Abstract
Emerging drug resistance in clinical isolates of Staphylococcus aureus might be implicated to the overexpression of NorA efflux pump which is capable of extruding numerous structurally diverse compounds. However, NorA efflux pump is considered as a potential drug target for the development of efflux pump inhibitors. In the present study, NorA model was constructed based on the crystal structure of glycerol-3-phosphate transporter (PDBID: 1PW4). Molecular dynamics (MD) simulation was performed using NAMD2.7 for NorA which is embedded in the hydrated lipid bilayer. Structural design of NorA unveils amino (N)- and carboxyl (C)-terminal domains which are connected by long cytoplasmic loop. N and C domains are composed of six transmembrane α-helices (TM) which exhibits pseudo-twofold symmetry and possess voluminous substrate binding cavity between TM helices. Molecular docking of reserpine, totarol, ferruginol, salvin, thioxanthene, phenothiazine, omeprazole, verapamil, nalidixic acid, ciprofloxacin, levofloxacin, and acridine to NorA found that all the molecules were bound at the large hydrophobic cleft and indicated significant interactions with the key residues. In addition, structure-based virtual screening was employed which indicates that 14 potent novel lead molecules such as CID58685302, CID58685367, CID5799283, CID5578487, CID60028372, ZINC12196383, ZINC72140751, ZINC72137843, ZINC39227983, ZINC43742707, ZINC12196375, ZINC66166948, ZINC39228014, and ZINC14616160 have highest binding affinity for NorA. These lead molecules displayed considerable pharmacological properties as evidenced by Lipinski rule of five and prophecy of toxicity risk assessment. Thus, the present study will be helpful in designing and synthesis of a novel class of NorA efflux pump inhibitors that restore the susceptibilities of drug compounds.
Introduction
Staphylococcus aureus is one of the major emerging multidrug-resistant pathogenic bacteria whose infections have been increasing alarmingly causing 19,000 deaths per year.Citation1 Drug access is overdue by numerous resistant mechanisms such as drug inactivation, target-based mutation, reduced drug access, and efflux pumps. The multidrug efflux systems play an important role in imparting resistance in bacteria and this seems to be a major setback in designing antimicrobial agents. In recent times, the complete genome sequence of S. aureus explored 30 efflux pump genes which belong to the major facilitator super family (MFS).Citation2 MFS transporters reveal that all MFS proteins possess a uniform topology of 12 transmembrane (TM) α-helices which are connected by hydrophilic loops at amino (N) and carboxyl (C) termini in the cytoplasm.Citation3 MFS proteins are dependent on proton motif force that transports the assorted substrates such as ions, sugars, sugar phosphates, drugs, neurotransmitters, nucleosides, amino acids, and peptides across the TM through three distinct transport mechanisms such as uniport, symport, and antiport. However, substrate specificity and transport mechanism of all MFS proteins vary and are less understood. Analysis by using structural, computational, and biochemical techniques reveals that MFS transporters possess a single binding site, alternating access mechanism that involves rocker switch-type movement of the protein.Citation4,Citation5
NorA efflux pump is one of the major overexpressed efflux pumps in the blood stream clinical isolates of S. aueusCitation6,Citation7 and involved in the efflux of multiple diverse drug compounds such as quinolones, fluoroquinolones,Citation8–Citation10 quarternary ammonium compounds and antiseptics,Citation11,Citation12 phenothaizines and thioxanthenes,Citation13 verapamil and omeprazole,Citation14 totarol, reserpine, ferruginol, carnosic acid, and dyes (such as ethidium bromide, rhodamine, acridine, and biocides).Citation15 NorA belongs to MFS transporters which are embedded in the membranes of prokaryotes and eukaryotes.Citation16 Till now, the crystal structure of NorA efflux pump has not been resolved, although the crystal structures of a confined number of MFS proteins, namely GlpT (antiporter),Citation17 LacY (symporter),Citation18 EmrD,Citation19 and OxlT, were resolved.Citation20 Dysfunction of efflux pumps could potentiate the antibacterial activity of the drugs, and this strategy has been attracting many researchers around the globe to develop a novel potent antibacterial compound.Citation21 So far, numerous potent efflux pump inhibitors (EPIs) such as derivatives of piperine,Citation22 boronic species,Citation23 N-caffeyl phenalkymanide,Citation24 2-aryl-5-nitro-1H-indoles,Citation25 aryl benzothiophenes and diarylthiophene,Citation26 6-amino 8-methyl quinolone ester, and some plant-derived compoundsCitation27,Citation28 have been reported to target NorA. Conversely, these compounds are unable to enter into the clinical settings due to several pharmacological adverse side effects, especially when administered for longer duration. Besides, the continuous usage of these drugs causes development of drug resistance in pathogenic bacteria. Hence, the present investigation was undertaken to elucidate the structural characteristics and binding mechanism of NorA efflux pump and design potent EPIs for effective clinical management of S. aureus.
Materials and methods
Prophecy of TM helices
NorA protein sequence of S. aureus was retrieved from UniProt database, and TM helices were predicted using various servers such as TMHMM,Citation29 TMpred,Citation30 SOSUI,Citation31 DAS,Citation32 HMMTOP,Citation33 Predict protein,Citation34 and TopPred IICitation35 to confirm origin and end of the helices.
Homology modeling
Three-dimensional (3D) structure of NorA was elucidated on the basis of sequence identity with high score, less e-value, highest resolution, and R-factor of the template structure by performing BLASTP search against Protein Data Bank (PDB). The coordinates for the query structure through pairwise sequence alignment were assigned from template structure by using Clustal X.Citation36 Subsequently, the 3D models were built by using MODELLER 9.14,Citation37 and the least modeler objective (low discrete optimized protein energy score) was chosen. The final model was subjected to molecular dynamics (MD) simulations.
MD simulation
NorA model with the least DOPE score was improved by applying MD simulations using NAMD2.7 software.Citation38 Chemistry at HARvard Macromolecular Mechanics (CHARMM2.7) force field was used for lipids and proteinsCitation39 along with the three-site transferable intermolecular potential for water and palmitoyl oleyl phosphatidyl choline for lipids (). Initially, the membrane and the protein complex were minimized and equilibrated with 250,000 runs for 10 picoseconds, and the simulations were performed for 1,000,000 runs for 2 nanoseconds. Integrated motion time step of 2 fs was computed using multiple time step algorithms.Citation40 Short range forces were computed for every two time steps, and long range forces were calculated for every four time steps. The pair list of the nonbonded interactions was computed with a pair list distance of 14.0 Å. Short-range interactions, within 12 Å, were defined as van der Waals and electrostatic interactions. Long-range electrostatic interactions were taken into account using particle mesh Ewald approach.Citation41–Citation43 Pressure was maintained at 1 atm using the Langevin piston and temperature was controlled at 300 K using Langevin dynamics. Covalent interactions between hydrogen and heavy atoms were constrained using SHAKE/RATTLE algorithm.Citation44
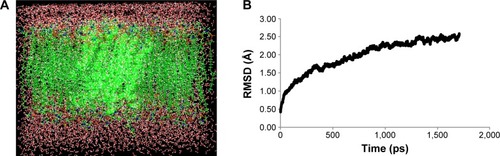
Figure 1 MD simulation of NorA efflux pump.
Abbreviations: RMSD, root-mean-square deviation; ps, picoseconds.
Model assessment
The quality of the model was assessed by calculating the stereochemical properties, compatibility of the atomic model (3D) with its own amino acid residues (1D), bond lengths, bond angles, and side-chain planarity using SAVES server (http://nihserver.mbi.ucla.edu/SAVES/). Ramachandran plot calculations were performed using PROCHECK to check the stereochemical quality of protein structure.Citation45 Environment profile was developed using Verify3DCitation46 and ERRAT.Citation47 The residue packing and atomic interactions were analyzed using WHATIF, and Ramachandran plot was analyzed using WHATCHECK.Citation48 Root-mean-square deviation (RMSD) of the model was calculated by superimposition of the 3D model with template using Swiss-Pdb Viewer.Citation49
Retrieval of ligands
Phytochemicals such as alkaloids (reserpine), terpenoids (ferruginol, totarol, salvin), xanthenes (thioxanthene, phenothiazone), verapamil, omeprazole, fluoroquinolones (levofloxacin, nalidixic acid, and ciprofloxacin), and dyes (acridine) were downloaded from PubChem. Reserpine analogs were retrieved from PubChem and ZINC database.
Virtual screening and docking
Structure-based virtual screening studies were carried out using AutoDock Vina 4.0Citation50 with PyRx.Citation51 Initially, all the ligand molecules were uploaded and energy minimized with universal force field using conjugate-gradient algorithm with 200 run iterations. Virtual screening was carried out against NorA efflux pump by using Lamarkian genetic algorithm. Docking parameters were set as follows: the number of individuals in the population was 150, maximum number of energy evaluations was 25,000, maximum number of generations was 27,000, top individual to survive to next generation was 1, gene mutation rate was 0.02, crossover rate was 0.8, Cauchy beta was 1.0, and genetic algorithm window size was 10.0. The grid was set to the binding pocket at X=29.3901, Y=−42.745, Z=−51.82; dimensions (Å) at X=90.000, Y=105.7097, Z=104.2448; and exhaustiveness at 8. The best docked ligand conformations were saved, and the bond angles, bond lengths, and hydrogen bonding interactions were analyzed using PyMOL.Citation52
Toxicity risk assessment and Lipinski rule of five
Lipinski rule of five, that is, molecular weight (<500 Da), H-bond acceptor (<10), H-bond donor (<5), and cLogP (<5) values; toxicity properties such as mutagenic, tumorogenic, irritant, and reproductive effects; and absorption, distribution, metabolize, excretion, and transport properties were assessed using OSIRIS server (http://www.organic-chemistry.org/prog/peo) and Molinspiration (http://www.molinspiration.com/cgi-bin/properties).
Results and discussion
Prophecy of TM helices
Protein sequence of NorA efflux pump (account number: Q6GIU7) consisting of 387 amino acids was retrieved from UniProt, and prophecy of TM helices has shown different helices at different positions, namely Helix-I (6–30), Helix-II (40–62), Helix-III (69–91), Helix-IV (94–119), Helix-V (127–151), Helix-VI (157–178), Helix-VII (201–227), Helix-VIII (237–259), Helix-IX (262–375), Helix-X (288–351), Helix-XI (326–350), and Helix-XII (357–375) (). However, Helix-IV, Helix-VI, and Helix-XI by TMpred; Helix-VI and Helix-XII by TMHMM; Helix-XII by SOUSI; and TopPred have not been recognized ().
Table 1 Prediction of TM helices of NorA efflux pump using different servers
NorA model construction
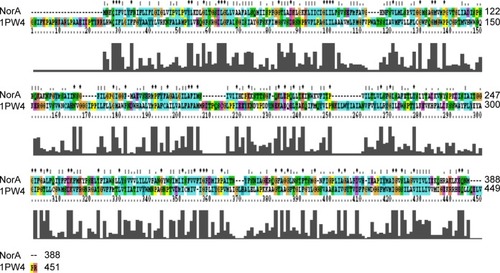
Glycerol-3-phosphate transporter (PDBID: 1PW4) belongs to the MFS and was selected based on highest resolution (3.3 Å), R-value (0.296), and R-free (0.325) as template.Citation53 Based on the spatial restraints, the coordinates of template to structurally conserved regions, structurally variable region, and N- and C-termini have been assigned to query the sequence (). Subsequently, all the side chains were fixed by rotamers and 100 models were generated, and the lowest DOPE score structure was selected as the best model for NorA. Variations in secondary structural elements such as irregular helices, sheets, and loops were identified by superimposing with template, and a small loop, observed between Ala20 and Val22 of Helix-I, was renovated using MOD loop server (https://modbase.compbio.ucsf.edu/modloop).
Figure 2 Pairwise alignment of NorA efflux pump of Staphylococcus aureus with glycerol-3-phosphate transporter (PDBID: 1PW4) of Escherichia coli.
To obtain stable conformation, hydrogens were added to the model initially, and the energy was minimized followed by MD simulations for 2 nanoseconds. Later on, trajectory graph was plotted by taking RMSD of Cα trace on X-axis and time (picoseconds) on Y-axis that showed a sharp increase up to 1.5 nanoseconds and achieved equilibrium at around 2 nanoseconds (). The refined model possesses 12 α-helices and one long cytoplasmic connecting loop, and the quality of the model was checked by SAVES server.
Model assessment
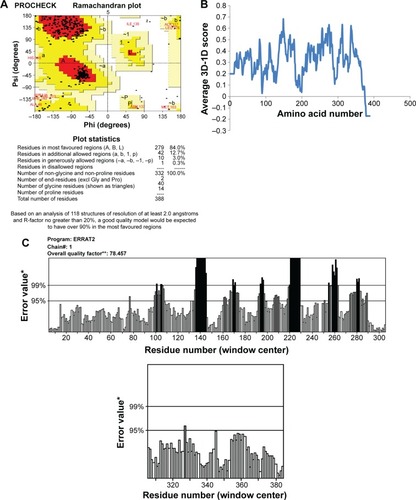
This final model was corroborated with Ramachandran plot calculations using PROCHECK which revealed 308 residues (92.8%) were aligned within the most favored region, 21 residues (6.3%) were located within the additionally allowed regions, three residues (0.9%) were plotted within the generously allowed region, and no residues were aligned within the disallowed region (). The environment profile of Verify3D–1D value was found to be mostly above zero (). The nonbonded interactions between various types of atoms were computed with ERRAT program which showed an overall quality factor of 80.0 (). WHATCHECK program calculated quality indicators such as second-generation packing quality, Ramachandran plot appearance, and chi-1/chi-2 rotamer normality and were found to be 2.7, 0.725, and 2.0, respectively; RMS Z-scores such as bond lengths, bond angles, omega angle restraints, side-chain planarity, improper dihedral distribution, and inside/outside distribution were found to be 0.98, 0.803, 0.564, 1.08, 1.15, and 1.23, respectively. Superimposition of NorA and GlpT recorded an RMSD of 0.86 Å for Cα atoms.
Figure 3 Assessment of NorA efflux pump structure.
Structure of NorA and binding-site analysis
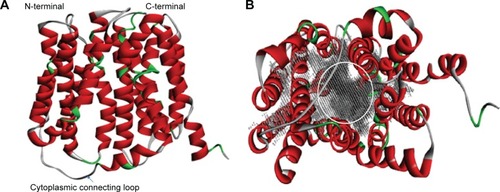
NorA efflux pump is a single polypeptide chain which exhibits 12 TM α-helices topology with two domains, N- and C-terminal domains, arranged as pseudo-twofold symmetry. Both N- and C-terminal domains are connected by a long cytoplasmic loop (Met172–Lys198) between Helix-VI and Helix-VII (). N-terminal domain consists of six TM α-helices: TM Helix-I (Met1–Ileu30), TM Helix-II (Leu40–Lys64), TM Helix-III (Gly66–Ala86), TM Helix-IV (Phe91–Ileu119), TM Helix-V (Gln124–Ile146), and TM Helix-VI (Arg156–Met172) and C-terminal domain consists of six TM α-helices: TM Helix-VII (Lys198–Tyr225), TM Helix-VIII (Asn234–Phe264), TM Helix-IX (Ser267–Leu286), TM Helix-X (Trp293–Ser318), TM Helix-XI (Gly326–Val353), and TM Helix-XII (Gly356–Arg380). Nevertheless, a large hydrophobic binding cleft is accompanied by eight TM helices of Helix-I, Helix-II, Helix-IV, and Helix-V from N terminal and Helix-VII, Helix-VIII, Helix-X, and Helix-XI from C terminal and composed of nonpolar residues such as Val44, Phe47, Gln51, Phe140, Ile244, Gly248, and Phe303 which are evolutionarily conserved among MFS transporters ().
Figure 4 3D structure of NorA efflux pump.
Molecular docking of NorA substrates
To find out the binding affinity of NorA substrates, molecular docking was performed, and the results are summarized in . From the docking results, reserpine showed highest binding energy of −8.7 kcal/mol and exhibited three binding interactions by the way of arene–arene interaction with aromatic ring of Phe317, arene–cationic interaction with Lys125, and two hydrogen bonding interactions by accepting the electrons from Arg324 and Phe129. Ferruginol and totarol are meroterpenes which showed binding affinities of −8.4 and −8.1 kcal/mol, respectively. Salvin is triterpene that exerts binding affinity of −7.1 kcal/mol; thioxanthene and phenothiazine have shown binding affinities of −8.1 and −7.1 kcal/mol, respectively, and these compounds were bound at the hydrophobic cleft by dint of hydrophobic interactions with nonpolar residues such as Phe13 and Leu17 of Helix-I, Phe47 and Gln51 of Helix-II, Met109 of Helix-IV, Ile136 and Phe140 of Helix-V, Leu212 and Phe216 of Helix-VII, and Phe341 of Helix-IX. Verapamil and omeprazole are proton pump and L-type calcium channel inhibitors that displayed binding affinities of −7.0 and −7.9 kcal/mol, respectively; verapamil forms one hydrogen bond by accepting the electron from the OH group of Tyr317, and omeprazole exhibits three bonds: two bonds by accepting the electrons from OH groups of Ser333 and one bond with OH group of Ser337. Quinolones such as nalidixic acid, ciprofloxacin, and levofloxacin are synthetic broad spectrum antimicrobial agents; alidixic acid showed lowest binding energy of −6.5 kcal/mol, and ciprofloxacin and levofloxacin showed binding energy of −8.1 and −7.8 kcal/mol, respectively. Nalidixic acid formed two bonds with OH group of Ser219 and Thr245, ciprofloxacin formed one hydrogen bond with amino group of Gln51, and levofloxacin interacted with nonpolar residues of hydrophobic cavity, that is, Phe13, Leu17, Ala105, Val108, Ile136, Phe140, Thr211, and Ala312. Acridine is a dye that shows binding affinity of −7.2 kcal/mol with nonpolar amino acids of the binding pocket ().
Table 2 Binding interactions, bond lengths, bond angles, and binding affinity of NorA substrate with active site residues of NorA efflux pump
Virtual screening and docking
In an effort to discover the selective novel potent NorA EPIs, virtual screening study was performed with the analogs of reserpine that monitored best binding lead molecules from the calculated binding affinities (). CID58685302 showed highest binding energy of −13.0 kcal/mol and bound at the vestibule of the binding pocket by means of four hydrogen bonding interactions: one with polar amide of Asn128 of Helix-V, one with hydroxyl group of Tyr316 of Helix-X, one with carboxylic group of Glu323, and one with amino group of Arg324 at the connecting loop of X–XI helices (). CID58685367 has shown binding affinity of −12.8 kcal/mol and formed two hydrogen bonds: one with polar amide on Gln51 of Helix-II and one bond with amino group on Lys130 of Helix-V (). CID5799283 showed binding energy of −12.4 kcal/mol and displayed one hydrogen bonding interaction with OH group on Ser333 of Helix-XI (). CID5578487 and CID60028372 have also shown similar binding affinity of −12.1 kcal/mol; CID5578487 showed two hydrogen bonds, that is, CO and NH groups with OH group of Ser219 and Ser337; CID60028372 interacted with three hydrogen bonds, that is, one bond with OH group of Thr333 and two bonds with OH group of Ser337 (). ZINC12196383 has shown good binding affinity of −12.4 kcal/mol and conferred two hydrogen bonding interactions formed by accepting electrons from the OH group on Ser219 of Helix-VII and Ser337 of Helix-XI, and its indole and aromatic rings interacted with nonpolar residues of binding cavity such as Phe16, Phe47, Gln51, Ala105, Ile136, and Phe140 (). ZINC72140751 has shown binding energy of −12.3 kcal/mol and displayed one hydrogen bonding interaction with OH group of Ser337 (). ZINC72137843, ZINC12196375, ZINC66166948, and ZINC39228014 have shown binding energy of −12.2, −11.7, −11.7, and −11.6 kcal/mol, respectively, and interacted with nonpolar residues of Phe13, Phe16, Leu17, and Gly20 of Helix-I; Phe47, Gln51, and Met55 of Helix-III; Ala105, Met109, Val112, Thr113, and Ile116 of Helix-IV; Asn128, Phe219, Thr131, Met132, Ile135, Ile136, and Phe140 of Helix-V; Tyr136 of Helix-X; and Gly326, Phe327, Gly330, Ser333, Thr334, Ser337, and Met338 of Helix-XI (). ZINC39227983, ZINC43742707, and ZINC14616160 compounds have shown best binding affinity of −11.8, −11.8, and −11.6 kcal/mol, respectively; ZINC39227983 exhibited one hydrogen bonding interaction with OH group on Thr211; and ZINC43742707 displayed one hydrogen bonding interaction with OH group of Ser337 (). ZINC14616160 displayed one hydrogen bonding interaction with polar residue Gln51 of the binding pocket ().
Table 3 Binding interactions, bond lengths, bond angles, and binding energies of lead molecules with active pocket residues of NorA efflux pump
Figure 6 Binding mode of lead compounds.

Toxicity risk assessment and Lipinski rule of five
Appraisal of pharmacological properties using Lipinski rule of five, such as molecular weight, H-bond donors, H-bond acceptors, cLogP, and toxicity properties reveals that lead molecules are found to be satisfied with these properties and are shown in . CID58685302, CID58685367, CID58685370, and CID57992283 have shown highest molecular weight. H-bond donors were predicted to be less than five, and H-bond acceptors less than ten. cLogP or partition coefficient plays a major role in accessing the drug in the body and value less than five indicates good absorption and distribution. Most of the compounds are found to be possessing best cLogP values except ZINC72137843, ZINC66166948, CID58685302, CID58685367, CID58685370, and CID57992283. Assessment of toxicity showed that none of the compounds exhibit serious adverse effects, but ZINC66166948 and CID5578487 show very low mutagenic property; CID58685302, CID58685367, and CID58685370 exhibit slight reproductive toxicity. Drug-likeness of the compounds was determined through the fragment of compounds which reveals that all the compounds showed good drug-likeness except ZINC39228014. Drug score was calculated for the above four risk factors which reveal that all the compounds displayed best drug score and were helpful in the development of EPIs for NorA.
Table 4 Lipinski rule of five and toxicity assessments of the lead compounds were predicted using Molinspiration and OSIRIS server
Conclusion
NorA pump consists of 12 TM helices and a large binding pocket between N- and C-terminal interfaces accommodates numerous drug compounds. Docking simulation studies of NorA substrates reveal that vital residues, namely Phe13, Phe14, Phe16, Leu17, Gly20, Phe47, Ala48, Gln51, Ala105, Gly106, Val108, Met109, Ile135, Ileu136, Phe140, Thr211, Leu212, Phe216, Ala312, and Phe341 present at the binding pocket play a pivotal role in extrusion of numerous compounds. In silico screening affords 14 good binding lead compounds which selectively inhibit the NorA efflux pump by competing with its substrates. Hence, these lead molecules will be useful for designing selective EPIs against NorA efflux pump of S. aureus.
Acknowledgments
The authors are grateful to the coordinator, Bioinformatics Infrastructure Facility, Department of Zoology, Sri Venkateswara University, Tirupati, for providing bioinformatics facilities.
Disclosure
The authors report no conflicts of interest in this work.
References
- KlevensRMMorrisonMANadleJInvasive methicillin-resistant Staphylococcus aureus infections in the United StatesJAMA2007298151763177117940231
- HassanKASkurrayRABrownMHActive export proteins mediating drug resistance in StaphylococciJ Mol Microbiol Biotechnol2007123–418019617587867
- PaoSSPaulsenITSaierMHJrMajor facilitator super familyMicrobiol Mol Biol Rev19986211349529885
- HuangYLemieuxMJSongJAuerMWangDNStructure and mechanism of the glycerol-3-phosphate transporter from Escherichia coliScience2003301563361662012893936
- LawCJYangQSoudantCMaloneyPCWangDNKinetic evidence is consistent with the rocker switch mechanism of membrane transport by GlpTBiochemistry20074643121901219717915951
- De MarcoCECushinLAFrempong-MansoESeoSMJaravazaTAKaatzGWEfflux-related resistance to norfloxacin, dyes, and biocides in bloodstream isolates of Staphylococcus aureusAntimicrob Agents Chemother20075193235323917576828
- Mohammed-AliMNJamalludeenNMIsolation and characterization of bacteriophage against methicillin resistant Staphylococcus aureusJ Med Microb Diagn20155213
- KaatzGWSeoSMRubleCAEfflux-mediated fluoroquinolone resistance in Staphylococcus aureusAntimicrob Agents Chemother1993375108610948517696
- NeyfakhAABorschCMKaatzGWFluoroquinolone resistance protein NorA of Staphylococcus aureus is a multidrug efflux transporterAntimicrob Agents Chemother200337128129
- GermanNWeiPKaatzGWKernsRJSynthesis and evaluation of fluoroquinolone derivatives as substrate-based inhibitors of bacterial efflux pumpsEur J Med Chem200843112453246318358571
- NoguchiNTamuraMNaruiKWakasugiKSasatsuMFrequency and genetic characterization of multidrug-resistant mutants of Staphylococcus aureus after selection with individual antiseptics and fluoroquinolonesBiol Pharm Bull20022591129113212230102
- LewisKKlibanovAMSurpassing nature: rational design of sterile-surface materialsTrends Biotechnol200523734334815922467
- KaatzGWMoudgalVVSeoSMKristiansenJEPhenothiazines and thioxanthenes inhibit multidrug efflux pump activity in Staphylococcus aureusAntimicrob Agents Chemother200347271972612543683
- VidaillacCGuillonJArpinCSynthesis of omeprazole analogues and evaluation of these as potential inhibitors of the multidrug efflux pump NorA of Staphylococcus aureusAntimicrob Agents Chemother200751383183817101679
- De MarcoCECushinLAFrempong-MansoESeoSMJaravazaTAKaatzGWEfflux-related resistance to norfloxacin, dyes, and biocides in bloodstream isolates of Staphylococcus aureusAntimicrob Agents Chemother20075193235323917576828
- SaierMHBeattyJTGoffeauAThe major facilitator super familyJ Mol Microbiol Biotechnol19991225727910943556
- HuangYLemieuxMJSongJAuerMWangDNStructure and mechanism of the glycerol-3-phosphate transporter from Escherichia coliScience2003301563361662012893936
- GuanLMirzaOVernerGIwataSKabackHRStructural determination of wild-type lactose permeaseProc Natl Acad Sci U S A200710439152941529817881559
- YinYHeXSzewczykPNguyenTChangGStructure of the multidrug transporter EmrD from Escherichia coliScience2006312577474174416675700
- HiraiTSubramaniamDStructure and transport mechanism of the bacterial oxalate transporter OxlTBiophys J20048753600360715339805
- WangYVenterHMaSEfflux pump inhibitors: a novel approach to combat efflux-mediated drug resistance in bacteriaCurrent Drug Targets2016171870271926424403
- NargotraASharmaSKoulJLQuantitative structure activity relationship (QSAR) of piperine analogs for bacterial NorA efflux pump inhibitorsEur J Med Chem200944104128413519523722
- FontaineFHequetAVoisin-ChiretASFirst identification of boronic species as novel potential inhibitors of the Staphylococcus aureus NorA efflux pumpJ Med Chem20145762536254824499135
- MichaletSCartierGDavidBN-caffeoylphenalkylamide derivatives as bacterial efflux pump inhibitorsBioorg Med Chem Lett20061761755175817275293
- SamosoronSBremnerJBBallALewisKSynthesis of functionalized 2-aryl-5-nitro-1H-indoles and their activity as bacterial NorA efflux pump inhibitorsBioorg Med Chem200514385786516203150
- FournierDChabertJMarquezBSynthesis and evaluation of new arylbenzo(b)thiophene and diarylthiophene derivatives as inhibitors of the NorA multidrug transporter of Staphylococcus aureusBioorg Med Chem200715134482449717498961
- Pereda-MirandaRKaatzGWGibbonsSPolyacylated oligosaccharides from medicinal Mexican morning glory species as antibacterials and inhibitors of multidrug resistance in Staphylococcus aureusJ Nat Prod200669340640916562846
- ThaiKMNgoTDPhanTVVirtual screening for novel Staphylococcus aureus NorA efflux pump inhibitors from natural productsMed Chem201511213515525181985
- SonnhammerELVon HeijneGKroghAA hidden Markov model for predicting transmembrane helices in protein sequencesProc Int Conf Intell Syst Mol Biol199861751829783223
- HofmannKStoffelWTMBASE: a database of membrane spanning protein segmentsBiol Chem1993374166170
- HirokawaTBoon-ChiengSMitakuSSOSUI: classification and secondary structure prediction system for membrane proteinsBioinformatics19981443783799632836
- CserzoMWallinESimonIVon HeijneGElofssonAPrediction of transmembrane alpha-helices in prokaryotic membrane proteins: the dense alignment surface methodProtein Eng19971066736769278280
- TusnadyGESimonIPrinciples governing amino acid composition of integral membrane proteins: Application to topology predictionJ Mol Biol199828324895069769220
- RostBSanderCCombining evolutionary information and neural networks to predict protein secondary structureProtein19941915572
- ClarosMGVon HeijneGTopPred II: an improved software for membrane protein structure predictionsComput Appl Biosci19941066856867704669
- ThompsonJDHigginsDGGibsonTJCLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choiceNucleic Acids Res19942222467346807984417
- SaliABlundellTLComparative protein modeling by satisfaction of spatial restraintsJ Mol Biol199323437798158254673
- KaleLSkeelRBhandarkarMNAMD: greater scalability for parallel molecular dynamicsJ Comput Phys1999151283312
- SchlickTSkeelRBrungerAKaleLBoardJAHermansJAlgorithmic challenges in computational molecular biophysicsJ Comput Phys1999151948
- JorgensenWLChandrasekharJMaduraJDImpeyRWKleinMLComparison of simple potential functions for simulating liquid waterJ Chem Phys198379926934
- GrubmullerHHellerHWindemuthASchultenKGeneralized Verlet algorithm for efficient molecular dynamics simulations with long-range interactionsMol Simul19916121142
- DardenTAPedersenLGMolecular modeling: an experimental toolEnviron Health Perspect199310154104128119250
- EssmannUBerkowitzMLDynamical properties of phospholipid bilayers from computer simulationBiophys J19997642081208910096903
- RyckaertJ-PCiccottiGBerendsenHJCNumerical integration of the Cartesian equations of motion of a system with constraints: molecular dynamics of n-alkanesJ Comp Phys1977233327341
- LaskoswkiRAMacArthurMWMossDSThorntonJMPROCHECK: a program to check the stereo chemical quality of protein structuresJ Appl Cryst199326283291
- EisenbergDLuthyRBowieJUVERIFY3D: assessment of protein models with three-dimensional profilesMethods Enzymol19972773964049379925
- ColovosCYeatesTOVerification of protein structures: Patterns of nonbonded atomic interactionsProtein Sci199329151115198401235
- HooftRWSanderCVriendGPositioning hydrogen atoms by optimizing hydrogen-bond networks in protein structuresProteins1996263633768990493
- GuexNPeitschMCSWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modelingElectrophoresis19971815271427239504803
- TrottOOlsonAJAutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreadingJ Comput Chem20101455461
- WolfLKPyRxC & EN20098731
- DeLanoWLThe PyMOL Molecular Graphics System DeLano Scientific version 1.7New York, NYSchrödinger, LLC
- KaliaNPMahajanPMehraRCapsaicin, a novel inhibitor of the NorA efflux pump, reduces the intracellular invasion of Staphylococcus aureusJ Antimicrob Chemother201267102401240822807321

