ABSTRACT
The functional diversity of the mammalian intestinal microbiome far exceeds that of the host organism, and microbial genes contribute substantially to the well-being of the host. However, beneficial gut organisms can also be pathogenic when present in the gut or other locations in the body. Among dominant beneficial bacteria are several species of Bacteroides, which metabolize polysaccharides and oligosaccharides, providing nutrition and vitamins to the host and other intestinal microbial residents. These topics and the specific organismal and molecular interactions that are known to be responsible for the beneficial and detrimental effects of Bacteroides species in humans comprise the focus of this review. The complexity of these interactions will be revealed.
Introduction
The human gut is home to one of the most dense and diverse microbial communities known.Citation1 The gene content of the gut microflora easily outnumbers that of the host by an astonishing 100-fold.Citation2 The gut microbiome includes a plethora of biological entities including bacteria, viruses, fungi, archaea, and protozoa.Citation3,Citation4 The human colon is the main site of habitation for bacterial residents with an estimated concentration of 1012/ml.Citation4
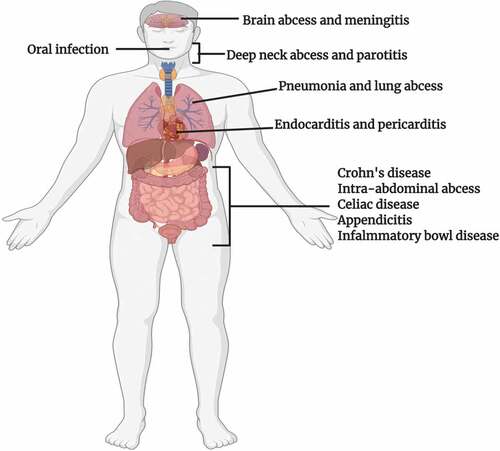
As the colon of the gastrointestinal tract (GIT) of mammals has the availability of diverse nutrient sources (derived from the host diet), this makes it a predilection site for numerous microbes.Citation5 Members of the genus Bacteroides are potential colonizers of the colon and account for a major fraction of the gut bacteriome.Citation6 These Gram-negative obligate anaerobes play multiple roles in the human gut bacteriome and are major players in sustaining the microbial food web of the gut.Citation7 As proven commensals, mutualists, and beneficial organisms, they not only play the role of “Providers” for the host and other microbes residing close to them, but also assist the host by providing numerous health benefits. Nevertheless, some species of Bacteroides may play dual beneficial and pathogenic roles based on their locations in the host, often being beneficial in the gut but opportunistic pathogens in other body locations. Common sites of Bacteroides infections and possible disease conditions are illustrated in . In this review we analyze the roles of Bacteroides species as beneficial organisms, gut competitors, and opportunistic pathogens. Also, recent relevant Bacteroides research findings will be evaluated.
Figure 1. Sites of infection and diseases caused by Bacteroides spp
1. The human gut glycome and bacteroides
A major factor that shapes the composition and physiology of the gut bacteriome is the influx of glycans into the intestines via host diet and mucosal secretions.Citation8 These glycans collectively form the human gut glycome, in which they are in constant interaction with each other and the microbial residents. The glycan landscape of the gut includes i) exogenous glycans derived from the host diet, ii) endogenous glycans expressed by the host cells, and iii) microbial glycans.Citation9 Conjugated glycoproteins and glycolipids are examples of gut glycans, and these may be either O-linked (attached to serine or threonine residues) like host mucins, or N-linked (attached to asparagine), attached to cell surfaces as the glycocalyx, or unconjugated oligoglycans, which are often found in plants and fungi.Citation10
In the human body mucus is present at the interface between many epithelial surfaces and their environments and is prevalent in the GIT.Citation11 Interactions between the gut bacteriome and mucus are considered to be pivotal for the assembly and stability of the microbiota that reside in the gut. The main component of the mucus layers in the GIT are mucins which are O-glycosylated glycoproteins.Citation12 The carbohydrate structures on these mucins are diverse due to which they can present microbes with a wide array of binding sites.Citation11 Recent work has established that the mucin- rich mucus layer acts as an essential barrier between the luminal microbiota and the underlying immune cells.Citation13
As gut commensals, Bacteroides spp. play multiple roles; they can provide protection from pathogens and supply nutrients to other microbial residents of the gut. Past research has revealed that mucin-type O-glycans are important contributors to their mutualistic roles and directly impact the interaction of Bacteroides spp with host tissues.Citation13 The Bacteroides thetaioatomicron (Bth) VPI- 5482 strain has 88 polysaccharide utilization loci (PUL) at its disposal for the degradation of various kinds of glycans including diet derived and host glycans.Citation14 The PUL of Bth enable it to effectively forage O-glycans during a shortage of glycans derived from plant polysaccharides.Citation15 Generally, mucins are considered to be important players in the fitness and stability of Bacteroides spp. Binding of Bth to mucins and their subsequent degradation regulates the genetic repertoire that assists in the synthesis of the outer capsule. B. fragilis (Bfr), like Bth, has genetic machinery to degrade and utilize glycans, including mucin-type O-glycans, for capsular polysaccharide synthesis, which is collectively required for optimal colonization and maintenance in the gut.Citation16 As Bth strains lack adhesive organelles, they utilize outer membrane glycan-binding proteins for attachment to food particles, mucus layers, and exfoliated epithelial cells.Citation17 Bth has a flexible glycan-foraging ability, and it allows easy switching to host polysaccharides when dietary polysaccharides become scarce. This imparts an overall stability to the ecosystem of the gut bacteriome, during nutritional deficiencies.
Mucins primarily play protective and lubricative roles, but they also facilitate microbial tropism by presenting glycans to bacterial residents of the gut including Bacteroides.Citation18 This, in turn, impacts the localization of these bacterial species and also gives them an extra nutritional source.Citation19 As such, mucin glycans have been predicted to be key players in the selection and thriving of bacterial communities across the gut bacteriome. Consistent with this prediction, recent research using mouse models and humans indicates an association between changes in mucin glycosylation profiles and deviations of overall bacterial community ecology along with altered abundances of Bacteroides strains.Citation18
2. Characteristics of polysaccharide utilization loci (PULs)
In the often nutrient-rich environment of the gut, one may assume that the microbes have easy access to desired nutrients, metabolizing them according to their physiological needs. However, in the colon, many of the desired nutrients, especially simple sugars, have already been absorbed and consumed in the small intestine. The remaining nutrients consist of long chain polysaccharides and oligosaccharides that are not readily absorbed by the epithelial cells of the colon, and resist digestion by host enzymes.Citation5 For access to these lumenal carbohydrates, bacterial residents may require 1) extracellular polysaccharide hydrolases, 2) receptor proteins on the bacterial cell surfaces, 3) appropriate sugar transport systems, and 4) cytoplasmic carbohydrate degrading enzymes.Citation9,Citation20,Citation21
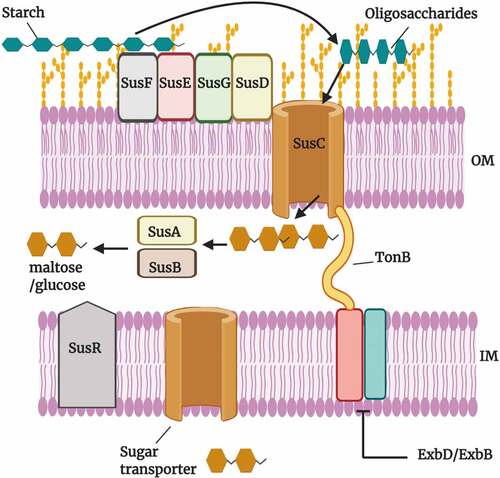
The polysaccharide utilization loci (PULs) of Bacteroides spp. may include secreted glycosidases, a complement of cell surface glycan-binding proteins, TonB-dependent outer membrane oligosaccharide receptor/transporters, uptake porters in the cytoplasmic membranes, and cytoplasmic carbohydrate-metabolic enzymes.Citation20 Syntheses of these proteins are activated depending on the availability of carbohydrate sensors and transcriptional regulators. These sophisticated PULs provide the major protein machinery for carbohydrate acquisition and the initiation of metabolism in many Bacteroides species.Citation9 These systems are pivotal to the colonization of nutritional niches and formation of gut-microbial ecosystems. One of the first identified PULs was the starch utilization system (Sus) of Bth.Citation21 The Sus of Bth includes numerous cell-surface proteins (SusDEF), a TonB-dependent outer membrane transporter (SusC), and three enzymes (SusABG) as shown in . The Sus proteins work in tandem for the capture and degradation of starch at the cell surface and for further digestion of the liberated malto-oligosaccharides in the periplasm. Many of the sequenced Bacteroides genomes possess PULs that show appreciable synteny with the Sus locus of Bth. However, there are some notable differences in the numbers of surface glycan-binding proteins like SusE and SusF. Also, the sequence similarities of these proteins differ as do the sizes of the predicted SusG hydrolase homologs.Citation21
Figure 2. The starch utilization system (Sus) of Bacteroides thetaiotaomicron.
The use of the PUL machinery by Bacteroides spp. enables them to get involved in inter-species cross-feeding relationships with their microbial neighbors. It has been shown that Bth influences the dynamics of flavonoid degradation and butyrate production of Eubacterium ramulus.Citation22 Flavonoids are phenolic compounds that are found in fruits and vegetables and arise as secondary metabolites in plants.Citation23 Quercetin is one of the most well characterized flavonoids and is the most abundant one found in nature. It has numerous health benefits, being anti-viral, anti-inflammatory, antioxidant and anti-carcinogenic.Citation24 Members of the gut microbiota, including E. ramulus, cleave the C-ring of quercetin during degradation and release 3,4-dihydroxyphenylacetate which has anti- proliferative activity in colon cancer cells.Citation25 Bth seems to lack the metabolic machinery to degrade quercetin, while E. ramulus lacks the ability to degrade starch. Bth metabolizes starch (PUL mediated) and provides both maltose and glucose to E. ramulus. In the presence of these sugars, E. ramulus can degrade quercetin while fermenting glucose to butyrate.Citation22 This PUL-mediated inter- species cross-feeding process is not only beneficial to gut residents for obtaining desired nutrients, it may also play beneficial roles for human health.
3. Outer membrane vesicles contribute to both health and disease
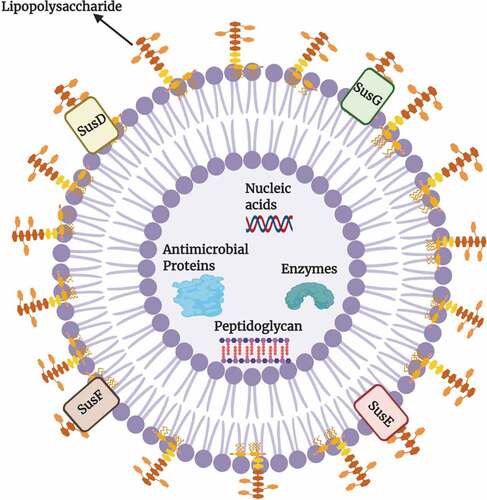
Enteric Gram-negative pathogens are known to produce outer membrane vesicles (OMVs) containing mediators of virulence.Citation26 These vesicles can be vehicles of pathogenicity as they can store and transport virulence factors over long distances.Citation27 In addition to spreaders of virulence factors, research indicates that these OMVs can be recognized as key modes of communication between bacterial spp. and host tissues, contributing to a wide array of functions including i) nutrient uptake, ii) transfer of genetic material, iii) biofilm formation, and iv) protection from antimicrobials.Citation28 Bacteroides have been shown to be major exporters of these OMVs with B. fragilis (Bfr) and Bth being two main players.Citation29,Citation30 The structure of a Bacteroides OMV is shown in . The glycosidases, lipid hydrolases and proteases present in these vesicles help recipient bacterial species break down complex polysaccharides, proteins, and lipids to obtain monosaccharides and small oligosaccharides, amino acids and peptides, and fatty acids and other lipid breakdown products.Citation31 As a result, the hydrolases of the OMVs play pivotal roles in the gut microbial ecosystem. By providing recipient bacteria (often called “cheaters”) with the required nutrients, the OMVs support the growth of other bacteria in the gut and contribute to overall gut homeostasis.Citation32
Figure 3. Structure of an Outer membrane vesicle (OMV) of Bacteroides.
The OMVs of Bth contain glycosyl hydrolases that help in the degradation of levan, a common non-structural carbohydrate in plants. The by-products of levan degradation include extracellular fructo-oligosaccharides that are important for the growth of other Bacteroides spp.Citation33–35 Another example of OMV-associated intra-genus support involves Bacteroides ovatus and Bacteroides vulgatus. The glycosyl hydrolases in the OMVs of B. ovatus break down inulin, the products of which are utilized by B. vulgatus.Citation36 Thus, the genus Bacteroides is an efficient public goods provider, and its services generally support other species in the microbial gut community.
4. Competition for shared nutrients in the gut
Gut microbes confront each other for available nutrients around them. Bacteroides spp. take part in exploitative competition and partially degrade polysaccharides for their own use instead of allowing other microbes to utilize them. For example, Bth only partially degrades the polysaccharide α-mannan and transports the products into its periplasm for further degradation into manno-oligosaccharides and mannose.Citation37 As many other microbes do not encode the transporters and enzymes required for the use of these partially degraded polysaccharides, they can be used exclusively by the bacteria that do have them. This illustrates how Bth can use its metabolic repertoire to increase its competitive advantage over other gut residents.
4.1. Antimicrobial toxins are used in bacterial competition
Bacteroides spp. take part in interference competition by the secretion of antimicrobial toxins in a contact-independent manner. After secretion of diffusible toxins from the cell, Bacteroides spp. may utilize various transporters (such as ABC exporters) to actively secrete the toxins to the environment of their microbial targets.Citation38 Several of the diffusible toxins produced by Bacteroides spp. have membrane attack complex/perforin (MACPF) domains. These domains are ubiquitous in eukaryotic cells and are involved in immunity and defense.Citation39 The first recognized Bacteroidales secreted antimicrobial protein (BSAP-1) was found in approximately 44% of Bfr strains. As a lipoprotein, it is released as cargo within OMVs into the cell surroundings, and it kills target bacteria by pore-formation.Citation40 BSAP-1 targets a β-barrel outer membrane protein of sensitive Bfr strains, thus exhibiting intraspecies killing.Citation41 Bfr strains either possess the bsap-1 gene encoding the toxin, or they lack the gene and are sensitive to it.Citation42 Producers of BSAP-1 have a gene adjacent to the bsap-1 gene that encodes an orthologue of the target outer membrane protein that serves as the receptor for BSAP-1 in the target bacterium.Citation43 This orthologue is structurally similar to the target receptor but is sufficiently different, so as not to be targeted by the toxin, but to protect the producer from it. This renders the BSAP-1-producing strains resistant to a potential antimicrobial attack by its own toxin. In a mouse model of competitive colonization, it was shown that BSAP-1 producers have increased fitness in the presence of sensitive isogenic strains. Also, according to meta-genomic data, the co-residence of BSAP-1 producers and sensitive strains is a rare event in the mammalian gut, thus suggesting that BSAP-1 is a key tool for intra-species dominance in vivo.Citation41 Two other MACPF toxins (BSAPs 2 and 3) are produced by B. uniformis and B. dorei/B. vulgatus, respectively.Citation43 The mode of action of these two toxins may be similar to that of BSAP-1, but their receptor targets in the sensitive strains are different from that of BSAP-1, as they target lipopolysaccharides.Citation42 Bfr also encodes a eukaryotic-like ubiquitin protein (BfUbb) that gives it a competitive advantage in the gut via intraspecies antagonism. Unlike BSAP 1–3, the mechanism of action is still unknown. Chaztidaki-lavanis et al. suggested a mechanism in which BfUbb is transported into target cells via a protein-specific uptake system that acts on an intracellular target rather than the outer membrane or lipopolysaccharide as is the case for BSAP 1–3.Citation44 Recently, Shumaker et al. discovered another MACPF toxin called BSAP-4, which shows 42% similarity to BSAP-1. It targets outer membrane proteins with calycin-like domains that are exposed on the surfaces of target cells.Citation42
4.2. Type vi secretion systems and contact dependent interbacterial antagonism
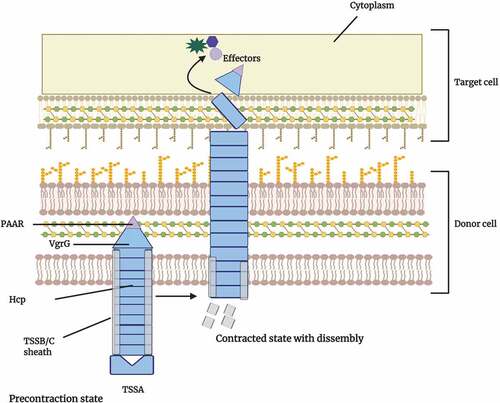
The generalized structures and topologies of Type VI secretion systems (T6SSs) are illustrated forBacteroides spp. Upon direct contact with target cells, these multi-protein machines release antimicrobial toxins, effectors that mediate interbacterial antagonism.Citation45 The T6SSs have structural and sequence similarity with the contractile tails of T4 bacteriophages, thus indicative of orthology between the systems.Citation46 After the secretion of T6SS effectors by the bacterium, synthesis of specific immunity proteins confers resistance to potential attacks by sister cells. Each effector protein is accompanied by a cognate immunity protein, typically encoded by a neighboring gene.Citation47
Figure 4. Model of the Bacteroidetes type VI secretion system
The T6SS loci of various Bacteroides spp. can segregate into three different genetic architectures, i) GA1, ii) GA2 and iii) GA3. The first two are present in numerous Bacteroides spp. while GA3 is exclusive to Bfr.Citation48 The wide distribution of GA1 and GA2 is due to their presence on integrative conjugation elements which facilitate their spread to other species.Citation49 Data from genomic and metagenomic analyses have revealed that the GA3 T6SS is found in 86% of Bfr strains.Citation50 These GA3 T6SSs seem to confer upon Bfr a competitive advantage in the gut as they antagonize other Bacteroides spp. and Parabacteroides spp. However other Bfr strains with the same GA3 T6SS region and immunity genes survive.Citation51 Analysis of infant bacteriomes showed that Bfr strains with GA3 structural genes are more prevalent (92%) compared to those in adults (74%).Citation52 This suggests the GA3 T6SS enables Bfr strains to dominate competition with other bacterial spp. during early gut bacteriome development. Numerous studies have been conducted in attempts to understand the role of GA3 T6SS in the gut microbiota of mice. In germ free mice, Bfr strains with the GA3 T6SS outcompete Bth strains that lack it, thus suggesting that the system provides Bfr with a competitive advantage for colonization.Citation53
The T6SS effectors of Proteobacteria are well characterized as are their targets in other bacterial cells including peptidoglycan, cell membranes and nucleic acids.Citation54 These anti-bacterial effectors can be classified into families of nucleases, phospholipases, peptidoglycan hydrolases and NAD(P)+-dependent glycohydrolases.Citation54 On the basis of homology, some putative effectors with similar functions have been predicted in Bacteroides spp., but many still remain unexplored with unknown functions and lacking recognizable domains. Due to a dependence on contact with the target cells, the target range of T6SS is limited to target cells within the immediate vicinity of the producers. T6SSs may prove to be important for Bacteroides spp. when competing locally for shared nutrients. This local action of T6SSs is in contrast to antimicrobial proteins which can exert effects on target cells at distant sites in the gut.
5. Prevalence of bacteroides species in children, adults and across different human populations
At birth, the gut is devoid of bacteria, but colonization starts shortly thereafter due to contact with the mother’s skin and environmental microbiota.Citation55 Microbial colonization of the gut is also dependent on the type of delivery method used during birth, as Bacteroides spp. have been found to be prevalent in the gut of infants delivered vaginally.Citation56,Citation57 Also, the gut bacteriome of infants delivered vaginally has significant resemblance to their mothers. In the infant gut, the Bacteroides population is variably abundant, and infants fed with formula milk have a higher percentage of Bacteroides spp. as compared to the breast fed.Citation58 Backheld et al. analyzed the development of the infant gut bacteriome during the first 12 months.Citation59 By the age of 4 months, an increased production of amino acids and vitamins was observed. During the 10–12 months period the gut bacteriome of infants showed an increase in the expression of genes for the degradation of complex sugars. This was attributed to a higher abundance of Bth, known for its vast glycan degrading repertoire and ability to degrade human milk oligosaccharides.
Overall, microbial diversity of the gut reaches a fairly stable composition at the age of 3 years with the Bacteroidetes phylum as one of the three major phyla (Firmicutes and Actinobacteria being the other two).Citation60 The introduction of solid food causes an increase in the bacterial load and diversity in the gut.Citation61 This is due to higher total short-chain fatty acid levels, and a dominance of Bacteroides spp. that are adept degraders of complex glycans. Also, dietary habits such as high fiber and animal protein foods can cause an increase in the Bacteroides population.Citation62
Various studies have been conducted to analyze the gut bacteriome in adults and children to get comparative insight into the respective microbiota. The most common techniques involved in these comparative studies have been 16S rRNA gene sequencing and shotgun metagenomic sequencing. Hollister et al. analyzed the gut microbiome of children of the age range of 7–12 years in Texas, USA by 16S rRNA sequencing and reported that members of the Bacteroides genus accounted for nearly 40% of the average healthy child gut bacteriome.Citation63 Despite the comparative findings that both child and adult gut bacteriomes contain similar numbers of operational taxonomic units (OTUs), significant differences among the two groups were observed with respect to Shannon and Simpson diversity indices. Overall, Bacteroides was the common genus among children and adults with some species having a similar prevalence in the two groups. However, species that were more prevalent in the adult gut on the basis of 16S reads included B. vulgatus and B. xylanisolvens. Another study on the comparative gut composition of children (1–4 years) and adults in North Carolina, USA indicated that Bacteroides spp. were more prevalent in children.Citation64 Zhong et al. examined the composition of the gut bacteriome of 281 school-going children (6–9 years) in the Netherlands.Citation65 Variation among the prevalence of various Bacteroides spp. was observed. The most prevalent species on the basis of detected annotated genes were B. ovatus followed by Bfr, Bth and B. xylanisolvens. Interestingly, there was a larger prevalence of Bacteroides spp. in the gut of children.
The prevalence of Bacteroides spp. in the adult gut depends mainly on different factors such as diet, environment, and antibiotic use.Citation66 However, important factors include dietary patterns, and the prevalence of species may vary in vegan, vegetarian and omnivorous diets. In a study based on the effects of dietary patterns on the gut bacteriome, Ferrocino et al. examined the fecal microbiota of 153 healthy volunteers (51 vegans, 51 vegetarians, and 51 omnivores) from four different locations in Italy.Citation67 Bfr was present in lower numbers in both vegans and vegetarians but was highly prevalent in the omnivorous participants. Data from the V3 region of 16S rRNA gene sequences showed a prevalence of B. salanitronis and B. coprocola in the omnivorous group, while B. vulgatus was specific to the vegetarians and B. salyersiae was prevalent in vegans. The prevalence of Bacteroides spp. in general has been linked to animal-based diets; however, Bth is prevalent in vegans and vegetarians.Citation68–70 Apparent discrepancies are common during categorization of certain bacterial spp. prevalence under a vegan/vegetarian diet vs an omnivorous diet. In this respect some Bacteroides spp. may play the role of outliers. This discrepancy in bacterial prevalence in different dietary patterns has been attributed to various causes, such as i) different methodologies for gut bacteriome profiling ii) variation in host genetics, iii) body mass index, and iv) consumption of red wine and aspartame (sugar substitute). 69,70
The Gut bacteriome varies among individuals across different geographical locations around the globe. The overall structure of the gut bacteriome is influenced by different intrinsic and extrinsic factors such as, i) physiology and genetics of the host, ii) health and disease, iii) antibiotic use, and iv) diet.Citation71 As dietary patterns vary across human populations in different geographical locations, so prevalence of Bacteroides spp. is also subject to variation. It has been demonstrated that Bacteroides species are prevalent in the guts of people living in Western countries (North America and Europe), as western diets are often high in fat and protein content.Citation72 Prevalence of Bacteroides spp. in children, adults and across different geographical locations is summed up in .
Figure 5. Prevalence of Bacteroides spp. in children, adults and across different geographical populations
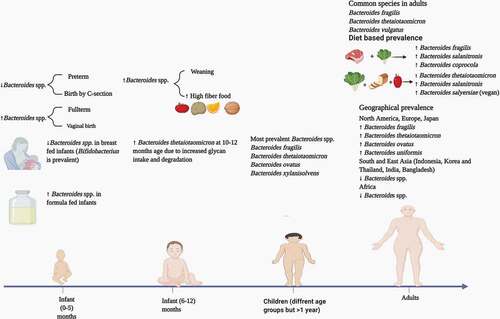
Generally, in Asian countries, fat and protein consumption is minimal, and carbohydrates (in the form of rice and wheat) are consumed widely.Citation73 However, due to diverse cultures and diet patterns, this cannot be considered as a trend as in many Asian populations, Bacteroides spp. have been observed to be present in significant numbers. For example, in a comparative study on the gut microbiota of populations from Japan and India, higher number of Bacteroides spp. were observed in Japanese participants who consumed a diet of animal origin in comparison to Indian adults who consumed a more plant-based diet.Citation74 The most prevalent species in the Japanese adults were Bfr, Bth, B. ovatus, and B. uniformis. A study on the prevalence of Bacteroides spp. in Belgium adults showed an abundance of B. uniformis, B. vulgatus and B. ovatus, similar to the data concerning Japanese adult gut bacteriomes.Citation75 Inhabitants of Indonesia, Korea and Thailand consume less meat and have a low prevalence of Bacteroides spp.Citation76
6. Bacteroides and their pathogenic characteristics
Bacteroides spp. are generally ‘friendly’ commensals while residing in the gut, but they tend to become opportunistic pathogens when lodged elsewhere.Citation77 For example, Bfr, a gut symbiont, has been found to be an opportunistic pathogen among the Bacteroides spp., as it is also the most common isolate from intra-abdominal abscesses.Citation78,Citation79 The translocation of Bacteroides spp. takes place through the intestinal mucosa into the normally sterile tissues, eventually producing different disease conditions.Citation8 This translocation from the gut to extraintestinal locations in the body may be attributed to various factors: i) a compromised immune system, ii) gut barrier disruption (leaky gut), iii) surgical injuries, iv) excessive antibiotic use and v) aging.Citation80,Citation81 Also, dietary patterns may lead to a lack of competition by other gut commensals due to which an overgrowth of Bacteroides spp. may occur. For example, an over-abundance of Bacteroides caccae results in the increased degradation of mucus, which helps reduce intestinal inflammation by decreasing bacterial interactions with intestinal epithelial cells in the intestine generally, and in the colonic mucus barrier specifically.Citation82 Due to thinner layers of mucus, the intestinal barrier function becomes compromised, resulting in the expulsion of potential pathogens to extraintestinal locations. In addition, Bacteroides spp. may transfer virulence genes, thus equipping sister cells and neighbors with virulence factors that may assist in pathogenesis in extra-intestinal organs.Citation83
Initially, during early infiltration in the extra-intestinal organs, aerobic bacteria dominate and cause tissue damage.Citation84 Then, the redox potential of oxygenated tissues decreases, and anaerobes such as Bacteroides begin to thrive, leading to inflammation, diarrhea, and the formation of intra-abdominal abscesses.Citation8 The dissemination of Bacteroides spp. outside of the gut lumen can thus lead to bacteremia and abscess formation in different body parts, sometimes even in the central nervous system.Citation85,
6.1. Virulence factors of Bacteroides
Bacteroides spp. possess some of the most complex polysaccharide capsular systems among bacteria, consisting of at least eight different polysaccharides (PSA – PSH).Citation86 A common function of the capsule is adherence to peritoneal surfaces. However, it also provides resistance to phagocytosis, thus playing an important role in bacterial fitness outside the colon.Citation87,Citation88 The lipopolysaccharides of Bacteroides spp. lack O-antigen and are approximately 1000 times less virulent than the lipopolysaccharide of E. coli.Citation89,Citation90 Like Salmonella spp., Bth chemically modifies the lipid A portion of the LPS, thereby increasing resistance to a subset of antimicrobial peptides.Citation91
Bfr can be classified into two subtypes based on their pathogenic potential 1) non-enterotoxigenic strains that do not encode the Bfr toxin, and 2) enterotoxigenic Bfr strains that do have the bft genes that encode the toxin.Citation92 Toxigenic strains of Bfr have been associated with different disease conditions of the human gut, including ulcerative colitis, toxin-mediated acute diarrhea, and bacteremia.Citation93,Citation94 The Bfr toxin (fragilysin) is one of the best-researched virulence factors among Bacteroides spp. It exists in three isoforms, BFT 1–3, with BFT-2 having the greatest potential to elicit tissue damage. Among isolates from humans, BFT-1 is the most common toxin variant, while the BFT-3 has a geographical propensity for Southeast Asia.Citation95,Citation96 As a heat-labile nickel ion-dependent metalloprotease, it shares substantial similarity with the tetanus and botulinum toxins. BFT is produced as a pre-protein and is cleaved by the fragipain cysteine protease to form the mature 20 kDa secreted toxin that is enterotoxic and cytotoxic in lamb ileal loop assays and HT29 cell lines.Citation97
Proteases of the C10 family are pivotal virulence factors in a variety of bacterial species such as Streptococcus pyogenes and Prevotella intermedia.Citation98 Thornton et al. reported homologues of the streptococcal virulence factor, SpeB, in Bfr with the encoding genes (bfp 1–4) located on mobile genetic elements, thus indicative of horizontal gene acquisition.Citation99 A detailed understanding of the mechanism of action of these Bfr toxins will require future research, but due to structural similarity to SpeB, the proposed action includes cleavage of cytokines, immunoglobulins, extracytoplasmic matrix proteins and fibronectin.
Many Gram-positive and Gram-negative bacteria, produce hemolysins/cytolysins that are powerful virulence factors which lyse and kill host immune cells.Citation100–102 These virulence factors not only contribute to the survival of the pathogens by providing access to nutrients but also weaken the immune system of the host.Citation103–105 These hemolysins/cytolysins may be used by opportunistic pathogens to develop system infections in the host. Also, gut commensals may employ these toxins for advantage in the highly competitive gut environment. Robertson et al. identified ten hemolysin paralogs, HlyA to HlyI and HlyIII, encoded within the genome of Bfr.Citation104 Further studies by Lobbo et al. showed that the expression of the hemolysins increases in an oxygen-rich environment and decreases during infection, mediated by the iron-dependent Fur transcriptional regulator. Bfr mutant strains (lacking genes hlyA/B) showed reduced fitness both in vitro and in vivo, suggesting that hemolysins may have roles in bacterial colonization of the gut.Citation83 To date, clear evidence is lacking regarding the specific role of hemolysins in the pathogenesis of Bfr diseases. In our previous study on Bacteroides (Zafar and Saier, 2018), we observed disparate patterns of distribution of hemolysins among seven strains including Bfr and Bth.Citation105 Only in the Bfr strain, did we observe a homologue of Hemolysin III, a powerful virulence determinant of Bacillus cereus.
The type 9 secretion system (T9SS) is a protein export pathway of the Fibrobacteres-Chlorobi-Bacteroidetes superphylum and has been associated with periodontal diseases in humans.Citation106 The components of the T9SS complex are not similar in sequence to those of other well-studied bacterial secretion systems. Studies on these systems indicate their role in the secretion of virulence factors that damage human tissues and manipulate host immune responses.Citation107 Other potential pathogenic functions include biofilm formation, adhesion and motility.Citation108,Citation109 So far, genomic studies have shown that components of T9SSs are present in a minority of the Bacteroides spp. including Bfr and Bth. However, the system is functional in the oral pathogen, Bacteroides forsythia, and the cargo proteins of its T9SS contribute to the evasion of host innate immunity.Citation110 Future studies aimed at revealing the presence of these virulence-promoting systems in other Bacteroides spp. will be of considerable interest.
6.2. Oxidative stress responses as virulence factors and protective mechanisms
As gut residents, Bacteroides spp. are exposed to various oxygen concentrations. During extraintestinal infections, Bacteroides translocate to the more oxygenated (up to Citation7% O2) peritoneal cavity, where additional oxidative stress is exerted by the host immune response, including the recruitment of polymorphonucleocytes.Citation111,Citation112 The ability to survive oxidative stress is a key virulence factor,Citation113 as pathogens must be able to withstand the conglomerate of oxidative host responses. This increased aerotolerance is achieved by the actions of various oxidoreductases including catalases, peroxidases and thioredoxins. In addition, the transcription factor OxyR is pivotal for the induction of numerous genes involved in oxidative stress response pathways.Citation114
The Bfr genes katA, ahpC and tpx encode catalase, alkyl hydroperoxidase and thioredoxin peroxidase, respectively. These proteins assist in the oxidative stress response by detoxifying peroxides.Citation115 Bacterioferritin co-migratory proteins are encoded within the genomes of numerous bacterial species. These proteins are members of the thiol-specific antioxidant protein family and play key roles in the prevention of free radical formation and resultant cellular oxidative damage.Citation116 Studies with Bfr by Nicholson et al. suggested that the bacterioferritin co- migratory protein, encoded within the recA operon, may play a role in maintaining metabolic fitness and genomic integrity in response to oxidative stress.Citation117 This may be accomplished by assisting in the reduction of hydroperoxides, thereby preventing lipid oxidation and DNA damage during oxidative stress.
6.3. Role of bacteroides species in oncogenesis
Recent research on the human gut microbiome has suggested that the microbiota play pivotal roles in the genesis of various types of cancer in humans.Citation118–120 A dysbiotic gut is more prone to cancer, as pathogens can exert negative effects on the host’s physiology, metabolism, and immune system, thus promoting tumor growth. It has been shown that gut dysbiosis is apparently linked to the growth of both local and distal tumors in the host.Citation121
Spermine oxidase is an FAD-dependent enzyme that oxidizes spermine and is generally important for the catabolism of polyamines in mammals.Citation122 The oxidative products of spermine oxidase activity are spermidine, the reactive oxygen species, hydrogen peroxide, and the aldehyde, 3-aminopropanal, each with the potential to produce cellular damage and aggravate pathogenesis.Citation123 With Bfr, activation of the host’s spermine oxidase can occur, which, in turn, generates hydrogen peroxide and other reactive oxygen species that contribute to DNA damage, increasing the prevalence of cancer.Citation124,Citation125
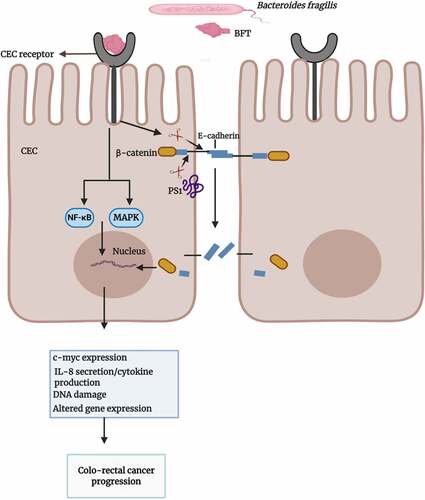
Recent research on enterotoxigenic Bfr has shown it to be a major initiator and promoter of colo-rectal cancer in humans.Citation126 The signaling pathways of colonic epithelial cells activated by Bfr is complicated with some mechanisms still being poorly understood. The zinc-dependent metalloprotease toxin of enterotoxigenic Bfr attaches to a colonic epithelial cell receptor and interacts with the host epithelial E-cadherin, a transmembrane protein essential for adhesion between colonic epithelial cells.Citation127,Citation128 Cleavage of E-cadherin results in the shedding of its 80- kDa extracellular ectodomain, followed by host cell presenilin-1/γ-secretase–mediated processing of the remaining intracellular fragment. This cleavage event leads to the disruption of intercellular junctions, activates the nuclear signaling protein, β-catenin, and induces the expression of the proto-oncogene, c-myc,Citation129 as shown in . The signaling mechanism may trigger cell proliferation and induce carcinogenic changes in the affected cells. BFT has been shown to activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and mitogen-activated protein kinase (MAPK) signaling pathways, leading to the release of interleukin-8 (IL-8) and tumor necrosis factor α (TNFα).Citation130,Citation131 IL-8 has been suggested to be a major player in tumor cell proliferation, tumor angiogenesis and growth.Citation132
Figure 6. Overview of the potential role of BFT in colo-rectal cancer
Around 5% of colorectal cancers occur in individuals who have an inherited mutation.Citation133 Familial adenomatous polyposis is a hereditary condition caused by germline mutations in the adenomatous polyposis coli tumor suppressor gene.Citation134 In this genetic background, numerous adenomatous polyps form in the epithelium of the colon, and malignant transformation may lead to colorectal cancer. Dejea et al. examined surgically resected tissue of patients with familial adenomatous polyposis and found that Bfr and E. coli (positive for the polyketide synthase (pks) island responsible for the synthesis of genotoxic colibactin) dominated the observed biofilms.Citation135 Further experiments in mice showed that mono-colonization with either species resulted in few to no tumors. However, tumorigenesis was observed in the mice colonized with both species. In vitro trials using mucin monolayers indicated that degradation of mucin by Bfr enhanced colonization by E. coli. Thus, a shift in the niche of this species could facilitate the delivery of genotoxic colibactin to colonic epithelial cells, increasing the risk of mutations in genes such as that of adenomatous polyposis. This revealed another association of Bacteroides spp. with another gut resident, this time as a partner in crime for the initiation of colon tumorigenesis.
In another study, Toprak et al. detected the bft gene in stool samples of 38% of colo-rectal cancer patients, but in only 12% of samples taken from a healthy control group.Citation136 Similar patterns of bft gene detection were reported by Haghi et al.Citation137 Over the past decade, evidence obtained from next generation sequencing has suggested a relationship between the gut microbiome and breast cancer.Citation138 This type of cancer is a global health concern as 1 in 8 women are expected to be affected in their lifetimes.Citation139 Approximately 65% of breast cancers are diagnosed as hormone receptor positive (HR+), that in cancerous cells, have receptors for estrogen (ER+) and progesterone (PG+). A mouse model of hormone receptor-positive breast cancer suggested that prior gut dysbiosis in the mice led to a significant increase in tumor dissemination in the blood, lungs, and distal lymph nodes.Citation140 This finding of tumor metastasis to distant sites supports the notion that the gut microbiome can be considered as an “endocrine gland”, at least when subjected to dysbiosis. Several bioactive metabolites secreted by gut microbial residents have been shown to affect immune cell functions and breast cancer cell growth in in vitro studies. Among these metabolites, short chain fatty acids, lithocholic acid, reactivated estrogen and cadverine can cause epithelial mesenchymal transitions and modulate mitochondrial metabolism in breast cancer.Citation141
Relevant to the present review, Bacteroides spp. are efficient producers of these metabolites. A recent murine study of antibiotic-based gut dysbiosis showed that cephalexin (a cephalosporin-type antibiotic) accentuated the decrease in microbiome diversity that was induced by the tumor itself, and it induced tumor formation, indicating that antibiotic use and breast cancer incidence may be interlinked.Citation142 This also supports the notion that a decrease of gut bacterial diversity correlates with cancer occurrence and metastasis. In the mouse gut, cephalexin decreased the number of butyrate producing bacteria including Odoribacter and Anaeotruncus, and increased the number of Bacteroides spp. As cephalexin is routinely used as a pre-surgery medication for breast cancer patients, future studies on the effects of other commonly used antibiotics on the gut microbiota will be of interest.
In a comparison of the gut microbiomes of healthy women and patients with invasive breast cancer, Cambell et al. reported increased levels of Bacteroides spp. in the cancer patients.Citation143 In another study by Parida et al., mice infected with enterotoxigenic Bfr exhibited morphological changes in the mammary glands.Citation144 Bfr toxin pretreated MCF7 cells exhibited increased tumor growth along with multifocal tumors in the mice models. MCF10A-KRas cells pretreated with Bfr toxin also showed increased tumor progression and multifocal tumors in mice. In vivo limiting dilution assays using breast tumors from Bfr toxin-pretreated MCF7 cells revealed a striking increase in tumor-initiating cells. Follow-up analyses of these tumors demonstrated increased migratory, invasive, and mammosphere-forming behavior, confirming that brief Bfr toxin exposure elicits long-term molecular changes in the cells.
6.4. Bacteroides and autoimmune disorders
The gut microbiota can be considered to be analogous to a fully functional organ of the body.Citation145 Occasionally, the host relationship with the microbial residents of the gut has few defined boundaries. Thus, comes the issue of the commonality of epitopes of the gut residents and the human body. It is possible that under certain conditions, some gut microbes can give rise to autoimmune (immune reaction to self-epitopes) conditions.Citation146,Citation147
It has been hypothesized that molecular mimicry by proteins of bacteria and viruses in the human body may lead to autoimmune conditions.Citation148 Recently, a shift in the symbiotic gut environment to a dysbiotic one has been linked to the occurrence of autoimmune conditions in humans.Citation149 A case of molecular mimicry has been observed in the genome of Bfr, where the gene, ubb, encodes BfUbb. This protein has been found to be 63% identical to the human ubiquitin, Ubc52.Citation150 Due to the similar molecular architecture of these two proteins, BfUbb may act as a mimic protein and generate antibodies that cross-react with self-epitopes. Ubiquitin plays several important physiological roles in the human body including regulation of the immune response and prevention of an autoimmune response to cellular debris by masking it.Citation151 Doubtlessly, the generation of cross-reactive anti-ubiquitin antibodies could play a role in the onset of autoimmune conditions.Citation152
Autoimmune inflammatory cardiomyopathy is a condition in which inflammation of the heart muscle is associated with impaired function of the myocardium.Citation153 Different genetic and environmental factors may be responsible for the onset of this condition.Citation154 However, a recent study by Gil-Cruz et al. has revealed that Bth may also play an important role in the occurrence of this condition.Citation155 The authors found that Bth encodes a cross-reactive β-galactosidase mimic peptide, which can activate myosin-specific T-cells in the gut and can also induce a humoral response involving IgA and IgG in response to gut commensals. Briefly, they showed that inflammation occurring in the gut results in migration of the immune cells to the heart. This seems to trigger autoimmunity accompanied by cardiomyopathy.
7. Bacteroides as beneficial microbes for human health
Bacteroides spp. appear to be key players in the immunomodulation of the human immune system. Bfr expresses eight capsular polysaccharides, and the immunomodulatory effects of capsular polysaccharide A (PSA) have been the subject to extensive research.Citation156 Documented beneficial effects of PSA in a nutshell include (i) stimulation, development, and homeostasis of the immune system,Citation157 and (ii) prevention of bacterial and viral infections.Citation158,Citation159 PSA is packaged into OMVs and delivered to host cells.Citation160
7.1. PSA of bacteroides fragilis, “an efficient immunomodulator”
Research on the effects of microbial peptides and carbohydrates on the immune system led to the concept that only peptides can induce adaptive T-cell responses. However, it is now established that zwitterionic-polysaccharides (carrying both negative and positive charges) such as PSA can induce CD4+ T cell–dependent immune responses.Citation161 Toll like receptors (TLRs) comprise a family of transmembrane pattern recognition receptors that detect different but overlapping microbial components such as pathogen-associated molecular patterns (PAMPs), and they assist in eliminating these PAMPs host cells via signal transduction. TLR’s can reside on the cell surface (TLR’s 1,5,6 and 10) or localize to the endosome (TLR’s 3,7, 8, 9 and 11) although TLR2 and 4 are found in both compartments.Citation162,Citation163 Bfr uses the ability of its PSA to activate TLR2 signaling for localization in mucosal niches of the gut,Citation164 while removal of TLR2 from CD4+ T cells results in an immune response against Bfr, thus limiting potential bacterial colonization.Citation165 Overall, PSA influences CD4+ T-cell development, regulates the immune balance of T-helper cells (Th1/Th2), and activates immunomodulatory IL-10.Citation166
Gut microbiota-virus interactions have been subject to extensive research in models of germ- free mice (born sterile and kept in a sterile environment) and antibiotic treated mice.Citation167 The gut microbes may enhance, reduce, or have no effect on viral infections.Citation168 These multifaceted roles in response to viral infections are possible by either direct modification of the virion or by immunomodulation of host responses.Citation169 There has been recent interest in the role of Bacteroides spp. and their metabolites (specifically PSA) in equipping the immune system to combat viral infections. The role of PSA during Herpes simplex encephalitis (the most common type of fatal sporadic encephalitis in humans) was analyzed by Ramakrishna et al. in a murine model.Citation159 This condition is caused by the herpes simplex virus 1 (HSV 1), and a review of past research on this type of pathogenesis indicates that during infection, immune pathology may lead to the uncontrolled dissemination of inflammatory neutrophils and monocytes into the brainstem.Citation170 In the immunomodulatory analysis of PSA during viral infection, pre-treatment of mice with PSA was followed by infection with HSV1 and delayed treatment with Acyclovir, the antiviral drug of choice. The PSA-treated mice exhibited high survival rates as compared to controls (pre-treatment with PBS), and decreased levels of brainstem inflammation were also observed. A comparison with other mice that lacked B-cells and IL-10 showed that IL-10 was the main anti-inflammatory factor secreted by CD4+ and CD8+ T-cells. These T-cells seem to be induced by the binding of the PSA to B-cells. This suggests that PSA of Bfr may provide robust protective anti-inflammatory responses during viral infections.
7.2. Other bacteroides-mediated benefits
Metabolites secreted by different Bacteroides spp. assist in maintaining stability of the immune system. These species are primary producers of short-chain fatty acids in the human gut, mostly in the form of acetate and propionate. These are important for the maintenance of intestinal homeostasis.Citation171 Both acetate and propionate are potent anti-inflammatory mediators as they inhibit the release of pro-inflammatory cytokines from neutrophils and macrophages.Citation172 Cruz-Bravo et al. described an anti-cancerous role of propionate which induced apoptosis in human colon carcinoma cells.Citation173 Also, butyrate increases expression of tight-junction proteins in the gut to reduce potential gut hyperpermeability. This, in turn, decreases inflammation and endotoxemia that are associated with leaky gut.Citation174 In the human gut, Bacteroides spp. are the principal synthesizers of Vitamin K, which is mainly produced by members of the human gut bacteriome.Citation175 It may prevent or treat osteoporosis by increasing the bone mineral density.Citation176
8. Conclusions
Bacteroides can be regarded as the quarterback of a human gut football team. They try to pass the best available nutrients and beneficial metabolites to their teammates. Like a quarterback played out of position, a Bacteroides specie will not be able to perform its advantageous roles, and in turn will start eliciting adverse effects on the performance of its most important teammate, the human host. Thus, in the proper body location and under appropriate environmental conditions, it will be a good friend, but in an inappropriate location, it may become a foe. It is amazing how these bacterial spp. are equipped with such sophisticated metabolic machinery that enables them to perform various roles (commensals, beneficial microbes, and opportunistic pathogens), reflective of an extended co-evolutionary process. In this regard, it is important to note how these bacteria can influence cancerous cell growth either positively or negatively. Some strains of Bfr may be promoters of various types of cancers, by inducing different physiological changes in the host that may result in DNA damage. Also, enterotoxigenic Bfr strains have proven to be carriers of cancer-promoting toxins in humans. In addition, Bfr may enhance the colonization of certain enteric pathogens in individuals with hereditary conditions leading to the formation of certain cancer types.Citation135 Another mechanism that may lead to cancer formation in the colon involves biofilms produced by certain Bacteroides spp. As this microbial structure may be formed in the ascending colon and contains certain metabolites (such as polyamines), it may increase the concentrations of reactive oxygens species.Citation177
Bacteroides spp. are also considered to be key players in cancer immunotherapy and prevention. For example, B. xylanisolvens DSM 23964 has been shown to promote the maturation of natural antibodies against various cancers in humans.Citation178 Another commensal strain, B. ovatus ELH-B2, has shown promise as a vaccine candidate for the development of a Thomsen-Friedenreich antigen (TFα)-specific anti-tumor vaccine.Citation179 In a nutshell the properties of cancer prevention and immunotherapy among these organisms could be related to the type of strains used and their functional capacities.
Further research on the role of these species as, “potential anticancer probiotics” could lead to improvements in existing cancer therapies. Future research will help to reveal physiological and metabolic details about the less well understood Bacteroides strains and their interactions. The role of polysaccharide A of Bfr, for example, in immunomodulation during viral infection is a fascinating but underdeveloped subject. Investigations focusing on its robust, protective, anti-inflammatory roles during various viral infections will certainly be a topic of increasing interest.
Disclosure statement
The authors have no conflicts of interest to disclose.
References
- Thursby E, Juge N. Introduction to the human gut microbiota. Biochem J. 2017;74(1823–36):3. doi:10.1042/BCJ20160510.
- Bull MJ, Plummer NT. Part 1: the Human Gut Microbiome in Health and Disease. Integr 5 Med (Encinitas). 2014;13:17–20.
- Barko PC, McMichael MA, Swanson KS, Williams DA. The Gastrointestinal Microbiome: A Review. J Vet Intern Med. 2018;32(1):9–25. doi:10.1111/jvim.14875.
- Sender R, Fuchs S, Milo R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016;14(8):e1002533. doi:10.1371/journal.pbio.1002533.
- Wexler AG, Goodman AL. An insider’s perspective: bacteroides as a window into the microbiome. Nat Microbiol. 2017;2:17026. doi:10.1038/nmicrobiol.2017.26.
- Kim S, Covington A, EG P. The intestinal microbiota: antibiotics, colonization resistance, and enteric pathogens. Immunol Rev. 2017;279(1):90–105. doi:10.1111/imr.12563.
- Wexler HM. Bacteroides: the good, the bad, and the nitty-gritty. Clin Microbiol Rev. 2007;20(4):593–621. doi:10.1128/CMR.00008-07.
- Koropatkin NM, Cameron EA, Martens EC. How glycan metabolism shapes the human gut microbiota. Nat Rev Microbiol. 2012;10(5):323–335. doi:10.1038/nrmicro2746.
- Flint HJ, Scott KP, Duncan SH, Louis P, Forano E. Microbial degradation of complex carbohydrates in the gut. Gut Microbes. 2012;3(4):289–306. doi:10.4161/gmic.19897.
- McKeen S, Young W, Fraser K, Roy NC, McNabb WC. Glycan Utilisation and Function in the Microbiome of Weaning Infants. Microorganisms. 2019:7. doi:10.3390/microorganisms7070190.
- Derrien M, van Passel MW, van de Bovenkamp JH, Schipper RG, de Vos WM, Dekker J. Mucin-bacterial interactions in the human oral cavity and digestive tract. Gut Microbes. 2010;1(4):254–268. doi:10.4161/gmic.1.4.12778.
- Corfield AP. The Interaction of the Gut Microbiota with the Mucus Barrier in Health and Disease in Human. Microorganisms. 2018:6. doi:10.3390/microorganisms6030078.
- Bergstrom KS, Xia L. Mucin-type O-glycans and their roles in intestinal homeostasis. 2 Glycobiology. 2013;23:1026–1037. doi:10.1093/glycob/cwt045.
- Martens EC, Chiang HC, Gordon JI. Mucosal glycan foraging enhances fitness and transmission of a saccharolytic human gut bacterial symbiont. Cell Host Microbe. 2008;4(5):447–457. doi:10.1016/j.chom.2008.09.007.
- Marcobal A, Southwick AM, Earle KA, Sonnenburg JL. A refined palate: bacterial consumption of host glycans in the gut. Glycobiology. 2013;23(9):4–1038. doi:10.1093/glycob/cwt040.
- Coyne MJ, Chatzidaki-Livanis M, Paoletti LC, Comstock LE. Role of glycan synthesis in colonization of the mammalian gut by the bacterial symbiont Bacteroides fragilis. Proc Natl Acad Sci U S A. 2008;105(35):13099–13104. doi:10.1073/pnas.0804220105.
- Chow J, Lee SM, Shen Y, Khosravi A, Mazmanian SK. Host-bacterial symbiosis in health and disease. Adv Immunol. 2010;107:243–274. doi:10.1016/B978-0-12-381300-8.00008-3.
- Tailford LE, Crost EH, Kavanaugh D, Juge N. Mucin glycan foraging in the human gut microbiome. Front Genet. 2015;6:81. doi:10.3389/fgene.2015.00081.
- Sonnenburg ED, Zheng H, Joglekar P, Higginbottom SK, Firbank SJ, Bolam DN. Specificity of polysaccharide use in intestinal bacteroides species determines diet-induced microbiota alterations. Cell. 2010;141(7):1241–1252. doi:10.1016/j.cell.2010.05.005.
- Grondin JM, Tamura K, Dejean G, Abbott DW, Brumer H. Polysaccharide Utilization Loci: fueling Microbial Communities. J Bacteriol. 2017:199. doi:10.1128/JB.00860-16.
- Foley MH, Cockburn DW, Koropatkin NM. The Sus operon: a model system for starch uptake by the human gut Bacteroidetes. Cell Mol Life Sci. 2016;73(14):2603–2617. doi:10.1007/s00018-016-2242-x.
- Rodriguez-Castano GP, Dorris MR, Liu X, Bolling BW, Acosta-Gonzalez A, Rey FE. Bacteroides thetaiotaomicron Starch Utilization Promotes Quercetin Degradation and Butyrate Production by Eubacterium ramulus. Front Microbiol. 2019;10:1145. doi:10.3389/fmicb.2019.01145.
- Tungmunnithum D, Thongboonyou A, Pholboon A, Yangsabai A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: an Overview. Medicines (Basel). 2018:5. doi:10.3390/medicines5030093.
- Zhao Y, Chen B, Shen J, Wan L, Zhu Y, Yi T. The Beneficial Effects of Quercetin, Curcumin, and Resveratrol in Obesity. Oxid Med Cell Longev. 2017;2017:1459497. doi:10.1155/2017/1459497.
- Gao K, Xu A, Krul C, Venema K, Liu Y, Niu Y. Of the major phenolic acids formed during human microbial fermentation of tea, citrus, and soy flavonoid supplements, only 3,4– dihydroxyphenylacetic acid has antiproliferative activity. J Nutr. 2006;136(1):52–57. doi:10.1093/jn/136.1.52.
- Jan AT. Outer Membrane Vesicles (OMVs) of Gram-negative Bacteria: A Perspective Update. Front Microbiol. 2017;8:1053. doi:10.3389/fmicb.2017.01053.
- Ellis TN, Kuehn MJ. Virulence and immunomodulatory roles of bacterial outer membrane vesicles. Microbiol Mol Biol Rev. 2010;74(1):81–94. doi:10.1128/MMBR.00031-09.
- Jones EJ, Booth C, Fonseca S, Parker A, Cross K, Miquel-Clopes A. The Uptake, Trafficking, and Biodistribution of Bacteroides thetaiotaomicron Generated Outer Membrane Vesicles. Front Microbiol. 2020;11:57. doi:10.3389/fmicb.2020.00057.
- Zakharzhevskaya NB, Vanyushkina AA, Altukhov IA, Shavarda AL, Butenko IO, Rakitina DV. Outer membrane vesicles secreted by pathogenic and nonpathogenic Bacteroides fragilis represent different metabolic activities. Sci Rep. 2017;7:5008. doi:10.1038/s41598-017-05264-6.
- Bryant WA, Stentz R, Le Gall G, Sternberg MJE, Carding SR, Wilhelm T. In Silico Analysis of the Small Molecule Content of Outer Membrane Vesicles Produced by Bacteroides thetaiotaomicron Indicates an Extensive Metabolic Link between Microbe and Host. Front Microbiol. 2017;8:2440. doi:10.3389/fmicb.2017.02440.
- Elhenawy W, Debelyy MO, Feldman MF. Preferential packing of acidic glycosidases and proteases into Bacteroides outer membrane vesicles. mBio. 2014;5:e00909–14. doi:10.1128/mBio.00909-14.
- Cecil JD, Sirisaengtaksin N, O’Brien-Simpson NM, Krachler AM. Outer Membrane Vesicle-Host Cell Interactions. Microbiol Spectr. 2019:7. doi:10.1128/microbiolspecPSIB-0001-2018.
- Han YW. Microbial levan. Adv Appl Microbiol. 1990;35:171–194. doi:10.1016/s0065-2164(08)70244-2.
- Adamberg S, Tomson K, Vija H, Puurand M, Kabanova N, Visnapuu T. Degradation of Fructans and Production of Propionic Acid by Bacteroides thetaiotaomicron are Enhanced by the Shortage of Amino Acids. Front Nutr. 2014;1:21. doi:10.3389/fnut.2014.00021.
- Poeker SA, Geirnaert A, Berchtold L, Greppi A, Krych L, Steinert RE. Understanding the prebiotic potential of different dietary fibers using an in vitro continuous adult fermentation model (PolyFermS). Sci Rep. 2018;8(1):4318. doi:10.1038/s41598-018-22438-y.
- Rakoff-Nahoum S, Foster KR, Comstock LE. The evolution of cooperation within the gut microbiota. Nature. 2016;533(7602):255–259. doi:10.1038/nature17626.
- Cuskin F, Lowe EC, Temple MJ, Zhu Y, Cameron E, Pudlo NA. Human gut Bacteroidetes can utilize yeast mannan through a selfish mechanism. Nature. 2015;517(7533):165–169. doi:10.1038/nature13995.
- Coyne MJ, Bechon N, Matano LM, McEneany VL, Chatzidaki-Livanis M, LE C. A family of anti-Bacteroidales peptide toxins widespread in the human gut microbiota. Nat Commun. 2019;10(1):3460. doi:10.1038/s41467-019-11494-1.
- Rosado CJ, Kondos S, Bull TE, Kuiper MJ, Law RH, Buckle AM. The MACPF/CDC family of pore-forming toxins. Cell Microbiol. 2008;10(9):1765–1774. doi:10.1111/j.1462-5822.2008.01191.x.
- Chatzidaki-Livanis M, Coyne MJ, Comstock LE. An antimicrobial protein of the gut symbiont Bacteroides fragilis with a MACPF domain of host immune proteins. Mol Microbiol. 2014;94:1361–1374. doi:10.1111/mmi.12839.
- Roelofs KG, Coyne MJ, Gentyala RR, Chatzidaki-Livanis M, Comstock LE. Bacteroidales Secreted Antimicrobial Proteins Target Surface Molecules Necessary for Gut Colonization and Mediate Competition In Vivo. mBio. 2016:7. doi:10.1128/mBio.01055-16.
- Shumaker AM, Laclare McEneany V, Coyne MJ, Silver PA, Comstock LE. Identification of a Fifth Antibacterial Toxin Produced by a Single Bacteroides fragilis Strain. J Bacteriol. 2019:201. doi:10.1128/JB.00577-18.
- McEneany VL, Coyne MJ, Chatzidaki-Livanis M, Comstock LE. Acquisition of MACPF domain-encoding genes is the main contributor to LPS glycan diversity in gut Bacteroides species. Isme J. 2018;12(12):2919–2928. doi:10.1038/s41396-018-0244-4.
- Chatzidaki-Livanis M, Coyne MJ, Roelofs KG, Gentyala RR, Caldwell JM, Comstock LE. Gut Symbiont Bacteroides fragilis Secretes a Eukaryotic-Like Ubiquitin Protein That Mediates Intraspecies Antagonism. mBio. 2017:8. doi:10.1128/mBio.01902-17.
- Coyne MJ, Comstock LE. Type VI Secretion Systems and the Gut Microbiota. Microbiol Spectr. 2019:7. doi:10.1128/microbiolspec.PSIB-0009-2018.
- Veesler D, Cambillau C. A common evolutionary origin for tailed-bacteriophage functional modules and bacterial machineries. Microbiol Mol Biol Rev. 2011;75(3):423–433. doi:10.1128/MMBR.00014-11.
- Coulthurst S. The Type VI secretion system: a versatile bacterial weapon. Microbiology. 2019;165(5):503–515. doi:10.1099/mic.0.000789.
- Coyne MJ, Roelofs KG, Comstock LE. Type VI secretion systems of human gut Bacteroidales segregate into three genetic architectures, two of which are contained on mobile genetic elements. BMC Genomics. 2016;17(1):58. doi:10.1186/s12864-016-2377-z.
- Coyne MJ, Zitomersky NL, McGuire AM, Earl AM, Comstock LE. Evidence of extensive DNA transfer between bacteroidales species within the human gut. mBio. 2014;5(3):e01305–14. doi:10.1128/mBio.01305-14.
- Verster AJ, Ross BD, Radey MC, Bao Y, Goodman AL, Mougous JD. The Landscape of Type VI Secretion across Human Gut Microbiomes Reveals Its Role in Community Composition. Cell Host Microbe. 2017;22(3):e4. doi:10.1016/j.chom.2017.08.010.
- Wexler AG, Bao Y, Whitney JC, Bobay LM, Xavier JB, Schofield WB. Human symbionts inject and neutralize antibacterial toxins to persist in the gut. Proc Natl Acad Sci U S A. 2016;113(13):3639–3644. doi:10.1073/pnas.1525637113.
- Alteri CJ, Mobley HLT. The Versatile Type VI Secretion System. Microbiol Spectr. 2016:4. doi:10.1128/microbiolspec.VMBF-0026-2015.
- Tang JY, Bullen NP, Ahmad S, Whitney JC. Diverse NADase effector families mediate interbacterial antagonism via the type VI secretion system. J Biol Chem. 2018;293(5):1504. doi:10.1074/jbc.RA117.000178.
- Sibinelli-Sousa S, Hespanhol JT, Nicastro GG, Matsuyama BY, Mesnage S, Patel A. A Family of T6SS Antibacterial Effectors Related to l,d-Transpeptidases Targets the Peptidoglycan. Cell Rep. 2020;31(12):107813. doi:10.1016/j.celrep.2020.107813.
- Houghteling PD, Walker WA. Why is initial bacterial colonization of the intestine important to infants’ and children’s health? J Pediatr Gastroenterol Nutr. 2015;60(3):294–307. doi:10.1097/MPG.0000000000000597.
- Milani C, Duranti S, Bottacini F, Casey E, Turroni F, Mahony J. The First Microbial Colonizers of the Human Gut: composition, Activities, and Health Implications of the Infant Gut Microbiota. Microbiol Mol Biol Rev. 2017:81. doi:10.1128/MMBR.00036-17.
- Fouhy F, Ross RP, Fitzgerald GF, Stanton C, Cotter PD. Composition of the early intestinal microbiota: knowledge, knowledge gaps and the use of high-throughput sequencing to address these gaps. Gut Microbes. 2012;3(3):203–220. doi:10.4161/gmic.20169.
- Wang M, Li M, Wu S, Lebrilla CB, Chapkin RS, Ivanov I. Fecal microbiota composition of breast-fed infants is correlated with human milk oligosaccharides consumed. J Pediatr Gastroenterol Nutr. 2015;60(6):825–833. doi:10.1097/MPG.0000000000000752.
- Backhed F, Roswall J, Peng Y, Feng Q, Jia H, Kovatcheva-Datchary P. Dynamics and Stabilization of the Human Gut Microbiome during the First Year of Life. Cell Host Microbe. 2015;17(5):690–703. doi:10.1016/j.chom.2015.04.004.
- Rodriguez JM, Murphy K, Stanton C, Ross RP, Kober OI, Juge N. The composition of the gut microbiota throughout life, with an emphasis on early life. Microb Ecol Health Dis. 2015;26:26050. doi:10.3402/mehd.v26.26050.
- Laursen MF, Bahl MI, Michaelsen KF, Licht TR. First Foods and Gut Microbes. Front Microbiol. 2017;8:356. doi:10.3389/fmicb.2017.00356.
- Rinninella E, Cintoni M, Raoul P, Lopetuso LR, Scaldaferri F, Pulcini G. Food Components and Dietary Habits: keys for a Healthy Gut Microbiota Composition. Nutrients. 2019:11. doi:10.3390/nu11102393.
- Hollister EB, Riehle K, Luna RA, Weidler EM, Rubio-Gonzales M, Mistretta TA. Structure and function of the healthy pre-adolescent pediatric gut microbiome. Microbiome. 2015;3(1):36. doi:10.1186/s40168-015-0101-x.
- Ringel-Kulka T, Cheng J, Ringel Y, Salojarvi J, Carroll I, Palva A. Intestinal microbiota in healthy U.S. young children and adults–a high throughput microarray analysis. PLoS One. 2013;8(5):e64315. doi:10.1371/journal.pone.0064315.
- Zhong H, Penders J, Shi Z, Ren H, Cai K, Fang C. Impact of early events and lifestyle on the gut microbiota and metabolic phenotypes in young school-age children. Microbiome. 2019;7(1):2. doi:10.1186/s40168-018-0608-z.
- Kurilshikov A, Wijmenga C, Fu J, Zhernakova A. Host Genetics and Gut Microbiome: challenges and Perspectives. Trends Immunol. 2017;38(9):633–647. doi:10.1016/j.it.2017.06.003.
- Ferrocino I, Di Cagno R, De Angelis M, Turroni S, Vannini L, Bancalari E. Fecal Microbiota in Healthy Subjects Following Omnivore, Vegetarian and Vegan Diets: culturable Populations and rRNA DGGE Profiling. PLoS One. 2015;10(6):e0128669. doi:10.1371/journal.pone.0128669.
- Hayashi H, Sakamoto M, Benno Y. Fecal microbial diversity in a strict vegetarian as determined by molecular analysis and cultivation. Microbiol Immunol. 2002;46(12):819–831. doi:10.1111/j.1348-0421.2002.tb02769.x.
- Singh RK, Chang HW, Yan D, Lee KM, Ucmak D, Wong K. Influence of diet on the gut microbiome and implications for human health. J Transl Med. 2017;15:73. doi:10.1186/s12967-017-1175-y.
- Tomova A, Bukovsky I, Rembert E, Yonas W, Alwarith J, Barnard ND. The Effects of Vegetarian and Vegan Diets on Gut Microbiota. Front Nutr. 2019;6:47. doi:10.3389/fnut.2019.00047.
- Voreades N, Kozil A, Weir TL. Diet and the development of the human intestinal microbiome. Front Microbiol. 2014;5:494. doi:10.3389/fmicb.2014.00494.
- Conlon MA, Bird AR. The impact of diet and lifestyle on gut microbiota and human health. Nutrients. 2014;7(1):17–44. doi:10.3390/nu7010017.
- Nakayama J, Heping Z, Lee YK. Asian gut Microbiome. Science Bulletin. 2017;62(12):816–817. doi:10.1016/j.scib.2017.04.001.
- Pareek S, Kurakawa T, Das B, Motooka D, Nakaya S, Rongsen-Chandola T. Comparison of Japanese and Indian intestinal microbiota shows diet-dependent interaction between bacteria and fungi. NPJ Biofilms Microbiomes. 2019;5:37. doi:10.1038/s41522-019-0110-9.
- Ishikawa E, Matsuki T, Kubota H, Makino H, Sakai T, Oishi K. Ethnic diversity of gut microbiota: species characterization of Bacteroides fragilis group and genus Bifidobacterium in healthy Belgian adults, and comparison with data from Japanese subjects. J Biosci Bioeng. 2013;116(2):265–270. doi:10.1016/j.jbiosc.2013.02.010.
- Lee YK, Conway P, Pettersson S, Nair GB, Surono I, Egayanti Y. ILSI Southeast Asia Region conference proceedings: the gut, its microbes and health: relevance for Asia. Asia Pac J Clin Nutr. 2017;26:957–971. doi:10.6133/apjcn.112016.09.
- Murphy EC, Morgelin M, Cooney JC, Frick IM. Interaction of Bacteroides fragilis and Bacteroides thetaiotaomicron with the kallikrein-kinin system. Microbiology. 2011;157(7):2094–2105. doi:10.1099/mic.0.046862-0.
- Polk BF. Bacteroides fragilis subspecies in clinical isolates. Ann Intern Med. 1977;86(5):569–571. doi:10.7326/0003-4819-86-5-569.
- Thadepalli H, Chuah SK, Qazi S, Thadepalli F, Gollapudi SV. Bacteroides fragilis-Induced Intra-Abdominal Abscess in an Experimental Model Treated with Telithromycin (HMR 3647). Chemotherapy. 2001;47(1):43–49. doi:10.1159/000048500.
- Vaishnavi C. Translocation of gut flora and its role in sepsis. Indian J Med Microbiol. 2013;31(4):334–342. doi:10.4103/0255-0857.118870.
- Archambaud C, Derre-Bobillot A, Lapaque N, Rigottier-Gois L, Serror P. Intestinal translocation of enterococci requires a threshold level of enterococcal overgrowth in the lumen. Sci Rep. 2019;9(1):8926. doi:10.1038/s41598-019-45441-3.
- Desai MS, Seekatz AM, Koropatkin NM, Kamada N, Hickey CA, Wolter M. A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell. 2016;167(1339–53):e21. doi:10.1016/j.cell.2016.10.043.
- Lobo LA, Jenkins AL, Jeffrey Smith C, Rocha ER. Expression of Bacteroides fragilis hemolysins in vivo and role of HlyBA in an intra-abdominal infection model. Microbiologyopen. 2013;2:326–337. doi:10.1002/mbo3.76.
- Pickard JM, Zeng MY, Caruso R, Nunez G. Gut microbiota: role in pathogen colonization, immune responses, and inflammatory disease. Immunol Rev. 2017;279(1):70–89. doi:10.1111/imr.12567.
- Sudhaharan S, Chavali P, Vemu L. Anaerobic brain abscess. Iran J Microbiol. 2016;8:120–124.
- Surana NK, Kasper DL. The yin yang of bacterial polysaccharides: lessons learned from B. fragilis PSA. Immunol Rev. 2012;245:13–26. doi:10.1111/j.1600-065X.2011.01075.x.
- Rodloff AC, Becker J, Blanchard DK, Klein TW, Hahn H, Friedman H. Inhibition of macrophage phagocytosis by Bacteroides fragilis in vivo and in vitro. Infect Immun. 1986;52(2):488–492. doi:10.1128/IAI.52.2.488-492.1986.
- Cress BF, Englaender JA, He W, Kasper D, Linhardt RJ, Koffas MA. Masquerading microbial pathogens: capsular polysaccharides mimic host-tissue molecules. FEMS Microbiol Rev. 2014;38(4):660–697. doi:10.1111/1574-6976.12056.
- Berezow AB, Ernst RK, Coats SR, Braham PH, Karimi-Naser LM, Darveau RP. The structurally similar, penta-acylated lipopolysaccharides of Porphyromonas gingivalis and Bacteroides elicit strikingly different innate immune responses. Microb Pathog. 2009;47(2):68–77. doi:10.1016/j.micpath.2009.04.015.
- Jacobson AN, Choudhury BP, Fischbach MA. The Biosynthesis of Lipooligosaccharide from Bacteroides thetaiotaomicron. mBio. 2018:9. doi:10.1128/mBio.02289-17.
- Cullen TW, Schofield WB, Barry NA, Putnam EE, Rundell EA, Trent MS. Gut microbiota. Antimicrobial peptide resistance mediates resilience of prominent gut commensals during inflammation. Science. 2015;347(6218):170–175. doi:10.1126/science.1260580.
- Zhang W, Zhu B, Xu J, Liu Y, Qiu E, Li Z. Bacteroides fragilis Protects Against Antibiotic-Associated Diarrhea in Rats by Modulating Intestinal Defenses. Front Immunol. 2018;9:1040. doi:10.3389/fimmu.2018.01040.
- Zamani S, Hesam Shariati S, Zali MR, Asadzadeh Aghdaei H, Sarabi Asiabar A, Bokaie S. Detection of enterotoxigenic Bacteroides fragilis in patients with ulcerative colitis. Gut Pathog. 2017;9(1):53. doi:10.1186/s13099-017-0202-0.
- Valguarnera E, Wardenburg JB. Good Gone Bad: one Toxin Away From Disease for Bacteroides fragilis. J Mol Biol. 2020;432(4):765–785. doi:10.1016/j.jmb.2019.12.003.
- Vu Nguyen T, Le Van P, Le Huy C, Weintraub A. Diarrhea caused by enterotoxigenic Bacteroides fragilis in children less than 5 years of age in Hanoi, Vietnam. Anaerobe. 2005;11(1–2):109–114. doi:10.1016/j.anaerobe.2004.10.004.
- Ulger Toprak N, Rajendram D, Yagci A, Gharbia S, Shah HN, Gulluoglu BM. The distribution of the bft alleles among enterotoxigenic Bacteroides fragilis strains from stool specimens and extraintestinal sites. Anaerobe. 2006;12(2):71–74. doi:10.1016/j.anaerobe.2005.11.001.
- Weikel CS, Grieco FD, Reuben J, Myers LL, Sack RB. Human colonic epithelial cells, HT29/C1, treated with crude Bacteroides fragilis enterotoxin dramatically alter their morphology. Infect Immun. 1992;60(2):321–327. doi:10.1128/iai.60.2.321-327.1992.
- Potempa J, Pike RN. Corruption of innate immunity by bacterial proteases. J Innate Immun. 2009;1(2):70–87. doi:10.1159/000181144.
- Thornton RF, Kagawa TF, O’Toole PW, Cooney JC. The dissemination of C10 cysteine protease genes in Bacteroides fragilis by mobile genetic elements. BMC Microbiol. 2010;10(1):122. doi:10.1186/1471-2180-10-122.
- Braun V, Focareta T. Pore-forming bacterial protein hemolysins (cytolysins). Crit Rev Microbiol. 1991;18(2):115–158. doi:10.3109/10408419109113511.
- Los FC, Randis TM, Aroian RV, Ratner AJ. Role of pore-forming toxins in bacterial infectious diseases. Microbiol Mol Biol Rev. 2013;77:173–207. doi:10.1128/MMBR.00052-12.
- Los FC, Ratner AJ. Pore-forming toxins in bacterial infections:</L> targets for novel drugs. Ned Tijdschr Geneeskd. 2014;158:A6668.
- Menestrina G, Dalla Serra M, Comai M, Coraiola M, Viero G, Werner S. Ion channels and bacterial infection: the case of β-barrel pore-forming protein toxins of Staphylococcus aureus. FEBS Lett. 2003;552(1):54–60. doi:10.1016/s0014-5793(03)00850-0.
- Robertson KP, Smith CJ, Gough AM, Rocha ER. Characterization of Bacteroides fragilis hemolysins and regulation and synergistic interactions of HlyA and HlyB. Infect Immun. 2006;74(4):2304–2316. doi:10.1128/IAI.74.4.2304-2316.2006.
- Zafar H, Saier MH Jr. Comparative genomics of transport proteins in seven Bacteroides species. PLoS One. 2018;13(12):e0208151. doi:10.1371/journal.pone.0208151.
- Lauber F, Deme JC, Lea SM, Berks BC. Type 9 secretion system structures reveal a new protein transport mechanism. Nature. 2018;564(7734):77–82. doi:10.1038/s41586-018-0693-y.
- Taguchi Y, Sato K, Yukitake H, Inoue T, Nakayama M, Naito M. Involvement of an Skp-Like Protein, PGN_0300, in the Type IX Secretion System of Porphyromonas gingivalis. Infect Immun. 2016;84(1):230–240. doi:10.1128/IAI.01308-15.
- Lasica AM, Ksiazek M, Madej M, Potempa J. The Type IX Secretion System (T9SS): highlights and Recent Insights into Its Structure and Function. Front Cell Infect Microbiol. 2017;7:215. doi:10.3389/fcimb.2017.00215.
- Kita D, Shibata S, Kikuchi Y, Kokubu E, Nakayama K, Saito A. Involvement of the Type IX Secretion System in Capnocytophaga ochracea Gliding Motility and Biofilm Formation. Appl Environ Microbiol. 2016;82(6):1756–1766. doi:10.1128/AEM.03452-15.
- Koneru L, Ksiazek M, Waligorska I, Straczek A, Lukasik M, Madej M. Mirolysin, a LysargiNase from Tannerella forsythia, proteolytically inactivates the human cathelicidin, LL-37. Biol Chem. 2017;398(3):395–409. doi:10.1515/hsz-2016-0267.
- Teixeira FL, Pauer H, Costa SB, Smith CJ, Domingues R, Rocha ER. Deletion of BmoR affects the expression of genes related to thiol/disulfide balance in Bacteroides fragilis. Sci Rep. 2018;8(1):14405. doi:10.1038/s41598-018-32880-7.
- Grant SS, Hung DT. Persistent bacterial infections, antibiotic tolerance, and the oxidative stress response. Virulence. 2013;4(4):273–283. doi:10.4161/viru.23987.
- Fraser T, Brown PD. Temperature and Oxidative Stress as Triggers for Virulence Gene Expression in Pathogenic Leptospira spp. Front Microbiol. 2017;8:783. doi:10.3389/fmicb.2017.00783.
- Sund CJ, Rocha ER, Tzianabos AO, Wells WG, Gee JM, Reott MA. The Bacteroides fragilis transcriptome response to oxygen and H2O2: the role of OxyR and its effect on survival and virulence. Mol Microbiol. 2008;67(1):129–142. doi:10.1111/j.1365-2958.2007.06031.x.
- Reott MA, Parker AC, Rocha ER, Smith CJ. Thioredoxins in redox maintenance and survival during oxidative stress of Bacteroides fragilis. J Bacteriol. 2009;191(10):3384–3391. doi:10.1128/JB.01665-08.
- Comtois SL, Gidley MD, Kelly DJ. Role of the thioredoxin system and the thiol peroxidases Tpx and Bcp in mediating resistance to oxidative and nitrosative stress in Helicobacter pylori. Microbiology. 2003;149(1):121–129. doi:10.1099/mic.0.25896-0.
- Nicholson SA, Smalley D, Smith CJ, Abratt VR. The recA operon: A novel stress response gene cluster in Bacteroides fragilis. Res Microbiol. 2014;165(4):290–299. doi:10.1016/j.resmic.2014.03.005.
- Zitvogel L, Galluzzi L, Viaud S, Vetizou M, Daillere R, Merad M. Cancer and the gut microbiota: an unexpected link. Sci Transl Med. 2015;7(271):271ps1. doi:10.1126/scitranslmed.3010473.
- Gopalakrishnan V, Helmink BA, Spencer CN, Reuben A, Wargo JA. The Influence of the Gut Microbiome on Cancer, Immunity, and Cancer Immunotherapy. Cancer Cell. 2018;33(4):570–580. doi:10.1016/j.ccell.2018.03.015.
- Baffy G. Gut Microbiota and Cancer of the Host: colliding Interests. Adv Exp Med Biol. 2020;1219:93–107. doi:10.1007/978-3-030-34025-4_5.
- Vivarelli S, Salemi R, Candido S, Falzone L, Santagati M, Stefani S. Gut Microbiota and Cancer: from Pathogenesis to Therapy. Cancers (Basel). 2019:11. doi:10.3390/cancers11010038.
- Cervelli M, Amendola R, Polticelli F, Mariottini P. Spermine oxidase: ten years after. Amino Acids. 2012;42(2–3):441–450. doi:10.1007/s00726-011-1014-z.
- Murray Stewart T, Dunston TT, Woster PM, Casero RA Jr. Polyamine catabolism and oxidative damage. J Biol Chem. 2018;293(48):18736–18745. doi:10.1074/jbc.TM118.003337.
- Liou GY, Storz P. Reactive oxygen species in cancer. Free Radic Res. 2010;44(5):479–496. doi:10.3109/10715761003667554.
- Snezhkina AV, Krasnov GS, Lipatova AV, Sadritdinova AF, Kardymon OL, Fedorova MS. The Dysregulation of Polyamine Metabolism in Colorectal Cancer Is Associated with Overexpression of c-Myc and C/EBPbeta rather than Enterotoxigenic Bacteroides fragilis Infection. Oxid Med Cell Longev. 2016;2016:2353560. doi:10.1155/2016/2353560.
- Zamani S, Taslimi R, Sarabi A, Jasemi S, Sechi LA, Feizabadi MM. Enterotoxigenic Bacteroides fragilis: A Possible Etiological Candidate for Bacterially-Induced Colorectal Precancerous and Cancerous Lesions. Front Cell Infect Microbiol. 2019;9:449. doi:10.3389/fcimb.2019.00449.
- Moncrief JS, Obiso R Jr., Barroso LA, Kling JJ, Wright RL, Van Tassell RL. The enterotoxin of Bacteroides fragilis is a metalloprotease. Infect Immun. 1995;63(1):175–181. doi:10.1128/IAI.63.1.175-181.1995.
- Wu S, Rhee KJ, Zhang M, Franco A, Sears CL. Bacteroides fragilis toxin stimulates intestinal epithelial cell shedding and gamma-secretase-dependent E-cadherin cleavage. J Cell Sci. 2007;120:1944–1952. doi:10.1242/jcs.03455.
- Wu S, Morin PJ, Maouyo D, Sears CL. Bacteroides fragilis enterotoxin induces c-Myc expression and cellular proliferation. Gastroenterology. 2003;124(2):392–400. doi:10.1053/gast.2003.50047.
- Kim JM, Cho SJ, Oh YK, Jung HY, Kim YJ, Kim N. Nuclear factor-kappa B activation pathway in intestinal epithelial cells is a major regulator of chemokine gene expression and neutrophil migration induced by Bacteroides fragilis enterotoxin. Clin Exp Immunol. 2002;130(1):59–66. doi:10.1046/j.1365-2249.2002.01921.x.
- Kim JM, Jung HY, Lee JY, Youn J, Lee CH, Kim KH. Mitogen-activated protein kinase and activator protein-1 dependent signals are essential for Bacteroides fragilis enterotoxin- induced enteritis. Eur J Immunol. 2005;35(9):2648–2657. doi:10.1002/eji.200526321.
- Yuan A, Chen JJ, Yao PL, Yang PC. The role of interleukin-8 in cancer cells and microenvironment interaction. Front Biosci. 2005;10:853–865. doi:10.2741/1579.
- Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990;61(5):759–767. doi:10.1016/0092-8674(90)90186-i.
- Giardiello FM, Krush AJ, Petersen GM, Booker SV, Kerr M, Tong LL. Phenotypic variability of familial adenomatous polyposis in 11 unrelated families with identical APC gene mutation. Gastroenterology. 1994;106(6):1542–1547. doi:10.1016/0016-5085(94)90408-1.
- Dejea CM, Fathi P, Craig JM, Boleij A, Taddese R, Geis AL. Patients with familial adenomatous polyposis harbor colonic biofilms containing tumorigenic bacteria. Science. 2018;359(6375):592–597. doi:10.1126/science.aah3648.
- Toprak NU, Yagci A, Gulluoglu BM, Akin ML, Demirkalem P, Celenk T. A possible role of Bacteroides fragilis enterotoxin in the aetiology of colorectal cancer. Clin Microbiol Infect. 2006;12(8):782–786. doi:10.1111/j.1469-0691.2006.01494.x.
- Haghi F, Goli E, Mirzaei B, Zeighami H. The association between fecal enterotoxigenic B. fragilis with colorectal cancer. BMC Cancer. 2019;19(1):879. doi:10.1186/s12885-019-6115-1.
- Fernandez MF, Reina-Perez I, Astorga JM, Rodriguez-Carrillo A, Plaza-Diaz J, Fontana L. Breast Cancer and Its Relationship with the Microbiota. Int J Environ Res Public Health. 2018:15. doi:10.3390/ijerph15081747.
- Buchta Rosean C, Bostic RR, Ferey JCM, Feng TY, Azar FN, Tung KS. Preexisting Commensal Dysbiosis Is a Host-Intrinsic Regulator of Tissue Inflammation and Tumor Cell Dissemination in Hormone Receptor-Positive Breast Cancer. Cancer Res. 2019;79(14):3662. doi:10.1158/0008-5472.CAN-18-3464.
- Burstein HJ, Cirrincione CT, Barry WT, Chew HK, Tolaney SM, Lake DE. Endocrine therapy with or without inhibition of epidermal growth factor receptor and human epidermal growth factor receptor 2: a randomized, double-blind, placebo-controlled phase III trial of fulvestrant with or without lapatinib for postmenopausal women with hormone receptor- positive advanced breast cancer-CALGB 40302 (Alliance). J Clin Oncol. 2014;32:3959–3966. doi:10.1200/JCO.2014.56.7941.
- Miko E, Kovacs T, Sebo E, Toth J, Csonka T, Ujlaki G. Microbiome-Microbial Metabolome-Cancer Cell Interactions in Breast Cancer-Familiar, but Unexplored Cells. 2019:8. 10.3390/cells8040293.
- Kirkup BM, McKee A, Makin KA, Paveley J, Caim S, Alcon-Giner C. Perturbation of the gut microbiota by antibiotics results in accelerated breast tumour growth and metabolic dysregulation. bioRxiv. 2019:553602. doi:10.1101/553602.
- Campbell MJ, McCune E, Johnson B, O’Meara T, Heditsian D, Brain S. Abstract 2830: breast cancer and the human oral and gut microbiomes. Cancer Res. 2019;79:2830. doi:10.1158/1538-7445.AM2019-2830.
- Parida S, Wu S, Muniraj N, Siddharth S, Nagaligam A, Sears CL. Abstract PR02: bacteroides fragilis toxin induces epithelial-to-mesenchymal transition and stem-like phenotype in breast epithelial cells and concomitantly activates Notch1 and βcatenin axes. Cancer Immunology Research. 2020;8:PR02–PR. doi:10.1158/1538-7445.MVC2020-PR06.
- Evans JM, Morris LS, Marchesi JR. The gut microbiome: the role of a virtual organ in the endocrinology of the host. J Endocrinol. 2013;218(3):R37–47. doi:10.1530/JOE-13-0131.
- Wu HJ, Wu E. The role of gut microbiota in immune homeostasis and autoimmunity. Gut Microbes. 2012;3(1):4–14. doi:10.4161/gmic.19320.
- Xu H, Liu M, Cao J, Li X, Fan D, Xia Y. The Dynamic Interplay between the Gut Microbiota and Autoimmune Diseases. J Immunol Res. 2019;2019:7546047. doi:10.1155/2019/7546047.
- Hebbandi Nanjundappa R, Ronchi F, Wang J, Clemente-Casares X, Yamanouchi J, Sokke UC. A Gut Microbial Mimic that Hijacks Diabetogenic Autoreactivity to Suppress Colitis. Cell. 2017;171(3):e17. doi:10.1016/j.cell.2017.09.022.
- Gianchecchi E, Fierabracci A. Recent Advances on Microbiota Involvement in the Pathogenesis of Autoimmunity. Int J Mol Sci. 2019:20. doi:10.3390/ijms20020283.
- Patrick S, Blakely GW. Crossing the eukaryote-prokaryote divide: A ubiquitin homolog in the human commensal bacterium Bacteroides fragilis. Mob Genet Elements. 2012;2(3):149–151. doi:10.4161/mge.21191.
- Weil R. Does antigen masking by ubiquitin chains protect from the development of autoimmune diseases? Front Immunol. 2014;5:262. doi:10.3389/fimmu.2014.00262.
- Stewart L, DME J, Blakely G, Patrick S. Antigenic mimicry of ubiquitin by the gut bacterium Bacteroides fragilis: a potential link with autoimmune disease. Clin Exp Immunol. 2018;194(2):153–165. doi:10.1111/cei.13195.
- Bracamonte-Baran W, Cihakova D. Cardiac Autoimmunity: myocarditis. Adv Exp Med Biol. 2017;1003:187–221. doi:10.1007/978-3-319-57613-8_10.
- Krejci J, Mlejnek D, Sochorova D, Nemec P. Inflammatory Cardiomyopathy: A Current View on the Pathophysiology, Diagnosis, and Treatment. Biomed Res Int. 2016;2016:4087632. doi:10.1155/2016/4087632.
- Gil-Cruz C, Perez-Shibayama C, De Martin A, Ronchi F, van der Borght K, Niederer R. Microbiota-derived peptide mimics drive lethal inflammatory cardiomyopathy. Science. 2019;366(6467):881–886. doi:10.1126/science.aav3487.
- Chatzidaki-Livanis M, Weinacht KG, Comstock LE. Trans locus inhibitors limit concomitant polysaccharide synthesis in the human gut symbiont Bacteroides fragilis. Proc Natl Acad Sci U S A. 2010;107(26):11976–11980. doi:10.1073/pnas.1005039107.
- Troy EB, Kasper DL. Beneficial effects of Bacteroides fragilis polysaccharides on the immune system. Front Biosci (Landmark Ed). 2010;15(1):25–34. doi:10.2741/3603.
- Sommese L, Pagliuca C, Avallone B, Ippolito R, Casamassimi A, Costa V. Evidence of Bacteroides fragilis protection from Bartonella henselae-induced damage. PLoS One. 2012;7:e49653. doi:10.1371/journal.pone.0049653.
- Ramakrishna C, Kujawski M, Chu H, Li L, Mazmanian SK, Cantin EM. Bacteroides fragilis polysaccharide A induces IL-10 secreting B and T cells that prevent viral encephalitis. Nat Commun. 2019;10(1):2153. doi:10.1038/s41467-019-09884-6.
- Shen Y, Giardino Torchia ML, Lawson GW, Karp CL, Ashwell JD, Mazmanian SK. Outer membrane vesicles of a human commensal mediate immune regulation and disease protection. Cell Host Microbe. 2012;12(4):509–520. doi:10.1016/j.chom.2012.08.004.
- Mogensen TH. Pathogen recognition and inflammatory signaling in innate immune defenses. Clin Microbiol Rev Table of Contents. 2009;22:240–273. doi:10.1128/CMR.00046-08.
- Leifer CA, Medvedev AE. Molecular mechanisms of regulation of Toll-like receptor signaling. J Leukoc Biol. 2016;100(5):927–941. doi:10.1189/jlb.2MR0316-117RR.
- Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat Immunol. 2010;11(5):373–384. doi:10.1038/ni.1863.
- Round JL, Lee SM, Li J, Tran G, Jabri B, Chatila TA. The Toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science. 2011;332(6032):974–977. doi:10.1126/science.1206095.
- Troy EB, Kasper DL. Beneficial effects of Bacteroides fragilis polysaccharides on the immune system. Front Biosci (Landmark Ed). 2010;15(1):25–34. doi:10.2741/3603.
- Cohen-Poradosu R, McLoughlin RM, Lee JC, Kasper DL. Bacteroides fragilis-stimulated interleukin-10 contains expanding disease. J Infect Dis. 2011;204(3):363–371. doi:10.1093/infdis/jir277.
- Kennedy EA, King KY, Baldridge MT. Mouse Microbiota Models: comparing Germ-Free Mice and Antibiotics Treatment as Tools for Modifying Gut Bacteria. Front Physiol. 2018;9:1534. doi:10.3389/fphys.2018.01534.
- Pfeiffer JK, Sonnenburg JL. The intestinal microbiota and viral susceptibility. Front Microbiol. 2011;2:92. doi:10.3389/fmicb.2011.00092.
- McCormick AL, Mocarski JES. . Viral modulation of the host response to infection. In: Arvin A, Campadelli-Fiume G, Mocarski E, Moore PS, Roizman B, Whitley R, editors. Human Herpesviruses: biology, Therapy, and Immunoprophylaxis. Cambridge; 2007.
- Mancini M, Vidal SM. Insights into the pathogenesis of herpes simplex encephalitis from mouse models. Mamm Genome. 2018;29(7–8):425–445. doi:10.1007/s00335-018-9772-5.
- Shimizu J, Kubota T, Takada E, Takai K, Fujiwara N, Arimitsu N. Propionate- producing bacteria in the intestine may associate with skewed responses of IL10-producing regulatory T cells in patients with relapsing polychondritis. PLoS One. 2018;13(9):e0203657. doi:10.1371/journal.pone.0203657.
- Vinolo MA, Rodrigues HG, Nachbar RT, Curi R. Regulation of inflammation by short chain fatty acids. Nutrients. 2011;3(10):858–876. doi:10.3390/nu3100858.
- Cruz-Bravo RK, Guevara-Gonzalez RG, Ramos-Gomez M, Oomah BD, Wiersma P, Campos-Vega R. The fermented non-digestible fraction of common bean (Phaseolus vulgaris L.) triggers cell cycle arrest and apoptosis in human colon adenocarcinoma cells. Genes Nutr. 2014;9:359. doi:10.1007/s12263-013-0359-1.
- Nagpal R, Wang S, Ahmadi S, Hayes J, Gagliano J, Subashchandrabose S. Human- origin probiotic cocktail increases short-chain fatty acid production via modulation of mice and human gut microbiome. Sci Rep. 2018;8(1):12649. doi:10.1038/s41598-018-30114-4.
- Walther B, Karl JP, Booth SL, Boyaval P. Menaquinones, bacteria, and the food supply: the relevance of dairy and fermented food products to vitamin K requirements. Adv Nutr. 2013;4:463–473. doi:10.3945/an.113.003855.
- Fujita Y, Iki M, Tamaki J, Kouda K, Yura A, Kadowaki E. Association between vitamin K intake from fermented soybeans, natto, and bone mineral density in elderly Japanese men: the Fujiwara-kyo Osteoporosis Risk in Men (FORMEN) study. Osteoporos Int. 2012;23(2):705–714. doi:10.1007/s00198-011-1594-1.
- Johnson CH, Dejea CM, Edler D, Hoang LT, Santidrian AF, Felding BH. Metabolism links bacterial biofilms and colon carcinogenesis. Cell Metab. 2015;21(6):891–897. doi:10.1016/j.cmet.2015.04.011.
- Ulsemer P, Toutounian K, Kressel G, Goletz C, Schmidt J, Karsten U. Impact of oral consumption of heat-treated Bacteroides xylanisolvens DSM 23964 on the level of natural TFalpha-specific antibodies in human adults. Benef Microbes. 2016;7:485–500. doi:10.3920/BM2015.0143.
- Ulsemer P, Henderson G, Toutounian K, Loffler A, Schmidt J, Karsten U. Specific humoral immune response to the Thomsen-Friedenreich tumor antigen (CD176) in mice after vaccination with the commensal bacterium Bacteroides ovatus D-6. Cancer Immunol Immunother. 2013;62(5):875–887. doi:10.1007/s00262-013-1394-x.
