin-resistant models.Citation120–Citation122 Indeed, the increase in Eubacterium spp. after FMT was associated with metabolic improvement in insulin-resistant individuals.Citation120 When orally administered to obese and insulin-resistant db/db mice, E. hallii have been shown to significantly improve insulin sensitivity and energy metabolism.Citation121 The stimulation of gut hormones and inhibition of food intake by SCFAs have been proposed as possible mechanisms of modulation of host metabolism by gut microbiota in T2D individuals.Citation123 Such a proposed mechanism is consistent with the observation that butyrate and propionate bound to FFAR2 receptor can regulate satiety hormones such as ghrelin (orexigenic peptide), glucagon-like peptide-1 (GLP-1), and peptide YY (PYY) (anorexigenic peptide)Citation124 (). Ghrelin, also known as the ‘hunger hormone’, stimulates appetite and is secreted before a meal, while GLP-1 and PYY are synthesized and released by enteroendocrine L cells and stimulate insulin secretion by pancreatic β cells, reduces food intake, and normalizes energy intake and weight loss. An opposite regulation of ghrelin and GLP-1/PYY by SCFAs, where GLP_1/PYY are upregulated and ghrelin is downregulated, ensures reduced food intake, satiety and reduced adiposity.Citation125 Ghrelin, has also been negatively associated with the butyrate-producing E. rectale.Citation126 Recent evidence from Zeevi et al., who performed a machine learning-based study on a large cohort (n = 800) in order to predict personalized postprandial glycemic response for individuals using an integrated feature dataset derived from dietary habits, gut microbiota, anthropometrics, physical activity, and blood parameters, also supports an affirmative role of Eubacterium spp. in insulin sensitivity.Citation127 In their study, 72 features from the gut microbiome were inferred to be predictive, among which E. rectale was reported to be one of the most robust with a higher abundance of the bacterium in the gut being positively associated with lower postprandial glycemic response (n = 430). Butyrate produced by Eubacterium spp. can also provide additional benefits to T2DM patients through HDAC inhibition-mediated pancreatic β-cell reprogramming to improve insulin sensitivity and satiety.Citation128 Finally, low-grade inflammation has been reported in T2DM, where inflammatory molecules are upregulated in insulin target tissues and contribute to insulin resistance.Citation129 For example, TLR4-dependent increase in production of pro-inflammatory cytokines through activation of macrophages and β-cells in pancreatic islets leads to dysregulation, functional impairment, and decreased viability of β-cells.Citation130 SCFAs produced by Eubacterium spp. can contribute to restoration of physiological inflammatory environments through mechanisms detailed above. Such a connection is also reinforced by the consistent decrease of other gut butyrate producers in T2DM.Citation116,Citation117 Current observations therefore consistently indicate Eubacterium spp. as a positive contributor in alleviating T2DM and should be considered as a potential therapeutic.
Butyrate-mediated contribution of Eubacterium spp. in inhibition of colorectal cancer and atherosclerosis
Dysbiosis of the gut microbiota is closely associated with incidence of various cancers including colorectal cancer. While chronic inflammation and reduced immune response resulting from dysbiosis has been reported to contribute to increased cancer incidence, commensal bacteria have been demonstrated to increase immune surveillance and decrease cancer incidence.Citation131,Citation132 Dietary fibers have been associated with lower risks of intestinal cancer development; this is primarily due to the strong anti-cancer effect of butyrate.Citation133–Citation136 Through modulation of various signaling pathways involved in cell survival and apoptosis, the anti-cancer activity of butyrate has been demonstrated in cancer cells and mouse models.Citation137,Citation138 Butyrate, while being the preferred energy source for colonocytes, is poorly metabolized in cancer cells due to the Warburg effect. This leads to cytoplasmic accumulation and subsequent translocation of butyrate into the nucleus where it acts as a HDAC inhibitor and negatively modulates PI3K/Akt and JAK2/STAT signaling pathways, resulting in inhibition of carcinogenesis and increased cancer cell apoptosis.Citation138 Additionally, inhibition of signaling pathways such as NF-κB and HIF-1 by butyrate have been reported to increase anti-cancer immune responses.Citation139 Interestingly, the anti-carcinogenic effect of butyrate is dose-dependent; a lower concentration of the butyrate (0.5–1 mM) promotes growth of non-cancerous colonocytes and apoptosis in cancerous ones, whereas at higher concentrations (greater than 2 mM) it can cause apoptosis in both.Citation140,Citation141 Indeed, butyrate induces proliferation of colonocytes at the cript of the colon, where its concentration is lower, but shows a pro-apoptotic effect closer to the lumen where its concentration increases; this also ensures normal turnover of cells in the intestine.Citation142
Butyrate producers including Eubacterium spp. are decreased in abundance in patients suffering from CRC.Citation143 Indeed, gut microbiomes in CRC patients are less fermentative in nature with significantly decreased abundance of butyrate fermenters from Clostridium cluster XIVa such as Eubacterium sp. and Roseburia sp.; depletion of the butyryl-CoA transferase in CRC subjects have also been reported.Citation136,Citation143 Among Eubacterium spp., E. rectale, E. hallii and E. ventriosum are reported to be significantly reduced in abundance in the gut of individuals with CRCCitation136,Citation143-Citation145 (). E. ventriosum has been proposed as biomarker for low risk of CRC, with significant enrichment in healthy individuals compared to CRC patients in diverse populations.Citation145 Additionally, butyrate levels in the colon share an inverse relationship with the incidence of CRC.Citation148,Citation149 This can be attributed to gut dysbiosis in patients with CRC, where butyrate-producing capacity of the gut microbiota is significantly reduced.Citation150,Citation151 Such change in gut microbiota can be caused by a consistently lower intake of dietary fibers and consequent-decreased levels of SCFAs, as often observed in individuals with CRC.Citation136 In the absence of butyrate, the intestinal tract can reach a state of chronic inflammation, that contributes to development and progression of CRC.Citation152,Citation153 Such consistent observations suggest there is merit in investigating the use of Eubacterium spp. strains as therapeutic interventions in CRC and related diseases. Indeed, Feng et al. have been granted patent rights for the use various strains of E. ventriosum and E. eligens in treating colitis and/or CRC.Citation154
Table 2. Case-control studies showing association of Eubacterium spp. with colorectal cancer and atherosclerosis.
Recent studies in humans and mice show that butyrate producers in the gut including Eubacterium spp. are negatively associated with atherosclerotic cardiovascular disease (ACVD).Citation146,Citation147,Citation155 Deep analysis of gut metagenomes from atherosclerotic subjects show a depletion of butyrate producers such as Eubacterium spp., Roseburia spp. and F. prausnitzii, compared to healthy individualsCitation146,Citation147 (). The gut environment in ACVD subjects have consequently been reported to be less fermentative and inflammatory in nature.Citation146 Notably, peptidoglycan (PG) and lipopolysaccharide (LPS) are pro-inflammatory, microbial pathogen-associated molecular patterns (PAMPs) that are recognized as risk factors in cardiovascular diseases (CVDs).Citation156 PG biosynthesis genes are enriched in ACVD metagenomes, which indicates greater peptidoglycan production that can lead to priming of the innate immune system and inflammation.Citation147 PG has also been observed in atherosclerotic plaques, while patients with a high CVD burden exhibit greater risk from circulating endotoxemia.Citation156 Interestingly, network analysis of ACVD gut microbiomes have revealed that microbes enriched in ACVD have a mutually exclusive relationship with butyrate producers including Eubacterium spp., thereby suggesting consistency of dysbiosis in ACVD patients that is represented by a depletion of butyrate producers.Citation146 Indeed, butyrate producers have been proposed to be protective against atherosclerosis.Citation155 In atherosclerotic mice, negative association of Eubacterium spp. and other butyrate producers with plasma cholesterol, MMP-9 and A-FABP (biomarkers for cardiovascular pathologies) have been reported.Citation155 It currently remains unclear if a dysbiotic gut is a read-out of atherosclerotic symptoms or vice-versa. However, since ACVD are manifestations of several factors such as lifestyle, diet and genetics, it is possible that lifestyle and diet may primarily contribute to a dysbiotic microbiota, which in turn may aggravate atherosclerotic development. Depletion of butyrate producers including Eubacterium spp. in the gut leads to reduced barrier integrity and translocation of pro-inflammatory microbial components such as PG and LPS across the intestinal epithelium. Inflammatory responses triggered by TLR4 bound by circulating LPS, primarily through the NF-κB pathway and the subsequent release of pro-inflammatory cytokines such as TNF-α, IL-6, IL-1, and IL-27, promotes the development of atherosclerosis.Citation156 Additionally, nucleotide-binding oligomerization domain-containing protein 1 (NOD1) and NOD2 can bind circulating PG and activate the NF-κB pathway to trigger inflammatory responses. Indeed, NOD1/2 knockouts in murine models have revealed these receptors as critical in maintaining intestinal barrier integrity and development of atherosclerosis.Citation156 Butyrate producers including Eubacterium spp., therefore, may play an important role in the gut-heart axis; they can restore a dysbiotic gut microbiota and modulate inflammation in ACVD subjects and merit further exploration as potential therapeutics.
Transformation of cholesterol by Eubacterium spp. provides protection against cardiovascular diseases
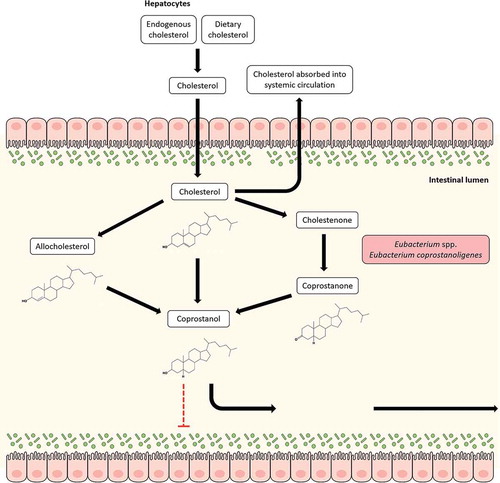
Conversion of cholesterol to coprostanol by intestinal bacteria was first reported in the 1930s and several studies have been carried out since to identify bacteria capable of transforming cholesterol to coprostanol. Many of the identified microbes were eventually assigned to the genus Eubacterium; Eubacterium coprostanoligenes HL (ATCC 51222) represents one such bacteria that was isolated from a hog sewage lagoon and has received considerable attention due to its cholesterol-reducing properties.Citation157 Although subsequently Bacteroides dorei, Lactobacillus sp. and Bifidobacterium sp. have been reported to have cholesterol utilization properties, these seem to be transient properties and may be lost, making E. coprostanoligenes HL the only available culturable gut isolate able to degrade cholesterol.Citation158 The presence of E. coprostanoligenes in the gut microbiota has been strongly associated with fecal coprostanol.Citation158 Recently, 3β-hydroxysteroid dehydrogenase homologs of E. coprostanoligenes have been identified in gut metagenomes that can transform cholesterol to coprostanol.Citation158 Interestingly, these intestinal sterol metabolism A genes (ismA) have been attributed to yet uncultured gut microbes which formed a coherent clade with E. coprostanoligenes in the tree of life and may represent novel Eubacterium species involved in cholesterol reduction in the gut.Citation158 The mechanism of cholesterol to coprostanol conversion has been investigated with three major proposed pathways and Eubacterium spp. have been found to be involved in all of these (). The first pathway involves a direct, stereospecific reduction of the 5,6-double bond in cholesterol,Citation159 while the second is an indirect pathway which includes at least three steps. The latter pathway, which has been demonstrated in E. coprostanoligenes HL, requires NADP+ and proceeds through the production of cholestenone and coprostanone intermediates.Citation158–Citation160 An additional third pathway has also been identified which involves isomerization of cholesterol to allocholesterol, which can be reduced to coprostanol by Eubacterium ATCC21-403 and 408 species.Citation161 The final pathway is, however, poorly studied.
Figure 3. Cholesterol metabolism by Eubacterium coprostanoligenes in the gut. Cholesterol can reach the gut from two sources: endogenous (synthesized in the liver) or exogenous (in the form of dietary uptake). Cholesterol can be reabsorbed from the gut. The cholesterol that is not reabsorbed can be metabolized by Eubacterium coprostanoligenes to coprostanol both directly and indirectly through the intermediate, coprostanone. It can also reduce cholesterol to coprostanol upon epimerization to allocholesterol through a pathway that remains poorly studied. Unlike cholesterol, coprostanol is taken up poorly in the intestine and most of it is excreted in feces, thereby providing a route for cholesterol removal from the gut and systemic circulation.
Nearly one gram of cholesterol from dietary and extra-dietary sources reach the human colon daily, where it is metabolized by commensal gut bacteria to coprostanol. Unlike cholesterol, coprostanol is poorly absorbed in the intestine, and is suggested to have an impact on modulation of cholesterol metabolism and serum cholesterol levels.Citation162 This notion has been reinforced by findings that an inverse relationship exists between plasma cholesterol levels and the ratio of cholesterol to coprostanol in the feces.Citation163 Cholesterol conversion to coprostanol has been therefore considered as a new strategy for management of cholesterol homeostasis in humans. As an extension, Eubacterium spp., which are highly involved in coprostanol metabolism in the gut have been investigated for their hypocholesterolemic effects. Li et al. reported a reduction in the plasma cholesterol levels and an increase in the coprostanol/cholesterol ratios in the digestive contents of hypercholesterolemic rabbits that were fed E. coprostanoligenes.Citation164 The effects observed in these rabbits were further ascribed to cholesterol reduction by E. coprostanoligenes due to its preferential colonization in the jejunum and ileum, both of which are sites for cholesterol absorption. Similar observations have also been reported in germ-free mice.Citation165 Additional results from a combined metabolomic and metagenomic study have identified multiple bacterial phylotypes including Eubacterium eligens ATCC 27750 (p = 1.477e-02) to be significantly correlated to high fecal coprostanol.Citation166
Atherosclerotic cardiovascular diseases (CVDs) are widely recognized as a major public health concern, where key risk factors in their development include an imbalance in blood cholesterol levels and high serum concentrations of low-density lipoprotein cholesterol.Citation167 Indeed, patients with ACVDs have higher cholesterol absorption in the gut.Citation158,Citation168 Notably, changes in the gut microbial community have been directly correlated to the rate of cholesterol converted to coprostanol, while a high efficiency of cholesterol transformation to coprostanol has been linked to a reduced risk of CVDs.Citation158,Citation169,Citation170 Due to their hypocholesterolemic effect, Eubacterium spp. and other cholesterol-reducing microbes can provide protection against CVDs. Indeed, gut Eubacterium spp. in atherosclerotic subjects show a significantly negative correlation with established markers of atherosclerosis such as low-density lipoproteins, cholesterol and white blood cells.Citation147 E. coprostanoligenes were also reduced in the murine gut when mice were fed a methionine-choline diet to induce nonalcoholic steatohepatitis, where damage to the liver inhibits the production of endogenous cholesterol.Citation171 Furthermore, cholesterol-reducing homologs of E. coprostanoligenes 3β-hydroxysteroid dehydrogenase identified in metagenome-assembled genomes from gut metagenomes have been associated with lower levels of serum cholesterol; the ismA genes were also correlated to higher levels of coprostanol and lower levels of cholesterol in stool.Citation158 The reduction of cholesterol to coprostanol still remains poorly understood and only few studies on cholesterol metabolizing bacteria are currently available. Greater investigative investment is necessary to garner a holistic understanding of the molecular mechanisms behind cholesterol-coprostanol metabolism in the gut, including Eubacterium spp., and to perfect hypocholesterolemic strategies.
Eubacterium spp. contribute to gut and hepatic health through modulation of bile acid metabolism
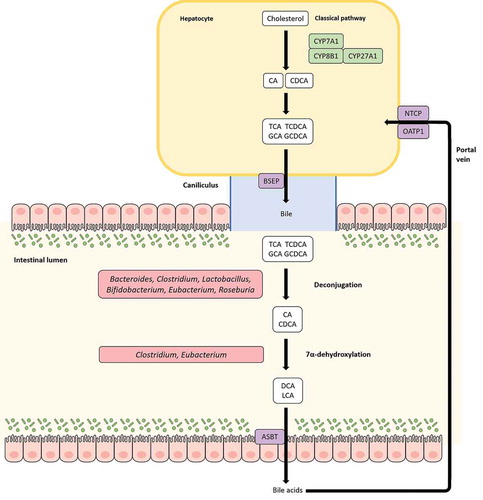
Bile acids (BA) are host-produced metabolites derived from cholesterol in liver pericentral hepatocytes. Cholic acid (CA) and chenodeoxycholic acid (CDCA) are the primary BAs produced in liver which are then conjugated to taurine or glycine before being temporarily stored in the gallbladder; these BAs subsequently undergo postprandial secretion to reach the gut. 95% of the total BA pool in the gut are absorbed efficiently and recycled back to the liver via the portal vein; this cyclic process is known as enterohepatic circulation. The rest serves as a substrate for bacterial metabolism in the gut and constitutes a critical route for cholesterol excretion from the body. BAs can occur in several forms including primary BA, secondary BA, conjugated, or unconjugated. Various members of the gut microbiota are capable of transforming BAs, thereby influencing the composition of the local BA pool along with various other aspects of host physiology. Gut microbes including Eubacterium spp. that possess the enzyme bile salt hydrolase (BSH) are able to hydrolyze the C-24 N-acyl amide bond in conjugated BAs to release glycine/taurine moietiesCitation121 (). Indeed, Eubacterium spp. along with other genera such as Roseburia and Clostridium constitute a major reservoir of BSHs in the gut.Citation172 Deconjugation increases the pKa of BAs to ~5, thereby making them less soluble which in turn leads to inefficient absorption and replenishment of the lost BA by de novo synthesis from cholesterol.Citation173 Additionally, BSH activity can disrupt micelle formation and absorption, resulting in a significant reduction of cholesterol levels.Citation159 Being reasonably widely distributed in the gut microbiota, BSH activity can thus be modulated to regulate weight gain and cholesterol levels in the host. Deconjugation also helps in bile detoxification through recapture and export of cotransported protons by the free BAs generated, thereby negating the pH.Citation174 Another way intestinal bacteria can transform BAs is through the oxidation and epimerization of hydroxyl groups at C3, C7, and C12 positions, resulting in the generation of isobile (β-hydroxy) salts.Citation175 Epimerization involves the reversible stereochemical change from α to β configuration and vice versa, generating a stable oxo-bile acid intermediate. This process is catalyzed by α- and β-hydroxysteroid dehydrogenases (HSDHs) and can be carried out by a single bacterial species containing both enzymes or through proto-cooperation between two species, with each contributing one enzyme. HSDH activity has been reported in several species including Eubacterium spp.Citation176
Figure 4. Bile acid (BA) modification by Eubacterium spp. and enterohepatic circulation. BAs are produced from cholesterol in the liver and are continually released into the bile canaliculi via the bile salt export pump (BSEP). The bile canaliculi drain into the gallbladder where BAs are temporarily stored and undergo postprandial release into the gut. Before release into the bile canaliculi, cholic acid (CA) and chenodeoxycholic acid (CDCA), the primary BAs produced in liver hepatocytes, can be conjugated to taurine/glycine moieties to form conjugated BAs (T/G-CA, T/G-CDCA). In the gut, primary BAs can be metabolized by gut bacteria including Eubacterium spp. into diverse secondary forms. BAs can undergo deconjugation to form deconjugated primary BAs and/or hydroxylation reactions to produce secondary BAs such as deoxycholic acid (DCA) and lithocholic acid (LCA). 95% of BAs are reabsorbed in the gut and recycled back to the liver through the portal vein, with conjugated BAs exhibiting highest rates of reabsorption. This circular movement of BAs from liver hepatocytes to the gut and back to the liver is known as the enterohepatic circulation.
Bacterial 7α-dehydroxylases in the gut convert primary BAs, CA and CDCA into deoxycholic acid (DCA) and lithocholic acid (LCA), respectively ().Citation173 Although quantitatively, 7α-hydroxylation represents the most important bacterial transformation of BAs in the gut, only few distinct members of the gut microbiota such as Eubacterium and Clostridium XIVa cluster have been reported to be capable of carrying out this reaction.Citation173,Citation177 Studies on Eubacterium strain VPI 12708 have identified enzymes encoded by the bile acid inducible (bai) operon which catalyze a multistep pathway for primary BA 7α-dehyroxylation.Citation178 DCA and LCA produced through 7α-dehyroxylation of primary BAs by Eubacterium spp. can have major impacts on gut health and homeostasis that are manifested primarily through bile acid signaling receptors. Both DCA and LCA are high-affinity ligands for the nuclear hormone receptor, farnesoid X receptor (FXR); activation of intestinal FXR by DCA or LCA upregulates the expression of the fibroblast growth factor 19 (FGF19), which in turn binds to the hepatic fibroblast growth factor receptor 4 (FGFR4) to subsequently downregulate bile acid synthesis in hepatocytesCitation179 (). FXR also promotes anti-inflammatory properties, primarily through inhibition of the NF-κB pathway and BA detoxification, through modulation of proliferator-activated receptor α (PPARα).Citation179 FXR activation has also been reported to induce expression of antimicrobial peptides, thereby contributing to control of pathobionts.Citation173,Citation180 TGR5, a GPCR that activates various intracellular pathways upon interaction with BAs, also binds LCA and DCA with the highest affinity in the BA pool (). Once activated, TGR5 stimulates the secretion of incretin hormone GLP-1 and insulin, thereby promoting energy expenditure.Citation181 Additionally, TGR5 can modulate inflammatory responses, which can be both pro- or anti-inflammatory in nature; BA-TGR5 signaling plays a critical role in the intricate balance of pro- and anti-inflammatory cytokines in the gut.Citation179 LCA and DCA also bind to the pregnane-X receptor (PXR), Vitamin D3 receptor (VDR) and constitutive androstance receptor (CAR) to variously influence BA homeostasis and BA detoxification.Citation179 The strong affinity of bile acid signaling receptors for microbiota-induced secondary BAs highlight how the gut microbiota including Eubacterium spp. can modulate BA homeostasis, BA detoxification, control and maintenance of bacterial growth in gut, inflammation and glycemic responses through BA signaling. BA metabolism by a healthy gut microbiome also provides protection against C. difficile infection (CDI). DCA, which predominates in feces under healthy conditions compared to CDI subjects, can stimulate germination of C. difficile spores, but importantly, inhibits the vegetative form of Clostridium difficile.Citation182 Dysbiosis of the gut leading to a decrease of secondary BA-producing bacteria and correlated with an increase in fecal primary BAs is permissive to the germination of C. difficile spores culminating in CDI.Citation183,Citation184 Indeed, restoration of gut BSH activity contributes to the efficacy of fecal microbiota transplantation (FMT) therapies in CDI patients.Citation185
Figure 5. Bile acid (BA) induced signaling pathways influence BA homeostasis and inflammation. BAs in the gut are taken up by enterocytes via the apical sodium-bile acid transporter (ASBT) and bind to the farnesoid X receptor (FXR) which in turn upregulates the expression of the fibroblast growth factor 19 (FGF19). FGF19 can then bind to FGF receptor 4 in hepatocytes to downregulate BA synthesis in liver through the JNK/ERK pathway. Additionally, BAs transported through the portal vein can inhibit BA synthesis in hepatocytes in a FXR-mediated manner by entry through the organic anion transporting polypeptide 1 (OATP1) or sodium-taurocholate cotransporting polypeptide (NTCP) and upregulating the BA synthesis inhibiting transcription factor small heterodimer protein (SHP). FXR can also influence BA homeostasis through the peroxisome proliferator-activated receptor alpha (PPARα). LCA and DCA produced by Eubacterium spp. are high-affinity ligands for TGR5, which upon binding of said BAs can modulate glycemic response, immune response, BA homeostasis and BA detoxification in various tissues.
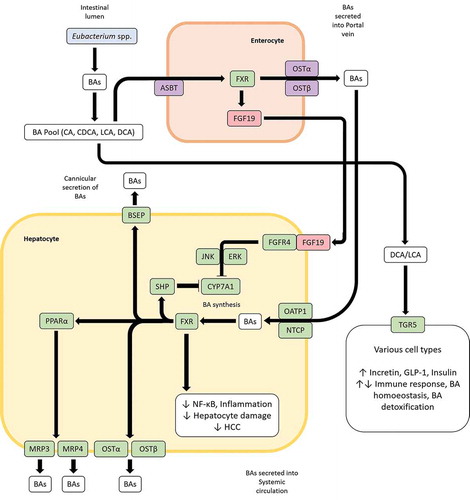
The gut microbiota, as modulated by diet and other factors can lead to a particular BA profile which in turn has important consequences. A high-fat diet (HFD) such as the Western diet overstimulates BA discharge into the intestine, leading to a dysbiotic gut microbiota and increased secondary BA production, especially LCA and DCA.Citation179 DCA and LCA are the most hydrophobic among the BA pool and elevated levels can be cytotoxic; detrimental effects exerted by DCA and LCA can disrupt the architecture and function of the colonic epithelium through oxidative damage to DNA, triggering of pro-inflammatory responses and increased cell proliferation. In HFD-fed mice, increased LCA/DCA was correlated to an increase in the abundance of Clostridum sordellii, a bacterium from Clostridium cluster XI.Citation186 Surprisingly, Clostridium cluster XIVa to which Eubacterium spp. belongs was reported as a minor contributor, even though they exhibit 7-α hydroxylation properties. Such an observation is consistent specifically for Eubacterium spp., which is negatively modulated by HFDs, as mentioned above. The greater reabsorption of secondary BAs in the intestine resulting from HFDs and subsequent transport to the liver causes hepatic inflammation.Citation179 A reduced FXR signaling due to increased inflammation results in decreased hepatic BA transporter function and increased BA sequestration in the liver; this can establish sustained hepatic inflammation, which can eventually lead to hepatocellular carcinoma (HCC).Citation180 Dysbiosis in liver disorders such as HCC, fatty acid liver disease (FLD), fibrosis and cirrhosis is additionally characterized by an elevation of aerobic, pro-inflammatory, BSH-rich bacteria such as Enterobacter and Enterococcus, which also contribute to an increased production of secondary BAs.Citation150 Indeed, the ratio between primary and secondary BAs in feces and the levels of conjugated and unconjugated BAs in serum are higher in nonalcoholic FLD (NAFLD) patients.Citation187 Eubacterium spp. is consistently found in lower proportions in liver disorders. Metagenomic shotgun sequencing of the gut microbiome of subjects suffering from fibrosis and cirrhosis has revealed a significant reduction of Eubacterium species such as E. rectale, E. hallii and E. eligens compared to healthy individualsCitation146,Citation188-Citation190 (). These metagenomes also tended to be less fermentative in nature, i.e. displayed lower abundances for fermentative butyrate producers such as Roseburia sp., Faecalibacterium sp. and others, besides Eubacterium spp.Citation146, 179
Table 3. Case-control studies showing association of Eubacterium spp. with liver diseases.
Secondary bile acids as produced by the gut microbiota may also play a critical role in the development and establishment of CRC. As mentioned above, butyrate inhibits colorectal carcinogenesis and a marked reduction of butyrate producers in the gut including Eubacterium spp. is commonly observed in patients with CRC. Several butyrate producers including Eubacterium spp., which belong to Clostridium cluster XIVa can additionally produce secondary BAs through 7-α hydroxylation of primary BAs.Citation173 In IBD subjects with chronic inflammation of the gut, significantly lower levels of secondary BAs with concurrently increased fecal-conjugated BAs and a marked decrease of Clostridium cluster XIVa is reported.Citation87,Citation101,Citation173,Citation191 A reduction in secondary BA levels contribute to a loss of the anti-inflammatory effects of secondary BAs on intestinal epithelial cells, thereby enhancing the chronic inflammation.Citation191 Even though the loss of butyrate producers and secondary BA producers in IBD have been made separately, the two groups share significant overlap and both are depleted in chronic inflammation of the gut.Citation87,Citation101,Citation150,Citation173,Citation191 Indeed, a recent bioinformatic analysis of gut metagenomes has revealed significantly decreased populations of butyrate producers F. prausnitzii and E. rectale in IBD patients.Citation192 Understandably, an absence of this group of butyrate and secondary bile acid-producing bacteria that includes Eubacterium spp. promotes the development of IBD and its eventual progression to CRC, where a similar dysbiotic gut microbiome is observed.Citation136,Citation152,Citation153 Indeed, modulation of bile acid profiles and/or gut microbiota are being pursued as novel therapeutic approaches for HCC and CRC.
Eubacterium spp. are involved in critical metabolic transformations in the gut
Metabolic transformations of specific compounds in the gut by the resident microbiota can be critical to human health. Substances can be taken up in the intestine that cannot be detoxified or broken down by the human body and thus, can result in toxicogenic effects. Eubacterium spp. have been shown to be capable of carrying out important metabolic transformations in the gut with positive effects on human health including detoxification of toxic compounds into much more benign forms. Multiple beneficial transformations by E. hallii were recently reported by Fekry et al. In their study, Fekry et al. found E. hallii to be highly proficient in the transformation of a highly abundant food-derived heterocyclic aromatic amine carcinogen – 2-amino-1-methyl-6-phenylimidazo(4,5-b)pyridine (PhIP) into a biologically unavailable form – 7-hydroxy-5-methyl-3-phenyl-6,7,8,9-tetrahydropyrido[3′,2′:4,5]imidazo [1,2-α]pyrimidin-5-ium chloride (PhIP-M1).Citation193 Additionally, PhIP transformations by E. hallii in the presence of simulated proximal and distal colon microbiota led to a 300-fold and 120-fold increase in its abundance, respectively, indicating great potential for use as a protective agent. In the same study, Fekry et al. also observed E. hallii to be capable of metabolizing glycerol to 3-hydroxypropionaldehyde (3-HPA), which exists as reuterin in aqueous solutions. Interestingly, reuterin has been shown to have inhibitory effects against Gram-positive and Gram-negative bacteria, fungi and yeast, possibly through increasing oxidative stress by modulating intracellular glutathione, thereby making it an attractive target for therapeutics.Citation194
In another instance, the transformation of 8-prenylanringenin (8-PN) from isoxanthohumol (IX) by gut microbes was investigated by Possemiers et al.Citation195 8-PN is known as a potent phytoestrogen and has been used to alleviate symptoms of menopause.Citation196 Production of 8-PN from IX, found commonly in hops and beers, has been found to be highly variable between individuals.Citation197 In their study, Possemiers et al. carried out supplementation of E. limosum, a bacterium known to carry out the transformation from IX to 8-PN and found that germ-free rats could indeed be induced by E. limosum to produce greater levels of 8-PN from IX upon transplantation of the microbiota from low 8-PN producing individuals. This probiotic effect of E. limosum requires further investigation if it can be applied to humans, especially with respect to the potential for variations in duration of effect and between individuals in terms of colonization efficiencies and other factors. The metabolic transformations described above add greatly to our understanding of the diverse array of benefits humans derive from gut Eubacterium spp. besides production of SCFAs. However, as highlighted already, further research is necessary to truly exploit all the potential benefits the Eubacterium genus has to offer.
Conclusion
The genus Eubacterium is a phylogenetically diverse group of microbes, a fact that makes associated taxonomic assignments challenging. Regardless, many current and former members of this genus exhibit compelling associations with gut health, and, as a major butyrate producer and core gut microbiota component, are immensely important. In this review, we have discussed how Eubacterium spp. is involved in various aspects of gut health through important contributions in SCFA, cholesterol and bile acid metabolism in the gut; we have also elaborated the phylogenetic characteristics of the genus and how it is modulated in the gut by diet and age. In the process we have outline how Eubacterium spp. play a major role in modulation of inflammation, regulation of immune responses, maintenance of barrier integrity in the gut, moderating glycemic response, and cholesterol homeostasis, among others. Strong correlations with beneficial effects in several clinical conditions have prompted further interest in the genus, with multiple species being considered for commercial endeavors as next generation probiotics/biotherapeutics.Citation154,Citation198-Citation201 Most notably, efforts are underway at Caelus Health, in collaboration with Danish bioscience firm Chr. Hansen, to create oral formulations containing E. hallii strains as a biotherapeutic to reduce insulin resistance in individuals with metabolic syndromes and to prevent the development of T2DM.Citation198 Given that the gut is a highly competitive and functionally non-redundant environment, recurring associations of Eubacterium spp. with positive clinical phenotypes combined with a simultaneous resolution of its modes of action establishes a consensus on its positive influence on human health. However, further studies are required to attribute causality to observed associations, i.e., understanding pathogenesis of clinical conditions with respect to gut microbiota. In what remains a major caveat in gut microbiology research today, our understanding of how much the gut microbiota – across all relevant species – influences a clinical condition and vice versa is still limited. Longitudinal studies with tightly controlled diet regimens where the gut microbiota and relevant health parameters are evaluated over protracted time periods may be necessary to elaborate such causalities. Even then, attribution of causation to specific species may prove to be difficult due to the tightly clustered functional niches in the gut.
A recurring motif suggests that in several clinical conditions, especially metabolic syndromes, diet, lifestyle, and other factors can induce the dysbiosis of the gut microbiota, which in turn creates an undesirable metabolic profile. The change in the effective proportions of these metabolites which, directly or indirectly modulate inflammation, barrier integrity, energy homeostasis, and so on, plays an important role in the development and progression of disease pathogenesis. Given the complexity of the processes involved, host-metabolite-microbiota crosstalk must be approached from a system biology standpoint using technologies such as metagenomics and metabolomics. It may be necessary to study involved components together and not in isolation, with therapeutic solutions aimed at modulation of all these components. To this end, further in vitro and in vivo characterization of Eubacterium spp. at the genomic, metagenomic and eventually at the ecological level is required. This will allow us to better understand how the relatively understudied Eubacterium spp. interacts with other members of the gut microbiome and how they are modulated by host factors and diet. Garnering such an understanding is crucial to the successful control and prevention of clinical conditions using clusters of commensal bacteria producing critical metabolites, as evidenced by Geirnhaert et al.Citation101 Much remains to be understood about the metabolic activities, immunomodulatory influences, and ecological role of Eubacterium spp., both in isolation and in combination, with other potential next-generation health-promoting microorganisms such as Akkermansia muciniphila and F. prausnitzii, to ensure its effective deployment in evidence-based gut therapeutics. However, based on evidence to date, there is a lot of cause for optimism.
Disclosure of potential conflicts of interest
No potential conflicts of interest were disclosed.
Additional information
Funding
References
- The Integrative HMP (iHMP) Research Network Consortium. The integrative human microbiome project. Nature. 2019;569:641–28.doi:10.1038/s41586-019-1238-8.
- Bäckhed F, Ley RE, Sonnenburg JL, Peterson DA, Gordon JI. Host-bacterial mutualism in the human intestine. science. 2005;307:1915–1920. doi:10.1126/science.1104816.
- Brunkwall L, Orho-Melander M. The gut microbiome as a target for prevention and treatment of hyperglycaemia in type 2 diabetes: from current human evidence to future possibilities. Diabetologia. 2017;60:943–951. doi:10.1007/s00125-017-4278-3.
- Douillard FP, de Vos WM. Biotechnology of health-promoting bacteria. Biotechnol Adv. 2019. doi:10.1016/j.biotechadv.2019.03.008.
- Wade WG. The genus Eubacterium and related genera. Prokaryotes. 2006;4:823–835.
- Ayeni FA, Biagi E, Rampelli S, Fiori J, Soverini M, Audu HJ, Cristino S, Caporali L, Schnorr SL, Carelli V, et al. Infant and adult gut microbiome and metabolome in rural bassa and urban settlers from Nigeria. Cell Rep. 2018;23(10):3056–3067. doi:10.1016/j.celrep.2018.05.018.
- Gomez A, Petrzelkova KJ, Burns MB, Yeoman CJ, Amato KR, Vlckova K, Modry D, Todd A, Jost Robinson C, Remis M, et al. Gut microbiome of coexisting BaAka pygmies and bantu reflects gradients of traditional subsistence patterns. Cell Rep. 2016;14(9):2142–2153. doi:10.1016/j.celrep.2016.02.013.
- Barrett HL, Gomez-Arango LF, Wilkinson SA, McIntyre HD, Callaway LK, Morrison M, Dekker Nitert M. A vegetarian diet is a major determinant of gut microbiota composition in early pregnancy. Nutrients. 2018;10(7):890. doi:10.3390/nu10070890.
- Mueller S, Saunier K, Hanisch C, Norin E, Alm L, Midtvedt T, Cresci A, Silvi S, Orpianesi C, Verdenelli MC. Differences in fecal microbiota in different European study populations in relation to age, gender, and country: a cross-sectional study. Appl Environ Microbiol. 2006;72(2):1027–1033. doi:10.1128/AEM.72.2.1027-1033.2006.
- Das B, Ghosh TS, Kedia S, Rampal R, Saxena S, Bag S, Mitra R, Dayal M, Mehta O, Surendranath A, et al. Analysis of the gut microbiome of rural and urban healthy indians living in sea level and high altitude areas. Sci Rep. 2018;8(1):10104. doi:10.1038/s41598-018-28550-3.
- Obregon-Tito AJ, Tito RY, Metcalf J, Sankaranarayanan K, Clemente JC, Ursell LK, Zech Xu Z, Van Treuren W, Knight R, Gaffney PM, et al. Subsistence strategies in traditional societies distinguish gut microbiomes. Nat Commun. 2015;6(1):6505. doi:10.1038/ncomms7505.
- Escobar JS, Klotz B, Valdes BE, Agudelo GM. The gut microbiota of Colombians differs from that of Americans, Europeans and Asians. BMC Microbiol. 2014;14:311. doi:10.1186/s12866-014-0311-6.
- Yassour M, Lim MY, Yun HS, Tickle TL, Sung J, Song Y-M, Lee K, Franzosa EA, Morgan XC, Gevers D, et al. Sub-clinical detection of gut microbial biomarkers of obesity and type 2 diabetes. Genome Med. 2016;8(1):17. doi:10.1186/s13073-016-0271-6.
- Jones RB, Alderete TL, Kim JS, Millstein J, Gilliland FD, Goran MI. High intake of dietary fructose in overweight/obese teenagers associated with depletion of Eubacterium and Streptococcus in gut microbiome. Gut Microbes. 2019;10:712–719. doi:10.1080/19490976.2019.1592420.
- Turpin W, Espin-Garcia O, Xu W, Silverberg MS, Kevans D, Smith MI, Guttman DS, Griffiths A, Panaccione R, Otley A, et al. Association of host genome with intestinal microbial composition in a large healthy cohort. Nat Genet. 2016;48(11):1413–1417. doi:10.1038/ng.3693.
- Almeida A, Mitchell AL, Boland M, Forster SC, Gloor GB, Tarkowska A, Lawley TD, Finn RD. A new genomic blueprint of the human gut microbiota. Nature. 2019;568(7753):499–504. doi:10.1038/s41586-019-0965-1.
- Karcher N, Pasolli E, Asnicar F, Huang KD, Tett A, Manara S, Armanini F, Bain D, Duncan SH, Louis P. Analysis of 1321 Eubacterium rectale genomes from metagenomes uncovers complex phylogeographic population structure and subspecies functional adaptations. Genome Biol. 2020;21:138. doi:10.1186/s13059-020-02042-y.
- Gibson GR, Hutkins R, Sanders ME, Prescott SL, Reimer RA, Salminen SJ, Scott K, Stanton C, Swanson KS, Cani PD, et al. Expert consensus document: the International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol. 2017;14(8):491–502. doi:10.1038/nrgastro.2017.75.
- Wade WG. Eubacterium. In: Trujillo ME, Dedysh S, DeVos P, Hedlund B, Kämpfer P, Rainey FA, Whitman WB, editors. Bergey’s manual of systematics of archaea and bacteria. Hoboken (NJ): Wiley; 2015. p. 1–36.
- Chaumeil P-A, Mussig AJ, Hugenholtz P, Parks DH. GTDB-Tk: a toolkit to classify genomes with the Genome Taxonomy Database. Bioinformatics. 2019. doi:10.1093/bioinformatics/btz848.
- Katoh K, Misawa K, Kuma K-I, Miyata T. MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 2002;30:3059–3066. doi:10.1093/nar/gkf436.
- Nguyen L-T, Schmidt HA, von Haeseler A, Minh BQ. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol. 2015;32:268–274. doi:10.1093/molbev/msu300.
- Letunic I, Bork P. Interactive tree of life (iTOL) v3: an online tool for the display and annotation of phylogenetic and other trees. Nucleic Acids Res. 2016;44:W242–W5. doi:10.1093/nar/gkw290.
- Nakazawa F, Sato M, Poco SE, Hashimura T, Ikeda T, Kalfas S, Sundqvist G, Hoshino E. Description of Mogibacterium pumilum gen. nov., sp. nov. and Mogibacterium vescum gen. nov., sp. nov., and reclassification of Eubacterium timidum (Holdeman et al. 1980) as Mogibacterium timidum gen. nov., comb. nov. Int J Syst Evol Microbiol. 2000;50:679–688. doi:10.1099/00207713-50-2-679.
- Taras D, Simmering R, Collins MD, Lawson PA, Blaut M. Reclassification of Eubacterium formicigenerans Holdeman and Moore 1974 as Dorea formicigenerans gen. nov., comb. nov., and description of Dorea longicatena sp. nov., isolated from human faeces. Int J Syst Evol Microbiol. 2002;52:423–428. doi:10.1099/00207713-52-2-423.
- HOFSTAD T, AASJORD P. Eubacterium plautii (Séguin 1928) comb. nov. Int J Syst Evol Microbiol. 1982;32:346–349.
- Willems A, Collins MD. Phylogenetic relationships of the genera acetobacterium and eubacterium sensu stricto and reclassification of eubacterium alactolyticum as pseudoramibacter alactolyticus gen. nov., comb. nov. Int J Syst Evol Microbiol. 1996;46:1083–1087.
- Duncan SH, Hold GL, Barcenilla A, Stewart CS, Flint HJ. Roseburia intestinalis sp. nov., a novel saccharolytic, butyrate-producing bacterium from human faeces. Int J Syst Evol Microbiol. 2002;52:1615–1620. doi:10.1099/00207713-52-5-1615.
- Shetty SA, Zuffa S, Bui TPN, Aalvink S, Smidt H, De Vos WM. Reclassification of Eubacterium hallii as Anaerobutyricum hallii gen. nov., comb. nov., and description of Anaerobutyricum soehngenii sp. nov., a butyrate and propionate-producing bacterium from infant faeces. Int J Syst Evol Microbiol. 2018;68:3741–3746. doi:10.1099/ijsem.0.003041.
- Allen-Vercoe E, Daigneault M, White A, Panaccione R, Duncan SH, Flint HJ, O’Neal L, Lawson PA. Anaerostipes hadrus comb. nov., a dominant species within the human colonic microbiota; reclassification of Eubacterium hadrum Moore et al. 1976. Anaerobe. 2012;18:523–529. doi:10.1016/j.anaerobe.2012.09.002.
- Rosero JA, Jí K, Sechovcová H, Mrázek J, Benada O, Fliegerová K, Havlík J, Kopečný J. Reclassification of Eubacterium rectale (Hauduroy et al. 1937) Prévot 1938 in a new genus Agathobacter gen. nov. as Agathobacter rectalis comb. nov., and description of Agathobacter ruminis sp. nov., isolated from the rumen contents of sheep and cows. Int J Syst Evol Microbiol. 2016;66:768–773. doi:10.1099/ijsem.0.000788.
- Sheridan PO, Duncan SH, Walker AW, Scott KP, Louis P, Flint HJ. Objections to the proposed reclassification of Eubacterium rectale as Agathobacter rectalis. Int J Syst Evol Microbiol. 2016;66:2106. doi:10.1099/ijsem.0.000969.
- Parks DH, Chuvochina M, Waite DW, Rinke C, Skarshewski A, Chaumeil P-A, Hugenholtz P. A standardized bacterial taxonomy based on genome phylogeny substantially revises the tree of life. Nat Biotechnol. 2018;36(10):996–1004. doi:10.1038/nbt.4229.
- Duncan SH, Belenguer A, Holtrop G, Johnstone AM, Flint HJ, Lobley GE. Reduced dietary intake of carbohydrates by obese subjects results in decreased concentrations of butyrate and butyrate-producing bacteria in feces. Appl Environ Microbiol. 2007;73:1073–1078. doi:10.1128/AEM.02340-06.
- Scott KP, Martin JC, Duncan SH, Flint HJ. Prebiotic stimulation of human colonic butyrate-producing bacteria and bifidobacteria, in vitro. FEMS Microbiol Ecol. 2014;87:30–40. doi:10.1111/1574-6941.12186.
- Cockburn DW, Orlovsky NI, Foley MH, Kwiatkowski KJ, Bahr CM, Maynard M, Demeler B, Koropatkin NM. Molecular details of a starch utilization pathway in the human gut symbiont Eubacterium rectale. Mol Microbiol. 2015;95:209–230. doi:10.1111/mmi.12859.
- Sheridan PO, Martin JC, Lawley TD, Browne HP, Harris HMB, Bernalier-Donadille A, Duncan SH, O'Toole PW, Scott KP, Flint HJ. Polysaccharide utilization loci and nutritional specialization in a dominant group of butyrate-producing human colonic Firmicutes. Microb Genom. 2016;2:e000043. doi:10.1099/mgen.0.000043
- Wu GD, Chen J, Hoffmann C, Bittinger K, Chen -Y-Y, Keilbaugh SA, Bewtra M, Knights D, Walters WA, Knight R, et al. Linking long-term dietary patterns with gut microbial enterotypes. Science (New York, NY). 2011;334(6052):105–108. doi:10.1126/science.1208344.
- Araújo JR, Tomas J, Brenner C, Sansonetti PJ. Impact of high-fat diet on the intestinal microbiota and small intestinal physiology before and after the onset of obesity. Biochimie. 2017;141:97–106. doi:10.1016/j.biochi.2017.05.019.
- Moreira APB, Texeira TFS, Ferreira AB, Do Carmo Gouveia Peluzio M, de Cássia Gonçalves Alfenas R. Influence of a high-fat diet on gut microbiota, intestinal permeability and metabolic endotoxaemia. Br J Nutr. 2012;108:801–809. doi:10.1017/S0007114512001213.
- Khemayanto H, Shi B. Role of mediterranean diet in prevention and management of type 2 diabetes. Chin Med J (Engl). 2014;127:3651–3656.
- Ghosh TS, Rampelli S, Jeffery IB, Santoro A, Neto M, Capri M, Giampieri E, Jennings A, Candela M, Turroni S, et al. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: the NU-AGE 1-year dietary intervention across five European countries. Gut. 2020. gutjnl-2019-319654. doi:10.1136/gutjnl-2019-319654.
- Noriega BS, Sanchez-Gonzalez MA, Salyakina D, Coffman J. Understanding the impact of Omega-3 rich diet on the gut microbiota. Case Rep Med. 2016;2016:3089303. doi:10.1155/2016/3089303.
- O’Toole PW, Jeffery IB. Gut microbiota and aging. Science. 2015;350:1214–1215. doi:10.1126/science.aac8469.
- Shoaie S, Ghaffari P, Kovatcheva-Datchary P, Mardinoglu A, Sen P, Pujos-Guillot E, de Wouters T, Juste C, Rizkalla S, Chilloux J, et al. Quantifying diet-induced metabolic changes of the human gut microbiome. Cell Metab. 2015;22(2):320–331. doi:10.1016/j.cmet.2015.07.001.
- Schiffrin EJ, Morley JE, Donnet-Hughes A, Guigoz Y. The inflammatory status of the elderly: the intestinal contribution. Mutat Res. 2010;690:50–56. doi:10.1016/j.mrfmmm.2009.07.011.
- Magrone T, Jirillo E. The interaction between gut microbiota and age-related changes in immune function and inflammation. Immun Ageing. 2013;10:31. doi:10.1186/1742-4933-10-31.
- Biagi E, Candela M, Turroni S, Garagnani P, Franceschi C, Brigidi P. Ageing and gut microbes: perspectives for health maintenance and longevity. Pharmacol Res. 2013;69:11–20. doi:10.1016/j.phrs.2012.10.005.
- Maynard CL, Weaver CT. Intestinal effector T cells in health and disease. Immunity. 2009;31:389–400. doi:10.1016/j.immuni.2009.08.012.
- Biagi E, Nylund L, Candela M, Ostan R, Bucci L, Pini E, Nikkïla J, Monti D, Satokari R, Franceschi C, et al. Through ageing, and beyond: gut microbiota and inflammatory status in seniors and centenarians. PloS One. 2010;5(5):e10667–e. doi:10.1371/journal.pone.0010667.
- Galkin F, Aliper A, Putin E, Kuznetsov I, Gladyshev VN, Zhavoronkov A. Human microbiome aging clocks based on deep learning and tandem of permutation feature importance and accumulated local effects. bioRxiv. 2018;507780. doi:10.1101/507780
- Odamaki T, Kato K, Sugahara H, Hashikura N, Takahashi S, Xiao J-Z, Abe F, Osawa R. Age-related changes in gut microbiota composition from newborn to centenarian: a cross-sectional study. BMC Microbiol. 2016;16(1):90. doi:10.1186/s12866-016-0708-5.
- El Kaoutari A, Armougom F, Gordon JI, Raoult D, Henrissat B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat Rev Microbiol. 2013;11:497–504. doi:10.1038/nrmicro3050.
- Chassard C, Lacroix C. Carbohydrates and the human gut microbiota. Curr Opin Clin Nutr Metab Care. 2013;16:453–460. doi:10.1097/MCO.0b013e3283619e63.
- Topping DL, Clifton PM. Short-chain fatty acids and human colonic function: roles of resistant starch and nonstarch polysaccharides. Physiol Rev. 2001;81:1031–1064. doi:10.1152/physrev.2001.81.3.1031.
- Jakobsdottir G, Xu J, Molin G, Ahrné S, Nyman M. High-fat diet reduces the formation of butyrate, but increases succinate, inflammation, liver fat and cholesterol in rats, while dietary fibre counteracts these effects. PLoS One. 2013;8:e80476. doi:10.1371/journal.pone.0080476.
- Louis P, Flint HJ. Formation of propionate and butyrate by the human colonic microbiota. Environ Microbiol. 2017;19:29–41. doi:10.1111/1462-2920.13589.
- Moore WE, Holdeman LV. Human fecal flora: the normal flora of 20 Japanese-Hawaiians. Appl Microbiol. 1974;27:961–979. doi:10.1128/AEM.27.5.961-979.1974.
- Barcenilla A, Pryde SE, Martin JC, Duncan SH, Stewart CS, Henderson C, Flint HJ. Phylogenetic relationships of butyrate-producing bacteria from the human gut. Appl Environ Microbiol. 2000;66:1654–1661. doi:10.1128/AEM.66.4.1654-1661.2000.
- Louis P, Flint HJ. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol Lett. 2009;294:1–8. doi:10.1111/j.1574-6968.2009.01514.x.
- Walker AW, Ince J, Duncan SH, Webster LM, Holtrop G, Ze X, Brown D, Stares MD, Scott P, Bergerat A, et al. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. Isme J. 2011;5(2):220–230. doi:10.1038/ismej.2010.118.
- Louis P, Young P, Holtrop G, Flint HJ. Diversity of human colonic butyrate-producing bacteria revealed by analysis of the butyryl-CoA: acetateCoA-transferase gene. Environ Microbiol. 2010;12:304–314. doi:10.1111/j.1462-2920.2009.02066.x.
- Pham VT, Lacroix C, Braegger CP, Chassard C. Early colonization of functional groups of microbes in the infant gut. Environ Microbiol. 2016;18:2246–2258. doi:10.1111/1462-2920.13316.
- Engels C, Ruscheweyh H-J, Beerenwinkel N, Lacroix C, Schwab C. The common gut microbe eubacterium hallii also contributes to intestinal propionate formation. Front Microbiol. 2016;7:713. doi:10.3389/fmicb.2016.00713.
- Gänzle MG. Lactic metabolism revisited: metabolism of lactic acid bacteria in food fermentations and food spoilage. Curr Opin Food Sci. 2015;2:106–117.
- Chowdhury C, Sinha S, Chun S, Yeates TO, Bobik TA. Diverse bacterial microcompartment organelles. Microbiol Mol Biol Rev. 2014;78:438–468. doi:10.1128/MMBR.00009-14.
- Rivière A, Gagnon M, Weckx S, Roy D, De Vuyst L. mutual cross-feeding interactions between bifidobacterium longum subsp. longum NCC2705 and Eubacterium rectale ATCC 33656 explain the bifidogenic and butyrogenic effects of arabinoxylan oligosaccharides. Appl Environ Microbiol. 2015;81:7767–7781. doi:10.1128/AEM.02089-15.
- Kanauchi O, Fujiyama Y, Mitsuyama K, Araki Y, Ishii T, Nakamura T, Hitomi Y, Agata K, Saiki T, Andoh A, et al. Increased growth of Bifidobacterium and Eubacterium by germinated barley foodstuff, accompanied by enhanced butyrate production in healthy volunteers. Int J Mol Med. 1999;3:175–179. doi:10.3892/ijmm.3.2.175.
- Schwab C, Ruscheweyh H-J, Bunesova V, Pham VT, Beerenwinkel N, Lacroix C. Trophic interactions of infant bifidobacteria and eubacterium hallii during L-fucose and fucosyllactose degradation. Front Microbiol. 2017;8:95. doi:10.3389/fmicb.2017.00095.
- Guilloteau P, Martin L, Eeckhaut V, Ducatelle R, Zabielski R, Van Immerseel F. From the gut to the peripheral tissues: the multiple effects of butyrate. Nutr Res Rev. 2010;23:366–384. doi:10.1017/S0954422410000247.
- Chen J, Li Y, Tian Y, Huang C, Li D, Zhong Q, Ma X. Interaction between microbes and host intestinal health: modulation by dietary nutrients and gut-brain-endocrine-immune axis. Curr Protein Pept Sci. 2015;16:592–603. doi:10.2174/1389203716666150630135720.
- Van den Abbeele P, Belzer C, Goossens M, Kleerebezem M, De Vos WM, Thas O, De Weirdt R, Kerckhof F-M, Van de Wiele T. Butyrate-producing Clostridium cluster XIVa species specifically colonize mucins in an in vitro gut model. Isme J. 2013;7(5):949–961. doi:10.1038/ismej.2012.158.
- Willemsen LE, Koetsier MA, van Deventer SJ, van Tol EA. Short chain fatty acids stimulate epithelial mucin 2 expression through differential effects on prostaglandin E(1) and E(2) production by intestinal myofibroblasts. Gut. 2003;52:1442–1447. doi:10.1136/gut.52.10.1442.
- Wang HB, Wang PY, Wang X, Wan YL, Liu YC. Butyrate enhances intestinal epithelial barrier function via up-regulation of tight junction protein Claudin-1 transcription. Dig Dis Sci. 2012;57:3126–3135. doi:10.1007/s10620-012-2259-4.
- Corrêa-Oliveira R, Fachi JL, Vieira A, Sato FT, Vinolo MAR. Regulation of immune cell function by short-chain fatty acids. Clin Trans Immunol. 2016;5:e73–e. doi:10.1038/cti.2016.17.
- Liu H, Wang J, He T, Becker S, Zhang G, Li D, Ma X. Butyrate: A double-edged sword for health? Adv Nutr (Bethesda, Md). 2018;9(1):21–29. doi:10.1093/advances/nmx009.
- Mowat AM, Agace WW. Regional specialization within the intestinal immune system. Nat Rev Immunol. 2014;14:667–685. doi:10.1038/nri3738.
- Singh N, Gurav A, Sivaprakasam S, Brady E, Padia R, Shi H, Thangaraju M, Prasad P, Manicassamy S, Munn D, et al. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity. 2014;40(1):128–139. doi:10.1016/j.immuni.2013.12.007.
- Macia L, Tan J, Vieira AT, Leach K, Stanley D, Luong S, Maruya M, Ian McKenzie C, Hijikata A, Wong C, et al. Metabolite-sensing receptors GPR43 and GPR109A facilitate dietary fibre-induced gut homeostasis through regulation of the inflammasome. Nat Commun. 2015;6(1):6734. doi:10.1038/ncomms7734.
- Iraporda C, Errea A, Romanin DE, Cayet D, Pereyra E, Pignataro O, Sirard JC, Garrote GL, Abraham AG, Rumbo M, et al. Lactate and short chain fatty acids produced by microbial fermentation downregulate proinflammatory responses in intestinal epithelial cells and myeloid cells. Immunobiology. 2015;220(10):1161–1169. doi:10.1016/j.imbio.2015.06.004.
- Sunkara LT, Jiang W, Zhang G. Modulation of antimicrobial host defense peptide gene expression by free fatty acids. Plos One. 2012;7:e49558. doi:10.1371/journal.pone.0049558.
- Puddu A, Sanguineti R, Montecucco F, Viviani GL. Evidence for the gut microbiota short-chain fatty acids as key pathophysiological molecules improving diabetes. Mediators Inflamm. 2014;2014:162021. doi:10.1155/2014/162021.
- Nagao-Kitamoto H, Kamada N. Host-microbial cross-talk in inflammatory bowel disease. Immune Netw. 2017;17:1–12. doi:10.4110/in.2017.17.1.1.
- Kang S, Denman SE, Morrison M, Yu Z, Dore J, Leclerc M, McSweeney CS. Dysbiosis of fecal microbiota in Crohn’s disease patients as revealed by a custom phylogenetic microarray. Inflamm Bowel Dis. 2010;16:2034–2042. doi:10.1002/ibd.21319.
- El Mouzan MI, Winter HS, Assiri AA, Korolev KS, Al Sarkhy AA, Dowd SE, Al Mofarreh MA, Menon R. Microbiota profile in new-onset pediatric Crohn’s disease: data from a non-Western population. Gut Pathog. 2018;10(1):49. doi:10.1186/s13099-018-0276-3.
- Knoll RL, Forslund K, Kultima JR, Meyer CU, Kullmer U, Sunagawa S, Bork P, Gehring S. Gut microbiota differs between children with inflammatory bowel disease and healthy siblings in taxonomic and functional composition: a metagenomic analysis. Am J Physiol Gastrointest Liver Physiol. 2017;312(4):G327–G39. doi:10.1152/ajpgi.00293.2016.
- Vermeiren J, Van den Abbeele P, Laukens D, Vigsnaes LK, De Vos M, Boon N, Wiele T. Decreased colonization of fecal Clostridium coccoides/Eubacterium rectale species from ulcerative colitis patients in an in vitro dynamic gut model with mucin environment. FEMS Microbiol Ecol. 2012;79(3):685–696. doi:10.1111/j.1574-6941.2011.01252.x.
- Maukonen J, Kolho K-L, Paasela M, Honkanen J, Klemetti P, Vaarala O, Saarela M. Altered fecal microbiota in paediatric inflammatory bowel disease. J Crohns Colitis. 2015;9(12):1088–1095. doi:10.1093/ecco-jcc/jjv147.
- Hirano A, Umeno J, Okamoto Y, Shibata H, Ogura Y, Moriyama T, Torisu T, Fujioka S, Fuyuno Y, Kawarabayasi Y, et al. Comparison of the microbial community structure between inflamed and non-inflamed sites in patients with ulcerative colitis. J Gastroenterol Hepatol. 2018;33(9):1590–1597. doi:10.1111/jgh.14129.
- Kolho K-L, Korpela K, Jaakkola T, Pichai MVA, Zoetendal EG, Salonen A, de Vos WM. Fecal microbiota in pediatric inflammatory bowel disease and its relation to inflammation. Am J Gastroenterol. 2015;110(6):921–930. doi:10.1038/ajg.2015.149.
- Ott S, Musfeldt M, Wenderoth D, Hampe J, Brant O, Fölsch U, Timmis KN, Schreiber S. Reduction in diversity of the colonic mucosa associated bacterial microflora in patients with active inflammatory bowel disease. Gut. 2004;53:685–693. doi:10.1136/gut.2003.025403.
- de Meij TGJ, de Groot EFJ, Peeters CFW, de Boer NKH, Kneepkens CMF, Eck A, Benninga MA, Savelkoul PHM, van Bodegraven AA, Budding AE, et al. Variability of core microbiota in newly diagnosed treatment-naïve paediatric inflammatory bowel disease patients. PloS One. 2018;13(8):e0197649–e. doi:10.1371/journal.pone.0197649.
- Rojas-Feria M, Romero-García T, Fernández Caballero-Rico JÁ, Pastor Ramírez H, Avilés-Recio M, Castro-Fernandez M, Chueca Porcuna N, Romero-Gόmez M, García F, Grande L, et al. Modulation of faecal metagenome in Crohn’s disease: role of microRNAs as biomarkers. World J Gastroenterol. 2018;24(46):5223–5233. doi:10.3748/wjg.v24.i46.5223.
- Gevers D, Kugathasan S, Denson LA, Vázquez-Baeza Y, Van Treuren W, Ren B, Schwager E, Knights D, Song S, Yassour M, et al. The treatment-naive microbiome in new-onset Crohn’s disease. Cell Host Microbe. 2014;15(3):382–392. doi:10.1016/j.chom.2014.02.005.
- Vester-Andersen MK, Mirsepasi-Lauridsen HC, Prosberg MV, Mortensen CO, Träger C, Skovsen K, Thorkilgaard T, Nøjgaard C, Vind I, Krogfelt KA, et al. Increased abundance of proteobacteria in aggressive Crohn’s disease seven years after diagnosis. Sci Rep. 2019;9(1):13473. doi:10.1038/s41598-019-49833-3.
- Pei L-Y, Ke Y-S, Zhao -H-H, Wang L, Jia C, Liu W-Z, Fu Q-H, Shi M-N, Cui J, Li S-C, et al. Role of colonic microbiota in the pathogenesis of ulcerative colitis. BMC Gastroenterol. 2019;19(1):10. doi:10.1186/s12876-019-0930-3.
- Mottawea W, Chiang C-K, Mühlbauer M, Starr AE, Butcher J, Abujamel T, Deeke SA, Brandel A, Zhou H, Shokralla S, et al. Altered intestinal microbiota-host mitochondria crosstalk in new onset Crohn’s disease. Nat Commun. 2016;7:13419. doi:10.1038/ncomms13419.
- Marchesi JR, Holmes E, Khan F, Kochhar S, Scanlan P, Shanahan F, Wilson ID, Wang Y. Rapid and noninvasive metabonomic characterization of inflammatory bowel disease. J Proteome Res. 2007;6(2):546–551. doi:10.1021/pr060470d.
- Vital M, Penton CR, Wang Q, Young VB, Antonopoulos DA, Sogin ML, Morrison HG, Raffals L, Chang EB, Huffnagle GB, et al. A gene-targeted approach to investigate the intestinal butyrate-producing bacterial community. Microbiome. 2013;1:8. doi:10.1186/2049-2618-1-8.
- Frank DN, Amand ALS, Feldman RA, Boedeker EC, Harpaz N, Pace NR. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc Natl Acad Sci USA. 2007;104:13780–13785. doi:10.1073/pnas.0706625104.
- Geirnaert A, Calatayud M, Grootaert C, Laukens D, Devriese S, Smagghe G, De Vos M, Boon N, Van de Wiele T. Butyrate-producing bacteria supplemented in vitro to Crohn’s disease patient microbiota increased butyrate production and enhanced intestinal epithelial barrier integrity. Sci Rep. 2017;7(1):11450. doi:10.1038/s41598-017-11734-8.
- Kanauchi O, Fukuda M, Matsumoto Y, Ishii S, Ozawa T, Shimizu M, Mitsuyama K, Andoh A. Eubacterium limosum ameliorates experimental colitis and metabolite of microbe attenuates colonic inflammatory action with increase of mucosal integrity. World J Gastroenterol. 2006;12:1071–1077. doi:10.3748/wjg.v12.i7.1071.
- Verdam FJ, Fuentes S, de Jonge C, Zoetendal EG, Erbil R, Greve JW, Buurman WA, de Vos WM, Rensen SS. Human intestinal microbiota composition is associated with local and systemic inflammation in obesity. Obesity (Silver Spring). 2013;21(12):E607–E15. doi:10.1002/oby.20466.
- Kasai C, Sugimoto K, Moritani I, Tanaka J, Oya Y, Inoue H, TAMEDA M, SHIRAKI K, ITO M, TAKEI Y, et al. Comparison of human gut microbiota in control subjects and patients with colorectal carcinoma in adenoma: terminal restriction fragment length polymorphism and next-generation sequencing analyses. Oncol Rep. 2016;35(1):325–333. doi:10.3892/or.2015.4398.
- Munukka E, Wiklund P, Pekkala S, Völgyi E, Xu L, Cheng S, Lyytikäinen A, Marjomäki V, Alen M, Vaahtovuo J, et al. Women with and without metabolic disorder differ in their gut microbiota composition. Obesity (Silver Spring). 2012;20(5):1082–1087. doi:10.1038/oby.2012.8.
- Simões CD, Maukonen J, Kaprio J, Rissanen A, Pietiläinen KH, Saarela M. Habitual dietary intake is associated with stool microbiota composition in monozygotic twins. J Nutr. 2013;143:417–423. doi:10.3945/jn.112.166322.
- Sepp E, Lõivukene K, Julge K, Voor T, Mikelsaar M. The association of gut microbiota with body weight and body mass index in preschool children of Estonia. Microb Ecol Health Dis. 2013;24:19231. doi:10.3402/mehd.v24i0.19231.
- Schwiertz A, Taras D, Schäfer K, Beijer S, Bos NA, Donus C, Hardt PD. Microbiota and SCFA in lean and overweight healthy subjects. Obesity (Silver Spring). 2010;18(1):190–195. doi:10.1038/oby.2009.167.
- Bach Knudsen KE. Microbial degradation of whole-grain complex carbohydrates and impact on short-chain fatty acids and health. Adv Nutr (Bethesda, Md). 2015;6:206–213. doi:10.3945/an.114.007450.
- Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444:1027–1031. doi:10.1038/nature05414.
- Kovatcheva-Datchary P, Shoaie S, Lee S, Wahlström A, Nookaew I, Hallen A, Perkins R, Nielsen J, Bäckhed F. Simplified intestinal microbiota to study microbe-diet-host interactions in a mouse model. Cell Rep. 2019;26(13):3772–83.e6. doi:10.1016/j.celrep.2019.02.090.
- Duncan SH, Lobley GE, Holtrop G, Ince J, Johnstone AM, Louis P, Flint HJ. Human colonic microbiota associated with diet, obesity and weight loss. Int J Obes (Lond). 2008;32(11):1720–1724. doi:10.1038/ijo.2008.155.
- Balamurugan R, George G, Kabeerdoss J, Hepsiba J, Chandragunasekaran AM, Ramakrishna BS. Quantitative differences in intestinal Faecalibacterium prausnitzii in obese Indian children. Br J Nutr. 2010;103:335–338. doi:10.1017/S0007114509992182.
- De Vadder F, Kovatcheva-Datchary P, Goncalves D, Vinera J, Zitoun C, Duchampt A, Bäckhed F, Mithieux G. Microbiota-generated metabolites promote metabolic benefits via gut-brain neural circuits. Cell. 2014;156(1–2):84–96. doi:10.1016/j.cell.2013.12.016.
- Hong J, Jia Y, Pan S, Jia L, Li H, Han Z, Cai D, Zhao R. Butyrate alleviates high fat diet-induced obesity through activation of adiponectin-mediated pathway and stimulation of mitochondrial function in the skeletal muscle of mice. Oncotarget. 2016;7(35):56071–56082. doi:10.18632/oncotarget.11267.
- Forslund K, Hildebrand F, Nielsen T, Falony G, Le Chatelier E, Sunagawa S, Prifti E, Vieira-Silva S, Gudmundsdottir V, Krogh Pedersen H, et al. Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature. 2015;528(7581):262–266. doi:10.1038/nature15766.
- Dao MC, Everard A, Aron-Wisnewsky J, Sokolovska N, Prifti E, Verger EO, Kayser BD, Levenez F, Chilloux J, Hoyles L, et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut. 2016;65(3):426. doi:10.1136/gutjnl-2014-308778.
- Qin J, Li Y, Cai Z, Li S, Zhu J, Zhang F, Liang S, Zhang W, Guan Y, Shen D, et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490(7418):55–60. doi:10.1038/nature11450.
- Karlsson FH, Tremaroli V, Nookaew I, Bergström G, Behre CJ, Fagerberg B, Nielsen J, Bäckhed F. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature. 2013;498(7452):99–103. doi:10.1038/nature12198.
- Vrieze A, Van Nood E, Holleman F, Salojärvi J, Kootte RS, Bartelsman JFWM, Dallinga–Thie GM, Ackermans MT, Serlie MJ, Oozeer R, et al. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology. 2012;143(4):913–6.e7. doi:10.1053/j.gastro.2012.06.031.
- Udayappan S, Manneras-Holm L, Chaplin-Scott A, Belzer C, Herrema H, Dallinga-Thie GM, Duncan SH, Stroes ESG, Groen AK, Flint HJ, et al. Oral treatment with Eubacterium hallii improves insulin sensitivity in db/db mice. Npj Biofilms Microbiomes. 2016;2(1):16009. doi:10.1038/npjbiofilms.2016.9.
- Kootte RS, Levin E, Salojärvi J, Smits LP, Hartstra AV, Udayappan SD, Hermes G, Bouter KE, Koopen AM, Holst JJ, et al. Improvement of insulin sensitivity after lean donor feces in metabolic syndrome is driven by baseline intestinal microbiota composition. Cell Metab. 2017;26(4):611–9.e6. doi:10.1016/j.cmet.2017.09.008.
- Lin HV, Frassetto A, Kowalik Jr EJ Jr., Nawrocki AR, Lu MM, Kosinski JR, Hubert JA, Szeto D, Yao X, Forrest G, et al. Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS One. 2012;7(4):e35240. doi:10.1371/journal.pone.0035240.
- Tolhurst G, Heffron H, Lam YS, Parker HE, Habib AM, Diakogiannaki E, Cameron J, Grosse J, Reimann F, Gribble FM, et al. Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein-coupled receptor FFAR2. Diabetes. 2012;61(2):364–371. doi:10.2337/db11-1019.
- Engelstoft MS, Schwartz TW. Opposite regulation of ghrelin and glucagon-like peptide-1 by metabolite G-protein-coupled receptors. Trends Endocrinol Metab. 2016;27:665–675. doi:10.1016/j.tem.2016.07.001.
- Queipo-Ortuño MI, Seoane LM, Murri M, Pardo M, Gomez-Zumaquero JM, Cardona F, Casanueva F, Tinahones FJ. Gut microbiota composition in male rat models under different nutritional status and physical activity and its association with serum leptin and ghrelin levels. PLoS One. 2013;8(5):e65465. doi:10.1371/journal.pone.0065465.
- Zeevi D, Korem T, Zmora N, Israeli D, Rothschild D, Weinberger A, Ben-Yacov O, Lador D, Avnit-Sagi T, Lotan-Pompan M, et al. Personalized nutrition by prediction of glycemic responses. Cell. 2015;163(5):1079–1094. doi:10.1016/j.cell.2015.11.001.
- Khan S, Jena G. The role of butyrate, a histone deacetylase inhibitor in diabetes mellitus: experimental evidence for therapeutic intervention. Epigenomics. 2015;7:669–680. doi:10.2217/epi.15.20.
- de Luca C, Olefsky JM. Inflammation and insulin resistance. FEBS Lett. 2008;582:97–105. doi:10.1016/j.febslet.2007.11.057.
- Cucak H, Mayer C, Tonnesen M, Thomsen LH, Grunnet LG, Rosendahl A. Macrophage contact dependent and independent TLR4 mechanisms induce β-cell dysfunction and apoptosis in a mouse model of type 2 diabetes. PLoS One. 2014;9:e90685. doi:10.1371/journal.pone.0090685.
- Biragyn A, Ferrucci L. Gut dysbiosis: a potential link between increased cancer risk in ageing and inflammaging. Lancet Oncol. 2018;19:e295–e304. doi:10.1016/S1470-2045(18)30095-0.
- Jacouton E, Chain F, Sokol H, Langella P, Bermúdez-Humarán LG. Probiotic strain lactobacillus casei BL23 prevents colitis-associated colorectal cancer. Front Immunol. 2017;8:1553. doi:10.3389/fimmu.2017.01553.
- Encarnação JC, Abrantes AM, Pires AS, Botelho MF. Revisit dietary fiber on colorectal cancer: butyrate and its role on prevention and treatment. Cancer Metastasis Rev. 2015;34:465–478. doi:10.1007/s10555-015-9578-9.
- Waldecker M, Kautenburger T, Daumann H, Busch C, Schrenk D. Inhibition of histone-deacetylase activity by short-chain fatty acids and some polyphenol metabolites formed in the colon. J Nutr Biochem. 2008;19:587–593. doi:10.1016/j.jnutbio.2007.08.002.
- Chen J, Vitetta L. Inflammation-modulating effect of butyrate in the prevention of colon cancer by dietary fiber. Clin Colorectal Cancer. 2018;17:e541–e4. doi:10.1016/j.clcc.2018.05.001.
- Chen H-M, Yu Y-N, Wang J-L, Lin Y-W, Kong X, Yang C-Q, Yang L, Liu Z-J, Yuan Y-Z, Liu F, et al. Decreased dietary fiber intake and structural alteration of gut microbiota in patients with advanced colorectal adenoma. Am J Clin Nutr. 2013;97(5):1044–1052. doi:10.3945/ajcn.112.046607.
- Donohoe DR, Holley D, Collins LB, Montgomery SA, Whitmore AC, Hillhouse A, Curry KP, Renner SW, Greenwalt A, Ryan EP, et al. A gnotobiotic mouse model demonstrates that dietary fiber protects against colorectal tumorigenesis in a microbiota- and butyrate-dependent manner. Cancer Discov. 2014;4(12):1387–1397. doi:10.1158/2159-8290.CD-14-0501.
- Candido EP. Sodium butyrate inhibits histone deacetylation in cultured cells. Cell. 1978;14(1):105–113. doi:10.1016/0092-8674(78)90305-7.
- Chen J, Jiang CC, Jin L, Zhang XD. Regulation of PD-L1: a novel role of pro-survival signalling in cancer. Ann Oncol. 2016;27(3):409–416. doi:10.1093/annonc/mdv615.
- Donohoe DR, Curry KP, Bultman SJ. Microbial oncotarget: bacterial-produced butyrate, chemoprevention and warburg effect. Oncotarget. 2013 February;4(2):182–183. doi:10.18632/oncotarget.915.
- Andriamihaja M, Chaumontet C, Tome D, Blachier F. Butyrate metabolism in human colon carcinoma cells: implications concerning its growth-inhibitory effect. J Cell Physiol. 2009;218:58–65. doi:10.1002/jcp.21556.
- Donohoe DR, Collins LB, Wali A, Bigler R, Sun W, Bultman SJ. The Warburg effect dictates the mechanism of butyrate-mediated histone acetylation and cell proliferation. Mol Cell. 2012;48:612–626. doi:10.1016/j.molcel.2012.08.033.
- Wang T, Cai G, Qiu Y, Fei N, Zhang M, Pang X, Jia W, Cai S, Zhao L. Structural segregation of gut microbiota between colorectal cancer patients and healthy volunteers. Isme J. 2012;6(2):320–329. doi:10.1038/ismej.2011.109.
- Ai D, Pan H, Li X, Gao Y, Liu G, Xia LC. Identifying gut microbiota associated with colorectal cancer using a zero-inflated lognormal model. Front Microbiol. 2019;10:826. doi:10.3389/fmicb.2019.00826.
- Yu J, Feng Q, Wong SH, Zhang D, Liang QY, Qin Y, Tang L, Zhao H, Stenvang J, Li Y, et al. Metagenomic analysis of faecal microbiome as a tool towards targeted non-invasive biomarkers for colorectal cancer. Gut. 2017;66(1):70–78. doi:10.1136/gutjnl-2015-309800.
- Jie Z, Xia H, Zhong S-L, Feng Q, Li S, Liang S, Zhong H, Liu Z, Gao Y, Zhao H, et al. The gut microbiome in atherosclerotic cardiovascular disease. Nat Commun. 2017;8(1):845. doi:10.1038/s41467-017-00900-1.
- Karlsson FH, Fåk F, Nookaew I, Tremaroli V, Fagerberg B, Petranovic D, Bäckhed F, Nielsen J. Symptomatic atherosclerosis is associated with an altered gut metagenome. Nat Commun. 2012;3(1):1245. doi:10.1038/ncomms2266.
- Bingham SA, Day NE, Luben R, Ferrari P, Slimani N, Norat T, Clavel-Chapelon F, Kesse E, Nieters A, Boeing H, et al. Dietary fibre in food and protection against colorectal cancer in the European prospective investigation into cancer and nutrition (EPIC): an observational study. Lancet (London, England). 2003;361(9368):1496–1501. doi:10.1016/S0140-6736(03)13174-1.
- Clausen MR, Bonnén H, Mortensen PB. Colonic fermentation of dietary fibre to short chain fatty acids in patients with adenomatous polyps and colonic cancer. Gut. 1991;32(8):923–928. doi:10.1136/gut.32.8.923.
- Chiang JYL. Bile acids: regulation of synthesis. J Lipid Res. 2009;50:1955–1966. doi:10.1194/jlr.R900010-JLR200.
- Chiang JYL. Bile acid metabolism and signaling. Compr Physiol. 2013;3:1191–1212. doi:10.1002/cphy.c120023.
- Gadaleta RM, van Mil SW, Oldenburg B, Siersema PD, Klomp LW, van Erpecum KJ. Bile acids and their nuclear receptor FXR: relevance for hepatobiliary and gastrointestinal disease. Biochim Biophys Acta. 2010;1801:683–692. doi:10.1016/j.bbalip.2010.04.006.
- Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell. 2010;140:883–899. doi:10.1016/j.cell.2010.01.025.
- Feng Q, Zhang D, Liu C, Xiao L, Tang L, Wang J, inventors; BGI Shenzhen, assignee. Use of eubacterium in the prevention and treatment for colorectal cancer related diseases. China patent CN 106687130B. 2020 January 21.
- Chan YK, Brar MS, Kirjavainen PV, Chen Y, Peng J, Li D, Leung FC, El-Nezami H. High fat diet induced atherosclerosis is accompanied with low colonic bacterial diversity and altered abundances that correlates with plaque size, plasma A-FABP and cholesterol: a pilot study of high fat diet and its intervention with Lactobacillus rhamnosus GG (LGG) or telmisartan in ApoE(-/-) mice. BMC Microbiol. 2016;16:264.
- Ma J, Li H. The role of gut microbiota in atherosclerosis and hypertension. Front Pharmacol. 2018;9:1082.
- Freier TA, Beitz DC, Li L, Hartman PA. Characterization of Eubacterium coprostanoligenes sp. nov., a cholesterol-reducing anaerobe. Int J Syst Bacteriol. 1994;44(1):137–142. doi:10.1099/00207713-44-1-137.
- Kenny DJ, Plichta DR, Shungin D, Koppel N, Hall AB, Fu B, Vasan RS, Shaw SY, Vlamakis H, Balskus EP, et al. Cholesterol metabolism by uncultured human gut bacteria influences host cholesterol level. Cell Host Microbe. 2020. doi:10.1016/j.chom.2020.05.013.
- Kriaa A, Bourgin M, Mkaouar H, Jablaoui A, Akermi N, Soussou S, Maguin E, Rhimi M. Microbial reduction of cholesterol to coprostanol: an old concept and new insights. Catalysts. 2019;9(2):167. doi:10.3390/catal9020167.
- Ren D, Li L, Schwabacher AW, Young JW, Beitz DC. Mechanism of cholesterol reduction to coprostanol by Eubacterium coprostanoligenes ATCC 51222. Steroids. 1996;61:33–40. doi:10.1016/0039-128X(95)00173-N.
- Cuevas-Tena M, Alegría A, Lagarda MJ. Relationship between dietary sterols and gut microbiota: A review. Eur J Lipid Sci Technol. 2018;120:1800054. doi:10.1002/ejlt.201800054.
- Gérard P. Metabolism of cholesterol and bile acids by the gut microbiota. Pathogens. 2013;3:14–24. doi:10.3390/pathogens3010014.
- Sekimoto H, Shimada O, Makanishi M, Nakano T, Katayama O. Interrelationship between serum and fecal sterols. Jpn J Med. 1983;22:14–20. doi:10.2169/internalmedicine1962.22.14.
- Li L, Buhman KK, Hartman PA, Beitz DC. Hypocholesterolemic effect of Eubacterium coprostanoligenes ATCC 51222 in rabbits. Lett Appl Microbiol. 1995;20:137–140. doi:10.1111/j.1472-765X.1995.tb00410.x.
- Li L, Batt SM, Wannemuehler M, Dispirito A, Beitz DC. Effect of feeding of a cholesterol-reducing bacterium, Eubacterium coprostanoligenes, to germ-free mice. Comp Med. 1998;48:253–255.
- Antharam VC, McEwen DC, Garrett TJ, Dossey AT, Li EC, Kozlov AN, Mesbah Z, Wang GP. An integrated metabolomic and microbiome analysis identified specific gut microbiota associated with fecal cholesterol and coprostanol in clostridium difficile infection. PLoS One. 2016;11(2):e0148824. doi:10.1371/journal.pone.0148824.
- Krobot KJ, Yin DD, Alemao E, Steinhagen-Thiessen E. Real-world effectiveness of lipid-lowering therapy in male and female outpatients with coronary heart disease: relation to pre-treatment low-density lipoprotein-cholesterol, pre-treatment coronary heart disease risk, and other factors. Eur J Cardiovasc Prev Rehabil. 2005;12:37–45. doi:10.1177/204748730501200106.
- Matthan NR, Pencina M, LaRocque JM, Jacques PF, D’Agostino RB, Schaefer EJ, Lichtenstein AH. Alterations in cholesterol absorption/synthesis markers characterize Framingham offspring study participants with CHD. J Lipid Res. 2009;50:1927–1935. doi:10.1194/jlr.P900039-JLR200.
- Veiga P, Juste C, Lepercq P, Saunier K, Béguet F, Gérard P. Correlation between faecal microbial community structure and cholesterol-to-coprostanol conversion in the human gut. FEMS Microbiol Lett. 2005;242:81–86. doi:10.1016/j.femsle.2004.10.042.
- Beitz DC, Young JW, Li L, Buhman KK, inventors; Iowa State University Research Foundation, assignee. Oral administration of coprostanol producing microorganisms to humans to decrease plasma cholesterol concentration. United States patent US 5972685A. 1999 October 26.
- Ye JZ, Li YT, Wu WR, Shi D, Fang DQ, Yang LY, Bian X-Y, Wu -J-J, Wang Q, Jiang X-W, et al. Dynamic alterations in the gut microbiota and metabolome during the development of methionine-choline-deficient diet-induced nonalcoholic steatohepatitis. World J Gastroenterol. 2018;24:2468–2481. doi:10.3748/wjg.v24.i23.2468.
- Song Z, Cai Y, Lao X, Wang X, Lin X, Cui Y, Kalavagunta PK, Liao J, Jin L, Shang J, et al. Taxonomic profiling and populational patterns of bacterial bile salt hydrolase (BSH) genes based on worldwide human gut microbiome. Microbiome. 2019;7(1):9. doi:10.1186/s40168-019-0628-3.
- Ridlon JM, Kang DJ, Hylemon PB. Bile salt biotransformations by human intestinal bacteria. J Lipid Res. 2006;47:241–259. doi:10.1194/jlr.R500013-JLR200.
- De Smet I, Van Hoorde L, Vande Woestyne M, Christiaens H, Verstraete W. Significance of bile salt hydrolytic activities of lactobacilli. J Appl Bacteriol. 1995;79:292–301. doi:10.1111/j.1365-2672.1995.tb03140.x.
- Lefebvre P, Cariou B, Lien F, Kuipers F, Staels B. Role of bile acids and bile acid receptors in metabolic regulation. Physiol Rev. 2009;89:147–191. doi:10.1152/physrev.00010.2008.
- Urdaneta V, Casadesús J. Interactions between bacteria and bile salts in the gastrointestinal and hepatobiliary tracts. Frontiers Med. 2017;4(163). doi:10.3389/fmed.2017.00163.
- Ridlon JM, Kang DJ, Hylemon PB, Bajaj JS. Bile acids and the gut microbiome. Curr Opin Gastroenterol. 2014;30:332–338. doi:10.1097/MOG.0000000000000057.
- Björkhem I, Einarsson K, Melone P, Hylemon P. Mechanism of intestinal formation of deoxycholic acid from cholic acid in humans: evidence for a 3-oxo-delta 4-steroid intermediate. J Lipid Res. 1989;30:1033–1039.
- Jia W, Xie G, Jia W. Bile acid-microbiota crosstalk in gastrointestinal inflammation and carcinogenesis. Nat Rev Gastroenterol Hepatol. 2018;15:111–128. doi:10.1038/nrgastro.2017.119.
- Jia E-T, Liu Z-Y, Pan M, Lu J-F, Ge Q-Y. Regulation of bile acid metabolism-related signaling pathways by gut microbiota in diseases. J Zhejiang Univ Sci B. 2019;20:781–792. doi:10.1631/jzus.B1900073.
- Guo C, Chen W-D, Wang Y-D. TGR5, not only a metabolic regulator. Front Physiol. 2016;7:646. doi:10.3389/fphys.2016.00646.
- Allegretti JR, Kearney S, Li N, Bogart E, Bullock K, Gerber GK, Bry L, Clish CB, Alm E, Korzenik JR, et al. Recurrent Clostridium difficile infection associates with distinct bile acid and microbiome profiles. Aliment Pharmacol Ther. 2016;43(11):1142–1153. doi:10.1111/apt.13616.
- Theriot CM, Koenigsknecht MJ, Carlson PE Jr., Hatton GE, Nelson AM, Li B, Huffnagle GB, Z. Li J, Young VB. Antibiotic-induced shifts in the mouse gut microbiome and metabolome increase susceptibility to Clostridium difficile infection. Nat Commun. 2014;5(1):3114. doi:10.1038/ncomms4114.
- Theriot CM, Young VB. Microbial and metabolic interactions between the gastrointestinal tract and Clostridium difficile infection. Gut Microbes. 2014;5(1):86–95. doi:10.4161/gmic.27131.
- Mullish BH, McDonald JA, Pechlivanis A, Allegretti JR, Kao D, Barker GF, Kapila D, Petrof EO, Joyce SA, Gahan CGM, et al. Microbial bile salt hydrolases mediate the efficacy of faecal microbiota transplant in the treatment of recurrent Clostridioides difficile infection. Gut. 2019;68(10):1791–1800. doi:10.1136/gutjnl-2018-317842.
- Yoshimoto S, Loo TM, Atarashi K, Kanda H, Sato S, Oyadomari S, Iwakura Y, Oshima K, Morita H, Hattori M, et al. Obesity-induced gut microbial metabolite promotes liver cancer through senescence secretome. Nature. 2013;499(7456):97–101. doi:10.1038/nature12347.
- Mouzaki M, Wang AY, Bandsma R, Comelli EM, Arendt BM, Zhang L, Fung S, Fischer SE, McGilvray IG, Allard JP, et al. Bile acids and dysbiosis in non-alcoholic fatty liver disease. PLoS One. 2016;11(5):e0151829. doi:10.1371/journal.pone.0151829.
- Loomba R, Seguritan V, Li W, Long T, Klitgord N, Bhatt A, Dulai PS, Caussy C, Bettencourt R, Highlander SK, et al. Gut microbiome-based metagenomic signature for non-invasive detection of advanced fibrosis in human nonalcoholic fatty liver disease. Cell Metab. 2017;25(5):1054–62.e5. doi:10.1016/j.cmet.2017.04.001.
- Shao L, Ling Z, Chen D, Liu Y, Yang F, Li L. Disorganized gut microbiome contributed to liver cirrhosis progression: a meta-omics-based study. Front Microbiol. 2018;9:3166. doi:10.3389/fmicb.2018.03166.
- Qin N, Yang F, Li A, Prifti E, Chen Y, Shao L, Guo J, Le Chatelier E, Yao J, Wu L, et al. Alterations of the human gut microbiome in liver cirrhosis. Nature. 2014;513(7516):59–64. doi:10.1038/nature13568.
- Duboc H, Rajca S, Rainteau D, Benarous D, Maubert M-A, Quervain E, Thomas G, Barbu V, Humbert L, Despras G, et al. Connecting dysbiosis, bile-acid dysmetabolism and gut inflammation in inflammatory bowel diseases. Gut. 2013;62(4):531–539. doi:10.1136/gutjnl-2012-302578.
- Heinken A, Ravcheev DA, Baldini F, Heirendt L, Fleming RMT, Thiele I. Systematic assessment of secondary bile acid metabolism in gut microbes reveals distinct metabolic capabilities in inflammatory bowel disease. Microbiome. 2019;7:75. doi:10.1186/s40168-019-0689-3.
- Fekry MI, Engels C, Zhang J, Schwab C, Lacroix C, Sturla SJ, Chassard C. The strict anaerobic gut microbe Eubacterium hallii transforms the carcinogenic dietary heterocyclic amine 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP). Environ Microbiol Rep. 2016;8(2):201–209. doi:10.1111/1758-2229.12369.
- Vollenweider S, Lacroix C. 3-hydroxypropionaldehyde: applications and perspectives of biotechnological production. Appl Microbiol Biotechnol. 2004;64(1):16–27. doi:10.1007/s00253-003-1497-y.
- Possemiers S, Rabot S, Espín JC, Bruneau A, Philippe C, González-Sarrías A, Heyerick A, Tomás-Barberán FA, De Keukeleire D, Verstraete W, et al. Eubacterium limosum activates isoxanthohumol from hops (Humulus lupulus L.) into the potent phytoestrogen 8-prenylnaringenin in vitro and in rat intestine. J Nutr. 2008;138:1310–1316. doi:10.1093/jn/138.7.1310.
- Heyerick A, Vervarcke S, Depypere H, Bracke M, De Keukeleire D. A first prospective, randomized, double-blind, placebo-controlled study on the use of a standardized hop extract to alleviate menopausal discomforts. Maturitas. 2006;54:164–175. doi:10.1016/j.maturitas.2005.10.005.
- Bolca S, Possemiers S, Maervoet V, Huybrechts I, Heyerick A, Vervarcke S, Depypere H, De Keukeleire D, Bracke M, De Henauw S, et al. Microbial and dietary factors associated with the 8-prenylnaringenin producer phenotype: a dietary intervention trial with fifty healthy post-menopausal Caucasian women. Br J Nutr. 2007;98(5):950–959. doi:10.1017/S0007114507749243.
- Nieuwdorp M, Vos WMD, inventors; Caelus Pharmaceuticals BV, Academisch Medisch Centrum, assignees. Method for preventing and/or treating insulin resistance. United States patent US 9623055B2. 2017 April 18.
- Feng Q, Zhang D, Qin Y, inventors; BGI Shenzhen, assignee. Biomarkers for colorectal cancer. World Intellectual Property Organization WO 2015018307A1. 2015 February 12.
- Cutcliffe C, Eid JS, Bullard JH, Schicklberger MF, Cheng AT, inventors; Whole Biome, assignee. Methods and compositions relating to microbial treatment and diagnosis of skin disorders. World Intellectual Property Organization WO 2016149687A1. 2016 September 22.
- Mulder IE, Holt AB, McCluskey SM, Lennon GC, Ahmed S, inventors; 4D Pharma Research, assignee. Composition comprising a bacterial strain of the genus Eubacterium. United States patent US 10391128B2. 2019 August 27.
