ABSTRACT
Mucormycosis is a rare fungal infection; however, the number of cases increased during the last decades. The main risk factors are immunosuppression and uncontrolled diabetes mellitus. Although Lichtheimia species represent a common cause of mucormycosis in Europe, virulence and pathogenesis of this genus has not been investigated in detail yet. Using murine pulmonary infection models, we found that immunosuppression is essential for establishment of infection. The disease was characterized by necrosis, angioinvasion, thrombosis, and the lethal course of infection was associated with systemic activation of platelets. Furthermore, dissemination to internal organs was frequently observed. While the virulence potential of individual L. corymbifera and L. ramosa isolates differed, pathogenicity of both species was comparable. Although ketoacidosis promoted Rhizopus infection in mice, it did not predispose mice to infection with Lichtheimia in the absence of additional immunosuppression. This might partially explain the dominance of Rhizopus as cause of mucormycosis in countries with high prevalence of ketoacidotic patients.
Introduction
Mucormycosis is a life threatening infection in animals and humans, caused by ubiquitously distributed saprophytic fungi of the order Mucorales. Mucormycoses are rare, but the number of cases has increased during the last decades with mortality rates around 50%.Citation1 Risk factors for mucormycosis are immunosuppression and uncontrolled diabetes mellitus; cases in immunocompetent patients following trauma have been described.Citation1-3 Infections are usually initiated by either inhalation of spores, leading to sinusoidal or pulmonary mucormycosis, or are associated with traumatic or surgical wounds, facilitating cutaneous or deep-tissue infections.Citation1,3,4 Notably, infections can spread hematogenously and by direct invasion of adjacent tissue, leading to rhinocerebral, cerebral and other forms of mucormycosis.Citation1,3,4
70–80% of mucormycoses are caused by Rhizopus, Mucor, and Lichtheimia (former Absidia) species, with Lichtheimia as the second and third most common cause of mucormycosis in Europe and the USA, respectively.Citation1,3-5 The genus Lichtheimia contains at least 6 species,Citation6 of which only L. corymbifera, L. ramosa and L. ornata were isolated from clinical material,Citation7 suggesting differences in the virulence potential of the species. This hypothesis is supported by results from a chicken embryo model,Citation8,9 which furthermore revealed differences in the virulence potential of L. corymbifera and L. ramosa isolates. Whether these results are transferable to mammalian hosts has however not been determined yet.
Murine models have been successfully used to study pathogenesisCitation10-12 and to evaluate therapy efficacyCitation13-17 of mucormycosis caused by Rhizopus and Mucor. Depending on the localization of mucormycosis to be studied, the route of infection differs; systemic infection via the tail vein to mimic hematogenous dissemination,Citation13,14,18 and pulmonary infections facilitated by intranasal or intratracheal application of spores are most commonly used.Citation15-17 Chemically-induced diabetic ketoacidosis, leukopenia based on cyclophosphamide treatment and immunosuppression by corticosteroids are used to mimic common risk factors in humans.Citation12-18 As mucormycosis caused by Lichtheimia species has not yet been studied in detail in mammalian models, we established a murine model of pulmonary Lichtheimia infection based on published models for Rhizopus and Mucor. Corticosteroid treatment was confirmed as a risk factor for pulmonary infections by L. corymbifera and L. ramosa, and infection resembled human disease with necrosis, angioinvasion and thrombosis. However, ketoacidosis did not render mice susceptible to pulmonary Lichtheimia infection.
Material and methods
Ethics statement
Animal studies were conducted in accordance with the recommendations of the European Community and were approved by the Animal Care and Usage Committee of the Landesamt Thüringen, Bad Langensalza, Germany (permission no. 03–006/11 and 03–001/14).
Induction of immunosuppression and diabetic ketoacidosis
Outbred female CD-1 mice (Charles River Laboratories Germany) 6 to 8 weeks old were kept under specific pathogen-free conditions in groups of 5 in isolated ventilated cages with sterile food and water provided ad libitum. Immunosuppression was established by intraperitoneal injection of 25 mg cortisone acetate (Sigma Aldrich) in 200 µL PBS on day -3 and day 0.Citation19 To induce insulin-dependent ketoacidosis a single dose of 190 mg/kg streptozotocin (Sigma Aldrich) in 200 µl citrate buffer (0.1 M, pH 4.5, sterile-filtrated) was injected intraperitoneal after 4 h of fasting on day -10. After streptozotocin injection all animals received drinking water supplemented with 10% saccharose (Carl Roth GmbH, Germany) for 24 h. Development of hyperglycemia was evaluated by measuring the blood glucose concentration 4 d before and 7 d after streptozotocin treatment in blood from the lateral tail vain using the Ascensia MICROFILL system (Henry Schein) according to the manufacturer's instructions. Animals that did not develop hyperglycemia (blood glucose levels below 200 mg/dL) were excluded from the experiment before infection. Persistence of hyperglycemia was likewise determined at the end of the experiment (mean blood glucose 421 mg/dL). Untreated animals and mice treated with cortisone acetate analyzed on day 0 served as controls.
Cultivation of Lichtheimia and intranasal infection
The strains used in this study are listed in and are deposited in the Jena Microbial Resource Collection (www.jmrc.hki-jena.de). Spore suspensions were prepared as described previouslyCitation8,9; in brief, strains were revived from soil cultures (double-sterilized sand-loam-garden soil) and precultivated at 37°C in 10 mL modified liquid SUP medium (55 mM glucose, 30 mM potassium dihydrogen phosphate, 20 mM ammonium chloride, 5 mM di-potassium hydrogen phosphate, 1 mM magnesium sulfate and 0.5% yeast extract; all reagents from Carl Roth GmbH). The mycelium was further cultivated on modified SUP plates at 37°C for 7 d to allow sporulation. Spores were harvested, washed twice with sterile PBS (Dulbeccos, Thermo Fisher) and diluted in sterile PBS to the indicated concentrations. Intranasal infection was performed with 20 µL of the spore suspension as described previouslyCitation19 under general anesthesia (0.05 mg/kg fentanyl, 0.5 mg/kg medetomidin, and 5 mg/kg midazolam). Anesthesia was terminated by subcutaneous application of antidote (1.2 mg/kg naloxone, 2.5 mg/kg atipamezol, and 0.5 mg/kg flumazenil). Mice were monitored at least twice daily for development of clinical symptoms and humanely killed if they met predefined humane endpoints (≥ 25% weight loss, severe lethargy, severe dyspnea, inability to move coordinately) or at the end of the experiment. To assess the pathological alterations caused by the infection, all animals were necropsied and lung, liver, heart, kidneys and brain were analyzed by histology.
Table 1. Strains used in this study
Histology
Lung, kidneys, a lobe of the liver, heart and brain were aseptically collected during necropsy and fixed in neutral-buffered formalin. Paraffin-embedded sections were stained with either periodic acid Schiff (PAS) or hematoxylin and eosin (H&E) and microscopically analyzed using the Axiocam from Zeiss. A scoring system was used to quantify tissue alterations (fibrosis, necrotic tissue, immune cell infiltration, and presence of fungal elements, respectively) as follows: 0 = no alterations; 1 = single fields of small areas (< 50 µm) of alterations; 2 = multiple fields and/or larger areas (100 – 200 µm); 3 = severe tissue alterations, with the majority of the organ structure impaired. For quantification, each type of alteration (fibrosis, necrotic tissue, immune cell infiltration, and presence of fungal elements, respectively) was first scored individually. Then, single scores for the different types of alterations were added. The use of this additive scoring system was considered to be more precise as e.g. in lungs of mice who survived the infection, no fungal cells can be found but areas of necrosis and immune cell infiltration are still detectable.
Collection of blood and determination of platelet activation
100 µl of blood were collected under terminal anesthesia by retro-orbital bleeding, immediately transferred into a tube containing 10 µl EDTA solution (1.6 mg/ml), and gently mixed. 20 µl of the sample were analyzed on a BC-5300Vet (Mindray) configured for murine blood, providing 23 parameters, including platelet counts and mean platelet volume. Platelet-rich plasma (RPP) was prepared from whole-blood by centrifugation at 135 g for 15 min at room temperature. To detect platelet activation, platelets were stained for 30 min with fluorescence-labeled antibodies (BioLegend) directed against CD41 as platelet marker and CD63 as activation marker, followed by fixation with 1% formaldehyde. Half of the sample was used directly for flow cytometry (FC)-based quantification of CD41+ microparticles, which were gated according to their size. The rest of the sample was analyzed by FC for CD63 expression on the surface of CD41-positive platelets. Fibrinogen binding, which indicates activation of surface-bound GPIIb-IIIa complex, was quantified using fluorescence-labeled antibodies (BioLegend) as described for CD63. Plasma concentrations of soluble CD62P were determined using the Quantikine® ELISA Mouse sP-Selectin/CD62P kit (R&D Systems, USA) performed according to manufacturer instructions. Plasma was prepared by centrifugation of whole-blood at 1500 g for 15 min at room temperature.
Statistical analysis
The Graph Pad Prism 6 software (GraphPad Software Inc.) was used for statistical analysis. Survival data were analyzed using the Log-rank Mantel-Cox test. Gaussian distribution of the histological scores and flow cytometry data was confirmed by the D'Agostino & Pearson omnibus normality test. Histological scores were analyzed by one-way ANOVA with Tukey's multiple comparison test. Platelet activation markers were analyzed by unpaired, 2-sided t-test. Statistical significance was considered for p-values < 0.05.
Results
Immunosuppression is essential for the development of pulmonary Lichtheimia infection in mice
As inhalation of spores is the most common route of infection with mucormycetes, we performed intranasal infection. To determine whether this leads to reproducible numbers of spores reaching the lung, mice were killed 10 minutes after infection and pulmonary tissue (after removal of trachea and major bronchi) was homogenized and quantitatively cultured on SUP plates. Similar amounts of spores were recovered from all mice infected with the same dose (), indicating reliable delivery of spores to the lung.
Figure 1. Susceptibility of corticosteroid-treated mice to pulmonary Lichtheimia infection and histopathological alterations in the lung. (A) Number of spores reaching the lung after intranasal application. Female CD-1 mice (n = 3 mice/group) were killed 10 min after intranasal application of L. corymbifera FSU 9682 spores. (B) Dose-dependent survival of corticosteroid-treated mice intranasaly infected with L. corymbifera FSU 9682 (n = 5 mice/group). Log rank (Mantel-Cox) test was used to compare each infection dose to PBS treated control mice (#p = 0.05, ##p = 0.01). (C) Representative lung sections from mice infected with 5 × 106 spores of the L. corymbifera reference strain. Slides were stained with hematoxylin/eosin (HE) or Periodic Acid Schiff (PAS) as indicated in the upper right corner of each image. Representative HE and PAS stained slides from 2 consecutive sections are shown in the left and middle column, the right column represents an enlarged area
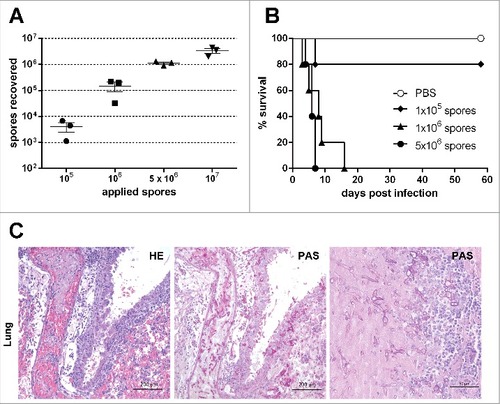
Figure 2. Corticosteroid-treated but not diabetic ketoacidotic mice succumb to infection with Lichtheimia corymbifera. (A) Survival of corticosteroid-treated and diabetic ketoacidotic mice intranasaly infected with 5 × 106 spores of L. corymbifera FSU 9682. Shown are the combined survival data from 2 independent experiments with n = 7–10 mice/group/experiment. Log-rank (Mantel-Cox) test was used to compare both groups, ##p < 0.01. (B) Representative PAS stained histological sections from lungs of PBS control mice (upper row) and infected mice 28 d p. inf. Black scale bar = 50 µm
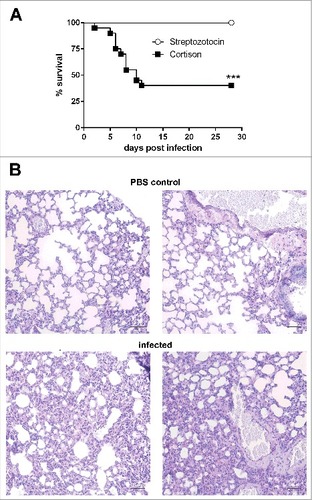
Immunosuppression is a risk factor for human mucormycosis,Citation1 and was shown to facilitate pulmonary infection by other mucormycetes in mice. To determine whether immunosuppression was necessary to establish infection, immunocompetent and mice immunocompromised with corticosteroids were infected in parallel with 105 to 5 × 106 spores (n = 5 per dose). While immunocompetent mice remained healthy, corticosteroid-treated mice infected with either 5 × 106 or 106 spores succumbed to infection (median survival time 6 and 8 days, respectively). 80% of the corticosteroid-treated mice receiving 105 spores survived the infection and were clinically healthy 58 d after infection (). The susceptibility of corticosteroid-treated mice to pulmonary L. corymbifera infection was confirmed in 2 additional independent experiments (5 × 106 spores/mouse, n = 20). Parallel experiments with immunocompetent mice (n = 10) did neither lead to clinical symptoms nor histological tissue alterations, indicating that immunosuppression is essential for pulmonary Lichtheimia infections in mice.
Murine pulmonary Lichtheimia infection is characterized by thrombosis, tissue necrosis and dissemination
In L. corymbifera-infected corticosteroid-treated mice the first clinical symptoms occurred 3 to 4 d post infection (p. inf.), characterized by ruffled fur and mild dyspnea. In these animals dyspnea commonly progressed to severe respiratory distress warranting euthanasia within 12 h to 72 h (11/35 mice). In animals with later onset of clinical disease, dyspnea was less prominent and lethargy and hypothermia led to euthanasia (7/35 mice). Individual animals presented with ataxia, torticollis or tremor and were killed due to their inability to ambulate (6/35 mice) between day 6 and 9 p. inf.. Mild clinical symptoms that occurred later than day 10 p. inf. (ataxia, lethargy or dyspnea) did not progress and were usually transient (11/35 mice).
Consistent with the clinical manifestation, the lungs of mice with overt dyspnea showed extensive dark red to black areas histologically characterized by hemorrhage, tissue destruction, fungal growth, angioinvasion and thrombosis (), reminiscent of typical mucormycosis in human patients.Citation20 Comparable pathology was also observed in the kidneys of some animals killed after day 5 p. inf. (16/35 mice, see Fig. S1). Occasionally, whitish areas were also observed in the liver (8/35), heart (4/35), or diaphragm (1/35), corresponding with tissue necrosis but not necessarily detection of fungal elements by histology (see Fig. S1). The neurologic symptoms observed in some mice suggested dissemination to the brain; this was histologically confirmed in some, but not all cases (see Fig. S1). Notably, the lung histopathology in animals that developed clinical disease later or at slower progression was more moderate than in acute cases and characterized by infiltration of lymphoid cells; it was however also frequently associated with dissemination into other organs.
Diabetic ketoacidosis alone does not promote pulmonary Lichtheimia infection
Uncontrolled diabetes mellitus leading to ketoacidosis is an important risk factor for mucormycoses in humans and has been shown to promote pulmonary Rhizopus infections in mice.Citation1,5,17,21,22 Insulin-dependent ketoacidosis occurs in genetically modified mice, in certain mouse strains fed with a special diet,Citation23 or can be achieved by treatment with streptozotocin, an antibiotic that causes pancreatic β-cell destruction and thereby insulin deficiency and hyperglycemia.Citation24 We chose the streptozotocin model as it has been used by others to investigate Rhizopus infections in mice.Citation17,25,26 Development of hyperglycemia was confirmed by determination of blood glucose, and animals infected with R. arrhizus (formerly R. oryzae; CBS 126971) as a positive control succumbed to infection (data not shown). To our surprise, in 2 independent experiments all diabetic ketoacidotic (DKA) mice survived L. corymbifera infection without developing clinical symptoms () and no fungal elements could be detected in the lungs and kidneys of DKA mice at the end of the experiments. However, infiltration of lymphoid cells in the lungs of DKA mice indicated a local immune response that likely mediated fungal clearance (). It should however be noted that DKA mice in this study were not additionally treated with corticosteroids, in contrast to previous studies by others.Citation17 Thus, we cannot exclude an exacerbating effect of DKA on the susceptibility of immunocompromised mice to Lichtheimia infections.
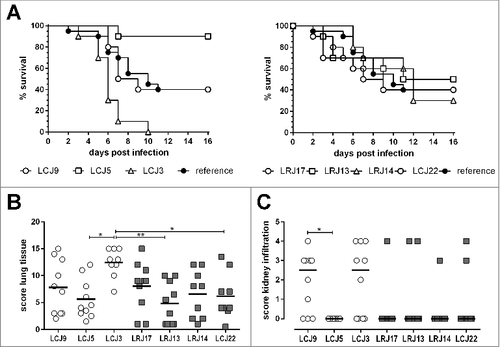
Figure 3. Strain specific virulence of different Lichtheimia isolates in a mouse model of pulmonary infection. (A) Survival of immunocompromised female CD-1 mice intranasaly infected with 5 × 106 spores of the indicated L. corymbifera and L. ramosa strains. Combined data from 2 independent experiments (n = 5 mice/group/experiment). ##p < 0.01, determined by Log-rank (Mantel-Cox) test. (B) Analysis of histological scores from lung and kidneys (C) (n = 8–10 mice/group). Statistical significance was calculated using one-way ANOVA with a Tukey's multiple comparison test, #p < 0.05 and ##p < 0.01
Virulence varies between different L. corymbifera isolates
Using a chicken embryo model, we previously observed comparable overall virulence potential of L. corymbifera and L. ramosa but also substantial variation in the virulence between distinct strains.Citation8 To determine whether this finding was transferable to infection of a mammalian host, we tested the virulence of 3 additional L. corymbifera and 4 L. ramosa strains in corticosteroid-treated and DKA mice.
None of the tested Lichtheimia strains was able to cause clinical symptoms in DKA mice (data not shown), confirming that, in contrast to Rhizopus, ketoacidosis does not significantly promote the development of pulmonary L. corymbifera infection in mice. However, infection of immunosuppressed mice revealed differences in the virulence potential of the L. corymbifera strains tested (median survival times ranging from 8 to 13.5 days; ). Whereas the reference strain and LCJ9 showed average virulence (median survival 10 and 8 days, respectively), LCJ5 was found to be strongly attenuated. Only LCJ3 was significantly (p < 0.01, Log-rank test) more virulent than the reference strain, with a median survival time of 6 d and higher absolute mortality. Additionally, quantification of the histological alterations using an additive scoring system for lung and kidneys supported the higher virulence of LCJ3 (, and see Fig. S2). In contrast, both survival and lesion scores were comparable for the L. ramosa strains tested (, ) and comparable to the L. corymbifera reference strain used for the establishment of the model (). In all cases of clinical infections, the pathological alterations resembled those described above for the L. corymbifera reference strain. Based on the previous experience that the initial onset of symptoms occurred no later than 12 d after infection, the experiment was terminated on day 16; all remaining mice at this time point were free of clinical symptoms and on necropsy showed no or only minor pulmonary and/or renal alterations characterized by lymphocytic infiltrates. The histological lung score correlated with the time of survival for both fungal species (see Fig. S3), suggesting that histological alterations and virulence are quantitatively associated. It should however be noted that histological samples from different time points after infection were used which limits the extend of the conclusions that can be drawn from this correlation.
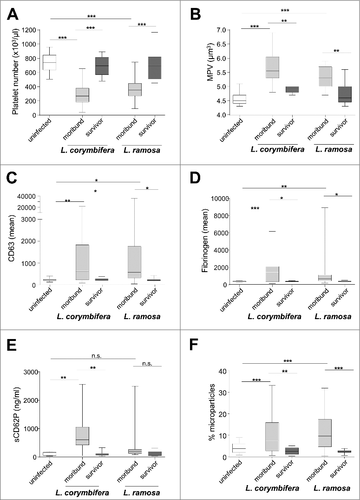
Figure 4. Platelet activation during Lichtheimia infection in corticosteroid-treated mice. (n = 6 mice/group/experiment). (A) Number of circulating platelets. (B) Mean platelet volume. (C) Surface expression of CD63 on circulating platelets. (D) Binding of fibrinogen to circulating platelets. (E) Quantification of soluble CD62P (sCD62P) in plasma. (F) Formation of platelet-derived microparticles circulating in the plasma. Statistical significance was calculated using 2-sided unpaired t-test, #p < 0.05 and ##p < 0.01
Lethal Lichtheimia infection is accompanied by increased platelet activation and reduced platelet numbers
Angioinvasion and thrombosis were commonly observed in infected tissues; to determine whether systemically platelet activation occurred, peripheral blood samples from PBS control mice, moribund animals and surviving mice 16 d p. inf. were analyzed. Moribund animals showed reduced numbers of circulating platelets () and increased mean platelet volume (), consistent with increased consumption of platelets during thrombus formation and increased release of young platelets from the bone marrow. Furthermore, the platelets circulating in the periphery were significantly more activated in moribund than in surviving mice as shown by enhanced CD63 expression on platelet surfaces (), implying dense granule release. Similarly, fibrinogen binding () on the platelet surface was higher in moribund than in surviving mice, suggesting enhanced tendency of clot formation and thrombosis.Citation27 Additionally, levels of secreted CD62P () and formation of microparticles () were increased in the plasma of moribund animals. These platelet-derived pro-inflammatory factors might contribute to excessive inflammation and pathophysiology of infected animals.Citation28
Discussion
Mucormycoses are rare but life-threatening fungal infections that usually affect patients with immunosuppression due to neutropenia, corticosteroid treatment, or diabetes.Citation1,29 Therapeutic interventions are often difficult and require a combination of surgical removal of infected foci and antifungal therapy, still resulting in overall high mortality.Citation1,20 Thus, there is a need to better understand these infections and murine models are an essential tool to investigate pathogenesis and assess treatment efficacy. In contrast to Rhizopus, Lichtheimia infections have not been systematically studied in mice yet. We therefore established a pulmonary infection model that is based on models widely used for Rhizopus, Aspergillus and other fungi, thereby allowing future comparative studies. Outbred mice were chosen for this study to avoid any possible influence of specific genetic backgrounds in inbred mouse lines on the results.
Similar to infection with Rhizopus,Citation12 immunosuppression significantly increased susceptibility of mice to pulmonary Lichtheimia infection. This is consistent with epidemiological studies in humans, in which only a minority of mucormycosis patients were identified as immunocompetentCitation1,3-5 and these patients commonly had severe trauma as a predisposing factor,Citation30 which likely allowed introduction of spores directly into deeper tissues. In contrast to our results, intravenous infection of immunocompetent mice resulted in severe systemic infection.Citation31 However, this route of inoculation leads to direct dissemination and thus likely mimics traumatic inoculation rather than pulmonary exposure. Consistent with this hypothesis, mucormycosis induced by intravenous infection affected mainly brain and kidneys but rarely the lung, reflecting observations in human patients with mucormycosis following trauma.Citation1,3,4
The pathological alterations that developed following intranasal challenge of corticosteroid-treated mice accurately mimicked clinical hallmarks of mucormycosis: vascular invasion associated with thrombosis and tissue infarction/necrosis.Citation20 Angioinvasion has been associated with the ability of the pathogen to hematogenously disseminate from the primary site of infection toward other organs,Citation20 which we also frequently observed following intranasal infection and establishment of pulmonary mucormycosis. Interestingly, we also observed strong systemic activation of platelets in mice with severe infection. The observation of necrotic areas in various organs in the absence of overt fungal growth might be the result of thrombotic ischemia, either mediated by general platelet activation or fungal growth in other parts of the organ. Whether platelets activation was a result of angioinvasion alone or whether specific fungal factors contribute to this phenomenon remains to be elucidated. It is however tempting to speculate that platelet activation with subsequent clotting and thrombosis as well as formation of pro-inflammatory factors such as sCD62P and microparticles contribute to pathogenesis and worsened outcome of infection. Overall, the clinical disease following intranasal infection of corticosteroid-treated mice accurately reflected the most common manifestation of mucormycosis in immunocompromised patients.
In our hands, around 50% of mice immunosuppressed with cortisone acetate reproducibly succumbed to infection with virulent L. corymbifera and L. ramosa strains, with some variation between experiments. It appears likely that more severe forms of immunosuppression, e.g., neutropenia or leucopenia, would further increase susceptibility as previously observed for pulmonary aspergillosis caused by A. terreusCitation19; there, 100% mortality could be achieved in leukopenic, but not cortisone acetate-treated mice. This suggests that the residual host defense system might be sufficient to clear the infection in some cortisone acetate-treated mice. Importantly, some strain-specific virulence differences may depend on the type of immunosuppression used, as has been demonstrated for the role of gliotoxin in the pathogenesis of aspergillosis.Citation32 Thus, it should be noted that the results presented here based on corticosteroid-treatment are not necessarily transferable to other modes of immunosuppression.
While we did not expect development of clinical mucormycosis in immunocompetent mice, we were surprised that none of the DKA mice developed clinical symptoms. Diabetic ketoacidosis has been described as an important clinical risk factor for mucormycosisCitation1-4 and DKA mice readily succumb to infection with R. arrhizus (formerly R. oryzae),Citation12,Citation17,Citation33 (and data not shown). Diabetic ketoacidosis is associated with increased expression of the endothelial heat shock host receptor glucose-regulated protein 78 (GRP78).Citation26 R. arrhizus CotH binds to GRP78 thereby mediating endocytosis.Citation26,34,35 R. arrhizus CotH has been shown to be essential for R. arrhizus virulence in a ketoacidotic mouse model.Citation34 As CotH genes are also present in the sequenced Lichtheimia genomes,Citation36,37 it could be assumed that they likewise contribute to infection. However, their function and expression in vivo has not been analyzed so far and it appears possible that functional differences in the CotH genes explain why DKA mice are less susceptible to pulmonary Lichtheimia infection. Furthermore, differences in the interaction with innate immune cells could contribute to the differential susceptibility of DKA mice to Rhizopus and Lichtheimia: L. corymbifera has been shown to be more susceptible to killing by neutrophilic granulocytes,Citation38,39 and this may explain the resistance of DKA mice to intranasal Lichtheimia infection. Finally, strain-specific differences might contribute to these findings: We showed that L. corymbifera strains differ in their virulence potential in immunocompromised mice, and thus, our findings do not exclude that specific Lichtheimia strains are capable of inducing infections in ketoacidotic animals. Reinhardt et al. used ketotic rabbits and found that 13/13 Rhizopus strains but only 1/13 L. corymbifera (Absidia corymbifera) strains were pathogenic in this model.Citation40 Of note, treatment with cortisone acetate also led to elevated blood glucose (175 ± 37 mg/dL on day 0 compared with 134 ± 11 mg/dL in untreated control animals). However, glucose levels were significantly lower than in DKA mice at day -3 and at the end of the experiment (421 ± 138 mg/dL and 499 ± 158 mg/dL; p < 0.0001 compared with cortisone acetate treated mice, Mann-Whitney test) and did not exceed 240 mg/dL. It therefore is unlikely that cortisone acetate treated mice in our model developed ketoacidosis.
An important difference between models used previously by others and our study is the lack of additional immunosuppression by corticosteroids in this study. Thus, while our data shows that DKA alone is not sufficient to promote Lichtheimia mucormycosis, it yet appears possible that DKA affects susceptibility to Lichtheimia in hosts with underlying immune dysfunction. This still needs to be determined experimentally. Furthermore, mucormycosis in diabetic hosts typically manifests as rhinocerebral infection.Citation1,3,4 We did not observe any clinical symptoms suggestive of affection of sinuses or the brain, e. g. torticollis, in DKA mice in our study; however, this does not exclude subclinical manifestations that could progress to life-threatening disease in the presence of other risk factors such as immunosuppression.
It is tempting to speculate that the differences in risk factors also affect the relative prevalence of Lichtheimia infections: In Europe, where Lichtheimia is a relatively common cause of mucormycosis, ketoacidosis was reported in less than 20% of the patients with mucormycosis.Citation3-5 In a global survey based on published data, ketoacidosis was present in 36% of the cases and Lichtheimia was less frequently isolated.Citation1 Furthermore, diabetic patients appear to be more frequently developed rhinocerebral mucormycosis, whereas pulmonary infections are more commonly associated with hematological malignancies, corticosteroid treatment or other forms of immunosuppression.Citation1,3,4 Studies linking risk factors, localization of mucormycosis and the fungal species involved are sparse; however, one study found that R. arrhizus was responsible for 85% of all rhinocerebral cases but only isolated from 17% of non-rhinocerebral forms.Citation3 This could imply that fungal-specific differences in the preferred route of entry and manifestation of mucormycosis exist. However, the available epidemiological data on mucormycosis is limited both regarding the number of studies, the number of cases included, and the methodology used in individual studies. Thus, no final conclusions can be drawn from the available data.
In summary, we established a murine pulmonary infection model which revealed that immunosuppression was essential for establishment of infection. The disease was characterized by necrosis, angioinvasion and thrombosis. Dissemination from the lung to other internal organs, especially the kidneys, was frequently observed. Although ketoacidosis induced by streptozotocin treatment promotes Rhizopus infection in mice, it did not predispose mice to infection with L. corymbifera or L. ramosa. This finding might partially explain the dominance of Rhizopus as causative agent of mucormycosis in countries with high prevalence of ketoacidotic patients. However, more comprehensive epidemiological studies are needed to elucidate whether associations between route of infection, distinct risk factors and the species causing mucormycosis do indeed exist.
Disclosure of potential conflicts of interest
No potential conflicts of interest were disclosed.
KVIR_S_1360460.zip
Download Zip (20.9 MB)Acknowledgments
We thank Birgit Weber for technical assistance and preparation of the histological slides, Caroline Semm for preparing the Lichtheimia spore suspensions, and Nicole Engert, Silke Machata, Maria Joanna Niemiec, and Birgit Weber for mouse husbandry.
Part of the data has been presented at the 65th Annual Meeting of the German Society for Hygiene and Microbiology (DGHM), 22.-25. September 2013, Rostock, Germany.
Funding
This work was supported by the Deutsche Forschungsgemeinschaft (CRC/TR 124 FungiNet, to IDJ and KV) and the Wissenschaftsfond (FWF, project P26117-B20, to CS and GR).
References
- Roden MM, Zaoutis TE, Buchanan WL, Knudsen TA, Sarkisova TA, Schaufele RL, Sein M, Sein T, Chiou CC, Chu JH, et al. Epidemiology and outcome of zygomycosis: A review of 929 reported cases. Clin Infect Dis. 2005; 41:634-53. doi:https://doi.org/10.1086/432579. PMID:16080086
- Adam RD, Hunter G, DiTomasso J, Comerci G Jr. Mucormycosis: Emerging prominence of cutaneous infections. Clin Infect Dis. 1994;19:67-76. doi:https://doi.org/10.1093/clinids/19.1.67. PMID:7948560
- Lanternier F, Dannaoui E, Morizot G, Elie C, Garcia-Hermoso D, Huerre M, Bitar D, Dromer F, Lortholary O; French Mycosis Study Group. A global analysis of mucormycosis in France: The RetroZygo study (2005-2007). Clin Infect Dis. 2012;54 Suppl 1:S35-43. doi:https://doi.org/10.1093/cid/cir880. PMID:22247443
- Skiada A, Pagano L, Groll A, Zimmerli S, Dupont B, Lagrou K, Lass-Florl C, Bouza E, Klimko N, Gaustad P, et al. Zygomycosis in Europe: Analysis of 230 cases accrued by the registry of the European Confederation of Medical Mycology (ECMM) working group on zygomycosis between 2005 and 2007. Clin Microbiol Infect. 2011;17:1859-67. doi:https://doi.org/10.1111/j.1469-0691.2010.03456.x. PMID:21199154
- Pana ZD, Seidel D, Skiada A, Groll AH, Petrikkos G, Cornely OA, Roilides E; Collaborators of Zygomyco.net and/or FungiScope™ Registries. Invasive mucormycosis in children: An epidemiologic study in European and non-European countries based on two registries. BMC Infect Dis. 2016;16:667; doi:https://doi.org/10.1186/s12879-016-2005-1. PMID:27832748
- de A. Santiago ALCM, Hoffmann K, Lima DX, de Oliveira RJV, Vieira HEE, Malosso E, et al. A new species of Lichtheimia (Mucoromycotina, Mucorales) isolated from Brazilian soil. Mycol Prog. 2014;13:343-52. doi:https://doi.org/10.1007/s11557-013-0920-8.
- Alastruey-Izquierdo A, Hoffmann K, de Hoog GS, Rodriguez-Tudela JL, Voigt K, Bibashi E, Walther G. Species recognition and clinical relevance of the zygomycetous genus Lichtheimia (syn. Absidia pro parte, Mycocladus). J Clin Microbiol. 2010;48:2154-70. doi:https://doi.org/10.1128/JCM.01744-09. PMID:20357218
- Schwartze VU, Hoffmann K, Nyilasi I, Papp T, Vagvolgyi C, de Hoog S, Voigt K, Jacobsen ID. Lichtheimia species exhibit differences in virulence potential. PloS One. 2012;7:e40908; doi:https://doi.org/10.1371/journal.pone.0040908. PMID:22911715
- Schwartze VU, Santiago AL, Jacobsen ID, Voigt K. The pathogenic potential of the Lichtheimia genus revisited: Lichtheimia brasiliensis is a novel, non-pathogenic species. Mycoses. 2014;57 Suppl 3:128-31. doi:https://doi.org/10.1111/myc.12230. PMID:25267009
- Kamei K. Animal models of zygomycosis–Absidia, Rhizopus, Rhizomucor, and Cunninghamella. Mycopathologia. 2001;152:5-13. doi:https://doi.org/10.1023/A:1011900630987. PMID:11694096
- Ibrahim AS, Gebremariam T, Lin L, Luo G, Husseiny MI, Skory CD, Fu Y, French SW, Edwards JE Jr., Spellberg B The high affinity iron permease is a key virulence factor required for Rhizopus oryzae pathogenesis. Mol Microbiol. 2010;77:587-604. doi:https://doi.org/10.1111/j.1365-2958.2010.07234.x. PMID:20545847
- Waldorf AR, Ruderman N, Diamond RD. Specific susceptibility to mucormycosis in murine diabetes and bronchoalveolar macrophage defense against Rhizopus. J Clin Invest. 1984;74:150-60. doi:https://doi.org/10.1172/JCI111395. PMID:6736246
- Ibrahim AS, Gebremariam T, Luo G, Fu Y, French SW, Edwards JE Jr., Spellberg B. Combination therapy of murine mucormycosis or aspergillosis with iron chelation, polyenes, and echinocandins. Antimicrob Agents Chemother. 2011;55:1768-70. doi:https://doi.org/10.1128/AAC.01577-10. PMID:21263057
- Salas V, Pastor FJ, Calvo E, Alvarez E, Sutton DA, Mayayo E, Fothergill AW, Rinaldi MG, Guarro J. In vitro and in vivo activities of posaconazole and amphotericin B in a murine invasive infection by Mucor circinelloides: Poor efficacy of posaconazole. Antimicrob Agents Chemother. 2012;56:2246-50. doi:https://doi.org/10.1128/AAC.05956-11. PMID:22290952
- Lewis RE, Albert ND, Liao G, Hou J, Prince RA, Kontoyiannis DP. Comparative pharmacodynamics of amphotericin B lipid complex and liposomal amphotericin B in a murine model of pulmonary mucormycosis. Antimicrob Agents Chemother. 2010;54:1298-304. doi:https://doi.org/10.1128/AAC.01222-09. PMID:20038620
- Lewis RE, Liao G, Wang W, Prince RA, Kontoyiannis DP. Voriconazole pre-exposure selects for breakthrough mucormycosis in a mixed model of Aspergillus fumigatus-Rhizopus oryzae pulmonary infection. Virulence. 2011;2:348-55. doi:https://doi.org/10.4161/viru.2.4.17074. PMID:21788730
- Luo G, Gebremariam T, Lee H, French SW, Wiederhold NP, Patterson TF, Filler SG, Ibrahim AS. Efficacy of liposomal amphotericin B and posaconazole in intratracheal models of murine mucormycosis. Antimicrob Agents Chemother. 2013;57:3340-7. doi:https://doi.org/10.1128/AAC.00313-13. PMID:23650163
- Ibrahim AS, Gebermariam T, Fu Y, Lin L, Husseiny MI, French SW, Schwartz J, Skory CD, Edwards JE Jr, Spellberg BJ. The iron chelator deferasirox protects mice from mucormycosis through iron starvation. J Clin Invest. 2007;117:2649-57. doi:https://doi.org/10.1172/JCI32338. PMID:17786247
- Slesiona S, Ibrahim-Granet O, Olias P, Brock M, Jacobsen ID. Murine infection models for Aspergillus terreus pulmonary aspergillosis reveal long-term persistence of conidia and liver degeneration. J Infect Dis. 2012;205:1268-77. doi:https://doi.org/10.1093/infdis/jis193. PMID:22438397
- Spellberg B, Edwards J Jr., Ibrahim A. Novel perspectives on mucormycosis: Pathophysiology, presentation, and management. Clin Microbiol Rev. 2005;18:556-69. doi:https://doi.org/10.1128/CMR.18.3.556-569.2005. PMID:16020690
- Kontoyiannis DP, Yang H, Song J, Kelkar SS, Yang X, Azie N, Harrington R, Fan A, Lee E, Spalding JR. Prevalence, clinical and economic burden of mucormycosis-related hospitalizations in the United States: A retrospective study. BMC Infect Dis. 2016;16:730; doi:https://doi.org/10.1186/s12879-016-2023-z. PMID:27905900
- Gebremariam T, Lin L, Liu M, Kontoyiannis DP, French S, Edwards JE Jr., Filler SG, Ibrahim AS. Bicarbonate correction of ketoacidosis alters host-pathogen interactions and alleviates mucormycosis. J Clin Invest. 2016;126:2280-94. doi:https://doi.org/10.1172/JCI82744. PMID:27159390
- Winzell MS, Ahren B. The high-fat diet-fed mouse: A model for studying mechanisms and treatment of impaired glucose tolerance and type 2 diabetes. Diabetes. 2004;53 Suppl 3:S215-9. doi:https://doi.org/10.2337/diabetes.53.suppl_3.S215. PMID:15561913
- Kitada M, Ogura Y, Koya D. Rodent models of diabetic nephropathy: Their utility and limitations. Int J Nephrol Renovasc Dis. 2016;9:279-90. doi:https://doi.org/10.2147/IJNRD.S103784. PMID:27881924
- Luo G, Gebremariam T, Clemons KV, Stevens DA, Ibrahim AS. Heat-killed yeast protects diabetic ketoacidotic-steroid treated mice from pulmonary mucormycosis. Vaccine. 2014;32:3573-6. doi:https://doi.org/10.1016/j.vaccine.2014.04.086. PMID:24814556
- Liu M, Spellberg B, Phan QT, Fu Y, Fu Y, Lee AS, Edwards JE Jr, Filler SG, Ibrahim AS. The endothelial cell receptor GRP78 is required for mucormycosis pathogenesis in diabetic mice. J Clin Invest. 2010;120:1914-24. doi:https://doi.org/10.1172/JCI42164. PMID:20484814
- Budnik I, Shenkman B, Hauschner H, Zilinsky I, Savion N. Role of heterotrimeric G proteins in platelet activation and clot formation in platelets treated with integrin alphaIIbbeta3 inhibitor. Platelets. 2017:1-5. [Advance online publication]. doi:https://doi.org/10.1080/09537104.2017.1295136. PMID:28406726.
- Varon D, Shai E. Platelets and their microparticles as key players in pathophysiological responses. J Thromb Haemost. 2015;13 Suppl 1:S40-6. doi:https://doi.org/10.1111/jth.12976. PMID:26149049
- Warkentien T, Rodriguez C, Lloyd B, Wells J, Weintrob A, Dunne JR, Ganesan A, Li P, Bradley W, Gaskins LJ, et al. Invasive mold infections following combat-related injuries. Clin Infect Dis. 2012;55:1441-9. doi:https://doi.org/10.1093/cid/cis749. PMID:23042971
- Skiada A, Pagano L, Groll A, Zimmerli S, Dupont B, Lagrou K, Lass-Florl C, Bouza E, Klimko N, Gaustad P, et al. Zygomycosis in Europe: Analysis of 230 cases accrued by the registry of the European Confederation of Medical Mycology (ECMM) working group on zygomycosis between 2005 and 2007. Clin Microbiol Infect. 2011;17:1859-67. doi:https://doi.org/10.1111/j.1469-0691.2010.03456.x. PMID:21199154
- Dannaoui E, Mouton JW, Meis JF, Verweij PE, Eurofung N. Efficacy of antifungal therapy in a nonneutropenic murine model of zygomycosis. Antimicrob Agents Chemother. 2002;46:1953-9. doi:https://doi.org/10.1128/AAC.46.6.1953-1959.2002. PMID:12019114
- Ben-Ami R, Lewis RE, Kontoyiannis DP. Enemy of the (immunosuppressed) state: An update on the pathogenesis of Aspergillus fumigatus infection. Br J Haematol. 2010;150:406-17. doi:https://doi.org/10.1111/j.1365-2141.2010.08283.x. PMID:20618330
- Spellberg B, Fu Y, Edwards JE Jr., Ibrahim AS. Combination therapy with amphotericin B lipid complex and caspofungin acetate of disseminated zygomycosis in diabetic ketoacidotic mice. Antimicrob Agents Chemother. 2005;49:830-2. doi:https://doi.org/10.1128/AAC.49.2.830-832.2005. PMID:15673781
- Gebremariam T, Liu M, Luo G, Bruno V, Phan QT, Waring AJ, Edwards JE Jr, Filler SG, Yeaman MR, Ibrahim AS. CotH3 mediates fungal invasion of host cells during mucormycosis. J Clin Invest. 2014;124:237-50. doi:https://doi.org/10.1172/JCI71349. PMID:24355926
- Ibrahim AS, Spellberg B, Avanessian V, Fu Y, Edwards JE Jr. Rhizopus oryzae adheres to, is phagocytosed by, and damages endothelial cells in vitro. Infect Immun. 2005;73:778-83. doi:https://doi.org/10.1128/IAI.73.2.778-783.2005. PMID:15664916
- Chibucos MC, Soliman S, Gebremariam T, Lee H, Daugherty S, Orvis J, Shetty AC, Crabtree J, Hazen TH, Etienne KA, et al. An integrated genomic and transcriptomic survey of mucormycosis-causing fungi. Nature Commun. 2016;7:12218; doi:https://doi.org/10.1038/ncomms12218. PMID:27447865
- Schwartze VU, Winter S, Shelest E, Marcet-Houben M, Horn F, Wehner S, Linde J, Valiante V, Sammeth M, Riege K, et al. Gene expansion shapes genome architecture in the human pathogen Lichtheimia corymbifera: An evolutionary genomics analysis in the ancient terrestrial mucorales (Mucoromycotina). PLoS Genet. 2014;10:e1004496; doi:https://doi.org/10.1371/journal.pgen.1004496. PMID:25121733
- Simitsopoulou M, Roilides E, Maloukou A, Gil-Lamaignere C, Walsh TJ. Interaction of amphotericin B lipid formulations and triazoles with human polymorphonuclear leucocytes for antifungal activity against Zygomycetes. Mycoses. 2008;51:147-54. doi:https://doi.org/10.1111/j.1439-0507.2007.01457.x. PMID:18254752
- Gil-Lamaignere C, Simitsopoulou M, Roilides E, Maloukou A, Winn RM, Walsh TJ. Interferon- gamma and granulocyte-macrophage colony-stimulating factor augment the activity of polymorphonuclear leukocytes against medically important zygomycetes. J Infect Dis. 2005;191:1180-7. doi:https://doi.org/10.1086/428503. PMID:15747255
- Reinhardt DJ, Licata I, Kaplan W, Ajello L, Chandler FW, Ellis JJ. Experimental cerebral zygomycosis in alloxan-diabetic rabbits: Variation in virulence among zygomycetes. Sabouraudia. 1981;19:245-56. doi:https://doi.org/10.1080/00362178185380421. PMID:6798698
