Abstract
Engineered nanoparticles (NPs) are widely used in many sectors, such as food, medicine, military, and sport, but their unique characteristics may cause deleterious health effects. Close attention is being paid to metal NP genotoxicity; however, NP genotoxic/carcinogenic effects and the underlying mechanisms remain to be elucidated. In this review, we address some metal and metal oxide NPs of interest and current genotoxicity tests in vitro and in vivo. Metal NPs can cause DNA damage such as chromosomal aberrations, DNA strand breaks, oxidative DNA damage, and mutations. We also discuss several parameters that may affect genotoxic response, including physicochemical properties, widely used assays/end point tests, and experimental conditions. Although potential biomarkers of nanogenotoxicity or carcinogenicity are suggested, inconsistent findings in the literature render results inconclusive due to a variety of factors. Advantages and limitations related to different methods for investigating genotoxicity are described, and future directions and recommendations for better understanding genotoxic potential are addressed.
Introduction
The rapidly growing nanotechnology industry will have significant economic and scientific impact in areas such as aerospace engineering, nanoelectronics, environmental remediation, and health care.Citation1 The design and development of novel engineered nanoparticles (NPs) are important to the industry due to beneficial physicochemical features that have led to over 800 NP-containing consumer products.Citation2 Hence, human exposure is high and continues to increase dramatically.
Due to their small size and great surface area coupled with physicochemical characteristics such as metal contaminations and charged surfaces, NPs may exhibit unpredictable genotoxic properties. Indirect DNA damage may be caused by induction of oxidative stress and inflammatory responses. Small NPs may cross cellular membranes and access the nucleus, where direct DNA interaction may result in damage. If NPs accumulate within a cell but do not readily gain access to the nucleus, direct DNA contact is possible during mitosis, when the nuclear membrane breaks down and gives rise to opportunity for DNA aberrations.
Occupational exposure of workers in the semiconductor, automotive, and aerospace industries is a primary concern, but NPs are also widely used in numerous cosmetics (such as lipstick, sunscreen, and antiaging creams), as well as medical sources (such as debris from dental prosthesis and orthopedic implants).Citation3–Citation5 There is promise that NPs could be administered as diagnostic aids, drug carriers, and therapeutic treatments for patients.Citation6,Citation7 With current and near-future exposure scenarios, workers in nanotechnology industries have the highest levels of chronic exposure to NPs, patients receiving NP-based treatments would have high-to-medium exposures over a limited duration, and consumers would likely have low, chronic exposures.
Among a variety of NPs, zinc oxide (ZnO) and silica NPs are in the most attractive positions for advanced nanotechnology industries and their potential applications, especially biomedical and pharmaceutical fields. They are also continuously directed for the advent of novel devices with multifunctionalities and multiple purposes, providing great benefits to human health. In particular, mesoporous ZnO and silica NPs have striking characteristics for application as drug carriers. They exhibit high surface area and porous interiors serving as reservoirs of drug molecules. The pore size and surrounding environment can influence the storage of various drugs of interest, whereas the size and shape of NPs can affect cellular uptake.Citation8 For example, mesoporous silica has been successfully utilized for delivery of ibuprofen into the pores via hydrogen bond interaction between ibuprofen and the silanol functional groups in the pore wall.Citation9 Moreover, highly mesoporous spherical three-dimensional ZnO nanoassemblies have been accomplished for loading doxorubicin hydrochloride as a model drug.Citation10 Both silica and ZnO NPs are generally regarded as essentially nontoxic and nonirritant beyond oral and topical pharmaceutical applications. In clinical use, individuals who may be more vulnerable to NP toxicity due to their pre-existing medical conditions thus require the examination of genotoxic potential and the underlying mechanism of action.
Key characteristics of nanoparticles
Due to large surface-area-to-volume ratio, NPs exhibit distinct physicochemical (eg, optical, magnetic, and electrical) and catalytic properties, rendering higher numbers of atoms binding on particle surfaces than their bulk counterparts.Citation11,Citation12 These characteristics of NPs promote their diffusion, reactivity, hardness, dimensionality, and suspension ability.
Usually, optical features of NPs are attributed to their ability to confine electrons to a very tiny size and to generate quantum effects. These optical absorption properties are related to their structure and shape. For instance, the yellow color of silver suspension in nanoform becomes a blue color in clustered form. Likewise, the color of gold NPs changes from blue to green to magenta, corresponding to their size and shape.
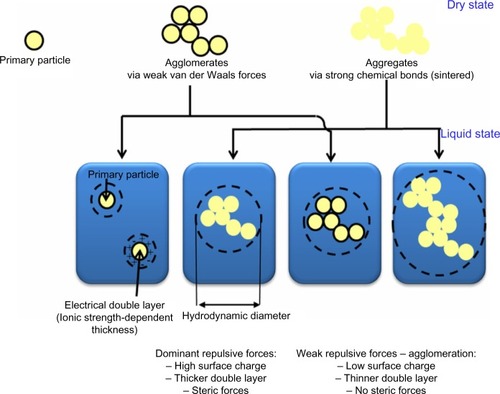
The suspension formation of NPs is also unique due to the great interaction force between their surface and suspension media, enabling density differences to be overcome.Citation13 In contrast, interactions of bulk material often result in either sinking or floating in liquid media. In aqueous media, NPs are dispersed due to electrostatic and steric repulsion of their surface charge (positive/negative).Citation14 Brownian motion and collision also have a crucial role in dispersion. As surface charges of NPs skew toward zero value, repulsive forces between NPs become decreased, eventually leading to their sedimentation by gravitational forces. The agglomeration process involves adhesion toward particles, mostly due to van der Waals forces resulting from their large surface-area-to-volume ratio at nanoscale ().Citation15 Due to agglomeration/aggregation, the physicochemical properties (eg, surface charge, size, size distribution, surface-area-to-volume ratio, surface reactivity) of NPs become altered, leading to mediation of their bioavailability and toxicities.Citation16,Citation17
Figure 1 Various states of nanoparticles in different forms of dry powder and liquid in suspension media.
In addition, diffusion of NPs is unique because it regulates their behavior in the surrounding environment. Indeed, particle diffusion coefficient is negatively proportional to the particle diameter. The smaller the particle size, the higher the diffusional forces, presenting the behavior tendency of gas or vapor.Citation18,Citation19 Thus, NPs with high diffusion coefficient display high mobility and consequently mix quickly in an aerosol. After their release in the environment, atmospheric diffusion rapidly promotes the migration of NPs, leading to them quickly traveling a long distance from the source and increasing detrimental health risks.Citation20
Additionally, other predominant properties of NPs are quantum confinement in semiconductors (eg, triple quantum dot silicon-based semiconductor, transition metal-doped ferromagnetic semiconducting silicon nanotubes, and ZnO semiconductor),Citation21–Citation23 surface plasmon resonance in particular metal NPs (eg, doped silicon nanocrystals, ZnO, and copper NPs),Citation24,Citation25 and superparamagnetism in magnetic materials (eg, multifunctional silica nanocomposites, gadolinium complexes, fluorophores, cell-penetrating peptides, and transition metal-doped ferromagnetic semiconducting silicon nanotubes).Citation22,Citation26,Citation27 For instance, ferroelectric materials (<10 nm in size) can switch their magnetization direction using room temperature thermal energy, rendering them inappropriate for memory storage. Copper NPs (>50 nm in size) are also regarded as superior durable materials that do not confer the same malleability and ductility as their bulk counterpart.Citation28
Regarding the unique features of NPs, their physicochemical properties should definitely be characterized prior to investigating the impact on human and environmental health. They include size, shape, structure, composition, purity, aggregation/agglomeration (size distribution), particle number, mass concentration, surface area, porosity, roughness, morphology, surface charge and chemistry, crystallinity, dispersity, and solubility. Measurement of primary particle size, hydrodynamic diameter, size distribution, zeta potential (surface charge), dispersity, concentration, and period of time in which agglomeration occurs provide better understanding of NP behavior relative to their cytotoxic and genotoxic responses.
Recent genotoxicological studies of metal oxide nanoparticles in in vitro and in vivo mammalian models
Zinc oxide nanoparticles
ZnO NPs are used in applications such as cosmetics, paints, drug carriers, and fillings in medical materials.Citation29 Also used as ultraviolet (UV) blocking materials, especially for UVA, their high catalytic activity in oxidation and photochemical reactions limits their use as UV blockers.Citation30 ZnO NPs are thought to be nontoxic and biocompatible.Citation31 Exposure to ZnO NPs has been associated with inflammatory responsesCitation32 and cytotoxicity.Citation33–Citation35 Little work regarding the genotoxic potential of ZnO NPs has been conducted. A previous investigation used Chinese hamster ovary cells to study chromosomal aberrations induced by ZnO NPs with a mean diameter of 100 nm promoted by UV light, finding increased clastogenicity under preirradiation and simultaneous irradiation conditions than in the dark.Citation29 This study indicates that ZnO NPs may cause photogenotoxicity, but a lack of information of the physicochemical properties makes the validity of the experiments questionable. Further assessment to fully investigate genotoxicity with a focus on size dependence and physiochemical features is required.
In vivo, the genotoxic potential of ZnO NPs has been investigated in animal models with systematic administration for 14 consecutive days,Citation36 as well as for 90 days.Citation37 The exposures increased liver enzyme and oxidative DNA breakage.
Recently, in vitro and in vivo, the mammalian toxicity of ZnO NPs as well as their toxicokinetics in various types of cells and animal models have been summarized ().Citation34,Citation35,Citation38–Citation41 In vitro, comet assays and the cytokinesis-blocked micronucleus present genotoxicity. Moreover, lung cells with in vitro exposure show cytotoxicity, increased oxidative stress, decreased mitochondrial membrane potential, and production of interleukin-8. Likely, the ZnO NPs are phagocytosed by macrophages and dissolved in lysosomes. In vivo, the ZnO NPs exhibit systemic distribution in target organs, including the liver, spleen, lung, kidney, and, in some cases, heart.
Table 1 Genotoxicity studies of ZnO and silica nanoparaticles using in vitro and in vivo mammalian models
The current review focuses on nanocosmetic ZnO sunscreens that have been thought not to be toxic, irritating, sensitizing, or photosensitizing after topical application.Citation42 The toxicity issue has also been examined by the European Commission, where sunscreen preparations containing ZnO NPs were reviewed.Citation42 Current studies have suggested that NPs do not exhibit increased penetration.Citation43,Citation44 However, populations with unhealthy skin or wounds still need to be cautious about long-term topical use of nanoscreens.
Silica nanoparticles
Silica induces inflammatory response via nuclear factor kappa B activation and oxidative stress responses both in vivo and in vitro,Citation32,Citation45,Citation46 but cytotoxicity is observed only at high concentrations.Citation47,Citation48 Silica NPs have been shown to enter the cell nucleus to potentially bind to the DNA phosphate backbone.Citation49 The silica NPs induce the reactive oxygen species (ROS),Citation46 especially the hydroxyl radical, a highly reactive molecule that may induce DNA strand breaks and oxidized bases.Citation50
Silica NPs have an impact on nuclear integrity, forming intranuclear protein aggregates and resulting in inhibition of replication, transcription, and cell proliferation.Citation49 Moreover, decreased replication activity as well as transcriptional activity were found for cells exposed to silica NPs. NPs of size >200 nm fail to penetrate the nucleus and do not alter nuclear structure and function or interfere with gene expression.Citation49 Nevertheless, there is limited evidence showing the genotoxic potential of silica NPs.Citation48,Citation51 A micronucleus assay report shows that these NPs do induce chromosomal damage.Citation52 To better indicate genotoxic potential, a battery of standardized tests quantifying different types of genetic aberrations are needed to cover all potential forms of inducible DNA damage as a result of exposure to NPs.
Recent work assessing systemic toxicological mechanisms of silica NPs in terms of cytotoxicity, genotoxicity, and phototoxicity has been reviewed and summarized ().Citation34,Citation35,Citation53–Citation57 Based on extensive physicochemical characterization, ecotoxicology, toxicology, safety, and epidemiology data, environmental and health risks seem to be unassociated with these particles if produced or utilized under current hygiene standards and recommendations. Silica NP interactions with membranes may induce the release of endosomal substance, ROS, cytokines, and chemokines, resulting in inflammatory responses.Citation58–Citation62 Silica NP toxicity is likely linked to mechanisms of interaction with outer and inner membranes, signaling responses, and vesicle trafficking pathways, but human health and environmental risks and the mechanisms of toxicity are not fully elucidated.
Taken together, genotoxicological investigations of ZnO and silica NPs have been carried out using in vitro and in vivo mammalian models, as summarized in .
Key mechanisms underlying nanoparticle-induced DNA damage
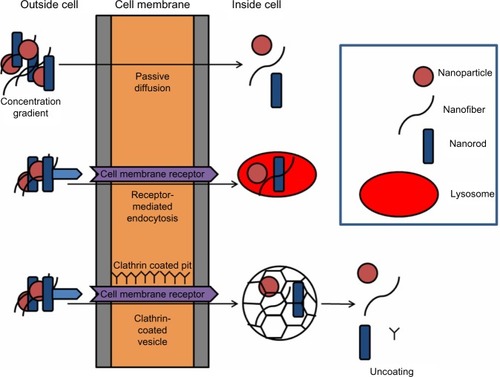
If NPs are able to enter the body through inhalation, dermal, or oral routes, direct and indirect mechanisms exist to stimulate DNA damage.Citation34,Citation35,Citation97 NPs may be able to penetrate into the cell, and subsequently the nucleus, through a number of routes ().Citation1 If NPs are located within the nucleus, direct interaction with DNA or DNA-associated proteins is possible. Indeed, silica NPs can enter the nucleus,Citation98,Citation99 inducing intranuclear protein aggregates and resulting in inhibition of replication, transcription, and cell proliferation.Citation49,Citation100,Citation101 Quantum dots have also been shown to penetrate the nucleus via the nuclear pore complexesCitation102 and interact with histone proteins.
Figure 2 Scheme illustrating possible routes of cellular uptake, including passive diffusion, receptor-related endocytosis, and clarthrin- or caveolae-dependent endocytosis. In brief, nanoparticles are in the correct size and shape. They may dock on membrane receptors, facilitating receptor-mediated endocytosis. Alternatively, clathrin- or caveolae-mediated endocytosis may occur, which results in the formation of pits in the region of 120 nm or up to 80 nm, respectively, which regulates the size of the material they are able to enclose.
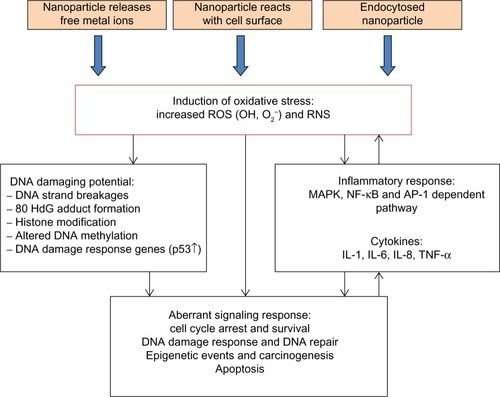
Genotoxicity may arise through indirect mechanisms where NPs do not physically interact with the DNA molecule but with other cellular components, such as those involved in the cell division process. Other cellular responses may be induced and give rise to genotoxicity, such as oxidative stress induction, inflammatory response, and aberrant signaling responses ().Citation1,Citation35,Citation97 Moreover, putative mechanisms underlying the detrimental effects of ZnO and silica NPs are proposed ().
Figure 3 Key indirect mechanisms underlying nanogenotoxicity. Nanoparticles (NPs) may cause oxidative stress induction, inflammatory responses, or aberrant cellular signaling. These responses may be implicated in cancer risk.
Abbreviations: NPs, nanoparticles; ROS, reactive oxygen species.
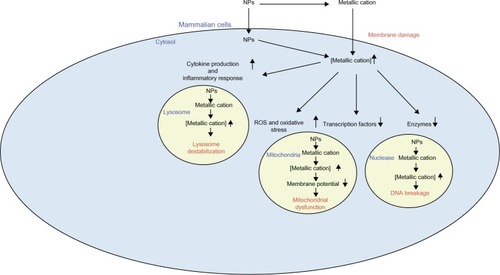
Figure 4 Putative mechanisms underlying the detrimental effects of zinc oxide and silica nanoparticles. These nanoparticles dissolve in the extracellular milieu, giving rise to increased extracellular metallic cations. This leads to increased intracellular respective metallic cations, resulting in decreased activity of particular enzymes and transcription factors. Moreover, this event can induce ROS generation and resulting oxidative stress, as well as stimulate various cytokine production and inflammatory responses. These phenomena, in turn, render membrane damage, DNA breakage, mitochondrial dysfunction, and lysosome destabilization.
Abbreviations: ROS, reactive oxygen species; RNS, reactive nitrogen species; IL, interleukin; TNF, tumor necrosis factor; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; MAPK, mitogen-activated protein kinases; AP-1, activator protein 1.
Oxidative stress
Oxidative stress is a redox imbalance within cells as a consequence of increased intracellular ROS and decreased antioxidants. ROS-induced DNA damage is categorized by single- and double-stranded DNA breaks, base modifications (eg, formation of 8-hydroxydeoxyguanosine adducts), and DNA crosslinks, all of which may be implicated to initiate and promote carcinogenesis if unrepaired.Citation103,Citation104
The transition metals ions (such as iron [Fe] and zinc) released from certain NPs are capable of converting cellular oxygen metabolic products (such as H2O2 and superoxide anions) to hydroxyl radicals and to DNA damaging species. Fe(II) can cause the production of H2O2 from molecular O2, which can diffuse through the cellular and nuclear membranes to react with DNA-bound Fe and lead to radical production, crosslinking thymine–tyrosine (DNA-histone protein) chromatin.Citation50 Free Fe ions can cause OH-mediated purine and pyrimidine modifications.Citation105 As a result, Fe-containing NPs are a concern as a surplus source of Fe within the cells and fuelling the generation of highly reactive hydroxyl radicals via the Fenton reaction.
In addition to the elemental and ionic composition of NPs, the inherent high surface area can enhance the production of ROS. The smaller the NP, the higher the oxidative stress produced.Citation106–Citation110 Researchers have reported that silica NP toxicity is dependent on size and may be associated with ROS generation.Citation54,Citation61,Citation111
Oxidative DNA damage, as determined by the comet assay and micronucleus assay,Citation108,Citation112,Citation113 has been demonstrated by several studies of the genotoxic effects of NPs. Oxidative stress activates specific signaling pathways, including mitogen-activated protein kinase and nuclear factor kappa B,Citation114 together with interference of antioxidant defenses, resulting in release of proinflammatory cytokines.Citation115 This signaling cascade is a key trigger of inflammation, a defensive reaction that leads to further ROS release from inflammatory cells (eg, neutrophils).Citation116,Citation117
Inflammation
Inflammation is an important physiological process in response to tissue injury mediated by inflammatory cells secreting cytokines (eg, interleukins and tumor necrosis factor protein families), migration inhibition factors, RNS (reactive nitrogen species), and ROS. These factors are involved in protective defense against infection and/or tissue injury. They promote DNA damage in the form of point mutations, DNA adducts, and chromosomal fragmentation, as well as inhibit DNA repair and induce aberrant methylation patterns.Citation60,Citation61 Expectedly, chronic inflammation has been associated with carcinogenesis.Citation118,Citation119
At present, a number of studies have shown that NPs can exhibit inflammatory responses. Their small size and great surface area are involved in facilitating inflammation, as previous studies demonstrate that ultrafine NPs display higher inflammatory potency in the lungs of rats following intratracheal instillation.Citation120,Citation121 The composition of the particle may be a determinant factor affecting the extent of the inflammatory response induced. For instance, in vitro, induced inflammation seems to follow the trend of silica and ZnO NPs.Citation111,Citation122,Citation123 Some NPs have oxidative DNA damaging potential via excessive formation of ROS and the release of metal ions, but also as a consequence of chronic inflammatory responses.
DNA damage responsive signaling
Exposure to NPs is associated with induction of oxidative stress, leading to damage to cellular components, most importantly DNA. Consequently, this damage can affect several cellular responses, including cell cycle arrest, apoptosis, and DNA repair. The DNA repair system is responsible for genetic stability and cell survival, and if repair fails to occur during or before replication of damaged DNA, mutagenic and possibly carcinogenic incidences may occur.
Once DNA is damaged, a key effector molecule, p53, is activated. Tumor suppressor gene p53 has been recognized as “the guardian of the genome” because of its essential role in arresting the cell cycle, activating transcription of genes that mediate DNA repair, and preventing the incidence of mutagenic conversion.Citation124 If DNA damage is extensively accumulated, p53 triggers apoptosis to eliminate the individual cell for the benefit of the organism. When these protective factors are compromised, stable heritable changes may undertake cellular transformation and, ultimately, carcinogenesis.
The pro-oxidative and proinflammatory properties have been found after exposure to various metal oxide NPs. ROS generation by ZnO NPs stimulates cellular processes: specifically, oxidant injury, inflammatory response, and cell death in different cell types such as mouse macrophages and human bronchial epithelial cells.Citation77 Moreover, ZnO NPs are thought to be linked to the incidence of metal fume fever. A previous study has indicated that pro-oxidant activity of ZnO NPs is attributable to particle dissolution.Citation125 Prevention of ZnO NP dissolution via Fe doping could decrease the pro-oxidative and proinflammatory effects of these particles.Citation125 Other NPs (silica, cationic polystyrene, and C60 fullerene) have been reported to exhibit pro-oxidative and proinflammatory properties in vitro and in vivo, including extensive accumulation of ROS, induction of oxidative stress, and stimulation of antioxidant and signaling pathways.Citation126–Citation128
Indeed, several reports have demonstrated that exposure to asbestos fibers in unregulated workplaces risks pleural and lung fibrosis (asbestosis), lung cancer, and pleural and peritoneal malignant mesothelioma.Citation129–Citation131 Asbestos may be regarded as a tumor promoter or cocarcinogen in the induction of lung cancers, particularly representing synergistic effects with chemical carcinogens in cigarette smoke.Citation132 Asbestos fibers are naturally occurring in rocks and soils and comprise six different types: amphibole types (crocidolite, amosite, anthophyllite, tremolite, and actinolite), which are rod-shaped and have higher durability relative to the only serpentine type (chrysotile).Citation133 Much evidence has been presented that exposure to asbestos gives rise to a spectrum of asbestos-related diseases, including malignant pleural mesothelioma. This suggests an obvious relationship between the specific NP and disease, probably due to ROS accumulation and resulting oxidative stress.Citation134–Citation137 However, there is no currently published report addressing a definite correlation between a disease outcome and exposure to a specific type of newly developed NP in humans. An active approach should be taken as a precaution. One effective strategy is to identify biomarkers associated with NP exposure.Citation138 Development of a panel of biomarkers as indicators of exposure-specific disease outcomes will require further time but would be well worth the effort for the identification of early biological responses related to current knowledge-based injury pathways.
Previous reports of metal fume fever, a flu-like illness with characteristics of self-limiting inflammation and oxidative stress response in the lung, indicate that it is caused by inhalation of highly concentrated metal oxide particles, particularly ZnO.Citation139–Citation143 Given the increase in application of NPs and the uncertainty of their potential health impacts, health surveillance of workers frequently exposed to NPs in the occupational setting is important.
Using liquid chromatography–tandem mass spectrometry analysis, more than 30 proteins were thought to be responsible for incidental NP-induced oxidative stress. Some of these proteins may serve as markers for exposure to pro-oxidative substances.Citation144,Citation145 Alterations of other particle-induced proteomes include modification of nitrotyrosine-based protein, activation of unfolding protein response, and incremental expression of ATF4, an endoplasmic reticulum stress-related transcription factor.Citation144,Citation146 In animal studies, oxidative stress-altered proteomic profiles were found in the bronchoalveolar lavage fluid and lung tissue in mouse asthma models.Citation147,Citation148 A more recent study demonstrates the expression of polymeric immunoglobulin receptor, complement C3, neutrophil gelatinase-associated lipocalin, chitinase 3-like protein 3, chitinase 3-like protein 4, and acidic mammalian chitinase in the lung to be associated with the adjuvant effect of ultrafine particles on the oxidant activity and the primary immune response (allergic sensitization).Citation148,Citation149 Increased chitinase 3-like protein 3 expression is associated with the boosting of ambient ultrafine particles on the secondary immune response of inhalation exposure.Citation150 Furthermore, alteration of oxidative stress-associated proteome was observed in the bronchoalveolar lavage fluid from C57BL/6 mice exposed to ZnO NP via pharyngeal aspiration, indicating that proteomics may be used to identify biomarkers related to the exposure of certain NPs. Because oxidative stress and inflammatory response are also responsible for the toxicity of various NPs, the technology of proteomics has the potential to identify the biomarkers associated with NP exposure and the resulting deleterious effects of injury pathways.
The ideal biomarkers for evaluating environmental and occupational exposures should provide a mechanistic, molecular, and biological basis for the diseases and be exposure specific to reflect early adverse health effects, have clinical relevance, and be easy to use. Although identification of biomarkers that meet all these criteria is a challenge, it is feasible to study NP exposure-associated early biological events such as oxidative stress and inflammation.
Artifacts and limitations influencing nanogenotoxicity studies
Although little is known about the toxicokinetics of NPs, key factors gaining attention are the physicochemical properties influencing cellular uptake and subsequent physiological consequences. Parameters involved in genotoxicological responses are uncertain, and the evidence points to different factors participating in modulating molecular interactions.
Size, shape, and surface area
The nanometer size (<100 nm) of particles is considered a primary feature representing unique properties over bulk counterparts. Decreased size increases the number of particles per unit mass, but small size can also demonstrate a health hazard due to their interference with biological components once internalized. As a consequence, the size influences absorption, distribution, metabolism, and excretion kinetics, the driving force behind the development of new nanomedicines and nanodevices for clinical health care.Citation6,Citation7 With regard to cellular uptake, size is a key factor in the different internalization mechanisms. Perhaps the most prominent mechanism is diffusion across the plasma membrane (either directly across the membrane or through membrane channels 10–30 nm wide), endocytosis, or energy-dependent mechanisms via a number of different routes ().
In addition to cellular uptake, size does influence toxicological outcome, as reported by many studies focusing on ZnO and silica NPs versus microparticles. For instance, inhalation studies show that NPs penetrate deeper into the lungs and become localized within various cell types, indicating a greater inflammatory response that is markedly associated with potential toxicity in comparison with their fine-sized counterparts.Citation98,Citation151–Citation154 Although genotoxic potential was not examined in these investigations, the established relationship between chronic inflammation, DNA damage, and carcinogenesis may provide insight into the adverse health effects of long-term NP inhalation.
Shape or morphology is another matter related to negative cellular effects of NPs. Fewer studies focusing on the toxicological relationship associated with this parameter have been established, though a previous publication has demonstrated that removal of structural defects from a particular NP was sufficient to substantially reduce inflammatory response and overall toxicity.Citation155 Previous studies have revealed that shape of NPs strongly governs uptake rate. Spherical NPs exhibit higher uptake than nanorods, whereas internalization of cylindrical materials is strongly influenced by a high aspect ratio.Citation156,Citation157
When the number of particles per mass unit increase, the overall surface area will also increase. The shape of NPs contributes to the overall surface area, such that spherical NPs have slightly smaller surface areas than an octagonal structure of the same size. This greater surface area promotes catalytic activity of the material, allowing an increase in its reactivity due to unsatisfied high energy bonds of surface atoms.Citation120 If NPs are able to gain access to the cellular milieu, the large surface area will give rise to more reactivity with biological components, resulting in unwanted cellular damage and oxidative stress.
Purity
Purity of NPs is a concern, as contamination of residual metal may cause stronger (geno) toxicological responses than the actual nanomaterial. Most metal catalysts are removed by postproduction processes. Purified NPs may contain up to 15% metal residual bymass. Efforts to purify NPs are under way to limit effects of impurities on toxicity.
Numerous studies undertake the synthesis of lead-rich carbon nanotubes for use as X-ray protection shields. However, Fe is one of the primary sources of damage via oxidative stress, resulting in Fenton or Haber–Weiss reactions.Citation158–Citation161 Indeed, Fe contaminants on carbon nanotubes have reportedly been shown to cause a substantial loss of glutathione and increased lipid peroxidation in alveolar macrophages, indicators of oxidative stress.Citation162 Conversely, a previous study found that single-walled carbon nanotubes induced dose-dependent lung lesions (granulomas) in mice, irregardless of purity.Citation163 Similarly, nickel and yttrium catalyst impurities entrapped within single-walled carbon nanotubes and multiwalled carbon nanotubes do not seem to be responsible for potential toxicity associated with these materials.Citation164 In general, in vitro studies utilizing metal chelators could provide more insight into the role of such impurities. However, the conflicting information may be attributed to the other physicochemical characteristics of NP used in the toxicological studies, and thus emphasizes the importance of full characterization and standardization of NPs.
Agglomeration (size distribution)
An inherent feature of several NPs is their hydrophobicity and tendency to agglomerate, especially under physiological conditions. Upon exposure to biological systems, most NPs will form aggregates rather than remain monodisperse. In genotoxicity testing, in vivo or in vitro dosing in an aqueous carrier or into an aqueous environment (with the exception of dust inhalation studies) is usually established, and exposure responses relate to the degree of agglomerated NP form.Citation165
Although NPs have a tendency to form larger aggregates, fibrous NPs represent a more complex situation. In addition to aggregation, the fibers may form a tangled structure depending upon rigidity, leading to a change in the dimensions and surface area of the original structure. Although fiber rigidity is dependent on the synthesis method, rigid NPs are attracted to one another by van der Waals forces, with a tendency to curve and twist, forming bundles.Citation166,Citation167 Agglomerates are larger and often more rigid than individual NPs, leading to new causes of toxicity.Citation168
Different approaches are being undertaken to improve the hydrophilicity of NPs. Concerning dissolution of NPs in suspension media, aggregation/agglomeration is still an obstacle due to van der Waals forces and resulting adhesion toward particles at nanoscale. Hence, efforts on dispersal methods are established to improve their solubilities by using various dispersing agents of both inorganic and organic stabilizers (eg, fetal serum, organosulfur compounds, polyethyleneglycol, dextran, liposomes, micelles) or chemically modified functionalization of particle surface (eg, polymeric macromolecules).Citation100,Citation169–Citation171
Indeed, the aggregate states can be overcome by use of organic small molecules harboring multiple functional groups such as carboxyl (COOH), amine (NH2), thiol (SH), phosphate, and sulfates. These stabilizers can be tailored for dispersibility into aqueous media or other biocompatible fluids.Citation172,Citation173 The organosulfur compound 2,3-meso dimercaptosuccinic acid (DMSA) containing two carboxylic and two thiol groups has been widely applied as a dispersing stabilizer. Magnetic NPs have been stabilized with DMSA for tissue- and cell-targeted delivery of therapeutic drugs in the lung.Citation174 In particular, the mechanism of proinflammatory effects of magnetic NPs and DMSA has been examined. Also, the postfunctionalization of NPs using methoxy polyethyleneglycol (PEG) 2000 silane has been successfully developed for stabilizing free thiols onto the surface of metal oxide NPs under physiological pH.Citation175
Moreover, various polymer molecules have been employed for steric stabilization of oxide NPs in aqueous and high ionic strength media.Citation176–Citation178 These polymeric stabilizers can affect the performance of nanomaterials, depending on the chemical nature of the polymer (ie, hydrophilicity/hydrophobicity, biocompatibility, and biodegradation), the molecular weight of the polymer, the conformation of the polymer, and the degree of particle surface. Other stabilizers, such as the amphiphilic molecules (eg, liposomes and micelles), have been successfully utilized to improve hydrophilicity of oxide NPs in biological or physiological media.Citation179,Citation180
The features of solubility or dispersity usually influence cytotoxic and genotoxic impact; however, these issues should be treated with caution. Previous reports have demonstrated that surface modification-solubilized NPs can alleviate toxicity as a result of the functionalization.Citation181,Citation182 Conversely, several studies have shown the surfactant-stabilized NPs to be more cytotoxic, and coating NPs with gold and silver apparently augments cytotoxicity in comparison with nondispersed or noncoated counterparts.Citation183
Extensive effort is necessary to refine the interplay between agglomeration, dispersal methods, and negative cellular effects. The degree of agglomeration under experimental conditions when conducting risk assessment may provide clearer interpretation of results.Citation184,Citation185 Given that agglomerated structure may not be in the nanoscale, risks regarding exposure may be substantially decreased due to reduced cellular uptake or an inability to cross biological barriers. However, overlooking the agglomeration-related issues in many studies may be a primary cause for the lack of consistency and often conflicting reports.
Surface charge and chemistry
An understanding of surface properties of NPs is essential for providing insight into their behavior under different experimental conditions. Surface charge and chemistry will influence the production of agglomerates and resulting toxicities according to factors such as the pH or ionic strength in the aqueous environment.Citation186 Thus, aggregation/disaggregation kinetics may be useful to decipher toxicities that may occur during the course of an in vitro experiment or according to the specific biological compartment where the NPs may become concentrated.
Surface charge has an important role in regulating cellular uptake of NPs. The plasma membrane is negatively charged (due to the phospholipids on the outer surface), as well as the intracellular environment. Thus, anionic NPs may be endocytosed at a lower rate compared with cationic NPs. Although this has been observed in practice using PEGylated polylactide and hydrogel NPs of similar sizes but varying charges,Citation157,Citation187 this effect does not preclude the uptake of negatively charged NPs.Citation188 However, cationic surface charges show greater cytotoxic responses as compared with those with anionic charges, although it is uncertain whether cell death is a direct consequence of surface charge or a result of increased uptake often associated with cationic NPs.Citation189 Furthermore, DNA is negatively charged. Thus, cationic NPs appear to interact more significantly with genetic material.
In addition to functionalizing the surface to promote their solubility, surface chemistry may be modified to attach biological components such as peptides for cell targeting or pharmaceuticals for drug delivery. These modifications will have toxicological impact toward the resultant NPs because cytotoxicity is strongly associated with coating the functional group.Citation190 Information on surface charge and chemistries is significant in elucidating uptake mechanisms and predicting biological interactions for the evaluation of toxicity.
Concluding remarks for nano(geno) toxicological studies
Currently, inadequate information on the genotoxic potential of NPs and the impact on persistent exposure to human health is a concern. Numerous literature reports focus on the cytotoxicity of NPs, specific aspects of physicochemical characteristics, association with other potential toxic effects, as well as consideration of DNA damage and cellular uptake, bioaccumulation, distribution, and retention. A growing body of studies point to particular NPs eliciting DNA damage potential, but inconclusive reports necessitate a discussion within the scientific community for clearer and more informative reports. Previous reviews have addressed potential considerations, including material characterization, use of standardized experimental methods, and association between in vitro and in vivo results,Citation191,Citation192 but should account for the following.
In toxicological studies of any NPs, detailed physicochemical characterization under real experimental conditions is preferential. Measurements should include size distribution, morphology, surface area, charge, surface modifications, chemical composition, crystallinity, and agglomeration. Moreover, information on the fabrication process should be provided.
Appropriate controls (positives and negatives) and standards need to be established. Sources of metal ions or the use of metal chelators should be addressed in experimental design to elucidate whether biological effects are the consequence of NP interactions, impurities, or degradation products released during exposure.
If the functionalized form of NPs is being assessed, the unfunctionalized form should also be included to directly investigate the effect of the surface modification.
NP dynamics during the period of genotoxicity assay need to be a concern: eg, what about the degree of agglomeration and size distribution? In vitro, do NPs sediment or remain in suspension? Metrological techniques required to provide clearer information are currently not available and need to be developed.
Once internalized, the long-term fate of NPs needs to be considered. Physicochemical properties before and after experimentation should be detailed but may require the development of novel methodologies to form answers.
A battery of genotoxicity tests with varying end points should be utilized to provide insight into the mechanism of action and to ensure a comprehensive view of the reactivity of NPs.
In addition to somatic cells, the genotoxic potential of NPs on germ cells should be conducted.
Extension of in vitro experiments over 24 hours might be necessary if longer treatment times are more informative on the genotoxic potential of NPs.
Well-designed experiments are needed to enable a concerted effort to better exploit NP-mediated hazards and to define similarities enabling further extrapolations. The underlying mechanisms responsible for the exposure effect observed is also important in effectively informing the safer design of future NP systems, ensuring biocompatibility with minimum deleterious health risks. Biomarker studies provide valuable information identifying early biological events associated with adverse health effects of engineered nanomaterials before the manifestation of clinical outcomes, potentially helping health surveillance of workers at higher risk due to their occupational settings.
Disclosure
The authors report no conflicts of interest in this work.
Acknowledgments
This research was supported by a grant (10182MFDS991) from the Ministry of Food and Drug Safety in 2010–13 and ‘The Ecoinnovation Project’ (412-112-110) by the Korean Ministry of Environment. This research project is also supported by a grant from Mahidol University, Thailand, and a grant from the Faculty of Environment and Resource Studies, Mahidol University, Thailand.
References
- SinghNManshianBJenkinsGJNanoGenotoxicology: the DNA damaging potential of engineered nanomaterialsBiomaterials20093023–243891391419427031
- Lux report. Nanomaterials state of the market: stealth success, broad impact Available from: http://portal.luxresearchinc.com/research/document/3735Accessed January 17, 2008
- BrownCFisherJInghamEBiological effects of clinically relevant wear particles from metal-on-metal hip prosthesesProc Inst Mech Eng H2006220235536916669401
- BallestriMBaraldiAGattiAMLiver and kidney foreign bodies granulomatosis in a patient with malocclusion, bruxism, and worn dental prosthesesGastroenterology200112151234123811677217
- HartAJHesterTSinclairKThe association between metal ions from hip resurfacing and reduced T-cell countsJ Bone Joint Surg Br200688444945416567777
- FerrariMCancer nanotechnology: opportunities and challengesNat Rev Cancer20055316117115738981
- SahooSKParveenSPandaJJThe present and future of nanotechnology in human health careNanomedicine200731203117379166
- DuXHeJElaborate control over the morphology and structure of mercapto-functionalized mesoporous silicas as multipurpose carriersDalton Trans201039389063907220730188
- MaS WYZhuYA simple room temperature synthesis of mesoporous silica nanoparticles for drug storage and pressure pulsed deliveryJournal of Porous Material2011182233239
- BarickKCNigamSBahadurDNanoscale assembly of mesoporous ZnO: a potential drug carrierJ Mater Chem20102064466452
- BormPJABerubeDA tale of opportunities, uncertainties and risksNano Today200835659
- DusinskaMFjellsboLMagdolenovaZTesting strategies for the safety of nanoparticles used in medical applicationsNanomedicine (Lond)20094660560719663587
- KumarAPandeyAShankerRDhawanAMicroorganism: a versatile model for toxicity assessment of engineered nanomaterialsCioffiNRaiMNano-Antimicrobials: Progress and ProspectsBerlin, GermanySpringer Verlag GmbH2012
- MaynardADNanotechnology: the next big thing, or much ado about nothing?Ann Occup Hyg20075111217041243
- ElsaesserAHowardCVToxicology of nanoparticlesAdv Drug Deliv Rev201264212913721925220
- NavarroEBaunABehraREnvironmental behavior and ecotoxicity of engineered nanoparticles to algae, plants, and fungiEcotoxicology200817537238618461442
- KumarAPandeyAKSinghSSShankerRDhawanAA flow cytometric method to assess nanoparticle uptake in bacteriaCytometry A201179970771221638764
- AitkenRJKoopmanPLewisSESeeds of concernNature20044327013485215525979
- AitkenRJ HSTranCLDonaldsonKMultidisciplinary approach to the identification of reference materials for engineered nanoparticle toxicologyNanotoxicology200827178
- FeliuNFadeelBNanotoxicology: no small matterNanoscale20102122514252020877789
- PanHHouseMGHaoXJiangHWFabrication and characterization of a silicon metal-oxide-semiconductor based triple quantum dotAppl Phys Lett201210026310915
- ShpaismanNGivanUKwiatMPevznerAElnathanRPatolskyFControlled synthesis of ferromagnetic semiconducting silicon nanotubesJ Phys Chem C20121161480008007
- SachetELosegoMDGuskeJFranzenSMariaJPMid-infrared surface plasmon resonance in zinc oxide semiconductor thin filmsAppl Phys Lett20131025051111051114
- RoweDJJeongJSMkhoyanKAKortshagenURPhosphorus-doped silicon nanocrystals exhibiting mid-infrared localized surface plasmon resonanceNano Lett20131331317132223413833
- ZhouWJHalpernARSeefeldTHCornRMNear infrared surface plasmon resonance phase imaging and nanoparticle-enhanced surface plasmon resonance phase imaging for ultrasensitive protein and DNA biosensing with oligonucleotide and aptamer microarraysAnal Chem201284144044522126812
- JoshiRFeldmannVKoestnerWMultifunctional silica nanoparticles for optical and magnetic resonance imagingBiological Chemistry2013394112513523096570
- OrlovAFAgagonovYBalagurovLABublikVTStudy of structural characteristics of ferromagnetic silicon implanted with manganeseCrystallography Reports2008535796799
- Science Daily. Nanoparticle Available from: http://www.sciencedaily.com/articles/n/nanoparticle.htmAccessed February 17, 2013
- DufourEKKumaravelTNohynekGJKirklandDToutainHClastogenicity, photo-clastogenicity or pseudo-photo-clastogenicity: genotoxic effects of zinc oxide in the dark, in pre-irradiated or simultaneously irradiated Chinese hamster ovary cellsMutat Res2006607221522416797222
- Yuan FangliHPChunleiYinShulanHuangJinlinLiPreparation and properties of zinc oxide nanoparticles coated with zinc aluminateJ Mater Chem200313634637
- Qi QiTZYuQingjiangWangRuiZengYiLiuLiYangHaibinProperties of humidity sensing ZnO nanorods-base sensor fabricated by screen-printingSens Actuators B Chem2008133638643
- SayesCMReedKLWarheitDBAssessing toxicity of fine and nanoparticles: comparing in vitro measurements to in vivo pulmonary toxicity profilesToxicol Sci200797116318017301066
- JengHASwansonJToxicity of metal oxide nanoparticles in mammalian cellsJ Environ Sci Health A Tox Hazard Subst Environ Eng200641122699271117114101
- KumarADhawanAGenotoxic and carcinogenic potential of engineered nanoparticles: an updateArch Toxicol201387111883190024068037
- MankeAWangLRojanasakulYMechanisms of nanoparticle-induced oxidative stress and toxicityBiomed Res Int2013201394291624027766
- SharmaVSinghPPandeyAKDhawanAInduction of oxidative stress, DNA damage and apoptosis in mouse liver after sub-acute oral exposure to zinc oxide nanoparticlesMutat Res20127451–2849122198329
- SurekhaPKishoreASSrinivasARepeated dose dermal toxicity study of nano zinc oxide with Sprague-Dawley ratsCutan Ocul Toxicol2012311263221830917
- RobJVandebrielWimDe JongHA review of mammalian toxicity of ZnO nanoparticlesNanotechnol Sci Appl20125617124198497
- SharmaVShuklaRKSaxenaNParmarDDasMDhawanADNA damaging potential of zinc oxide nanoparticles in human epidermal cellsToxicol Lett2009185321121819382294
- JangYeon SueLeeEun YoungParkYoon-HeeThe potential for skin irritation, phototoxicity, and sensitization of ZnO nanoparticlesMol Cell Toxicol201282171177
- LeeSeung HoPieJae EunKimYu RiLeeHee RaSonSang WookKimMeyoung-KonEffects of zinc oxide nanoparticles on gene expression profile in human keratinocytesMol Cell Toxicol201282113118
- SCCNFPThe scientific committee on cosmetic products and non-food products intended for consumers concerning zinc oxide2003 Available from: http://ec.europa.eu/health/ph_risk/committees/sccp/documents/out222_en.pdfAccessed April 15, 2014
- WangSQBalagulaYOsterwalderUPhotoprotection: a review of the current and future technologiesDermatol Ther2010231314720136907
- TranDTSalmonRPotential photocarcinogenic effects of nanoparticle sunscreensAustralas J Dermatol20115211621332685
- KaewamatawongTShimadaAOkajimaMAcute and subacute pulmonary toxicity of low dose of ultrafine colloidal silica particles in mice after intratracheal instillationToxicol Pathol200634795896517178696
- LinWHuangYWZhouXDMaYIn vitro toxicity of silica nanoparticles in human lung cancer cellsToxicol Appl Pharmacol2006217325225917112558
- ChangJSChangKLHwangDFKongZLIn vitro cytotoxicitiy of silica nanoparticles at high concentrations strongly depends on the metabolic activity type of the cell lineEnviron Sci Technol20074162064206817410806
- JinYKannanSWuMZhaoJXToxicity of luminescent silica nanoparticles to living cellsChem Res Toxicol20072081126113317630705
- ChenMvon MikeczAFormation of nucleoplasmic protein aggregates impairs nuclear function in response to SiO2 nanoparticlesExp Cell Res20053051516215777787
- ValkoMRhodesCJMoncolJIzakovicMMazurMFree radicals, metals and antioxidants in oxidative stress-induced cancerChem Biol Interact2006160114016430879
- BarnesCAElsaesserAArkuszJReproducible comet assay of amorphous silica nanoparticles detects no genotoxicityNano Lett2008893069307418698730
- WangJJSandersonBJWangHCytotoxicity and genotoxicity of ultrafine crystalline SiO2 particulate in cultured human lymphoblastoid cellsEnviron Mol Mutagen200748215115717285640
- Fruijtier-PollothCThe toxicological mode of action and the safety of synthetic amorphous silica-a nanostructured materialToxicology20122942–3617922349641
- ParkYHBaeCHJangYEffect of the size and surface charge of silica nanoparticles on cutaneous toxicityMol Cell Toxicol2013916774
- MusaMKannanTPMasudiSMAb RahmanIAssessment of DNA damage caused by locally produced hydroxyapatite-silica nanocom-posite using comet assay on human lung fibroblast cell lineMol Cell Toxicol2012815360
- ChoiJEParkY-HYoung LeeEA safety assessment of phototoxicity and sensitization of SiO2 nanoparticlesMol Cell Toxicol201172171176
- KimY-JIk YangSNeurotoxic effects by silica TM nanoparticle is independent of differentiation of SH-SY5Y cellsMol Cell Toxicol201174381388
- YoshidaTYoshiokaYMatsuyamaKSurface modification of amorphous nanosilica particles suppresses nanosilica-induced cytotoxicity, ROS generation, and DNA damage in various mammalian cellsBiochem Biophys Res Commun2012427474875223044420
- WangFJiaoCLiuJYuanHLanMGaoFOxidative mechanisms contribute to nanosize silican dioxide-induced developmental neurotoxicity in PC12 cellsToxicol In Vitro20112581548155621635944
- SergentJAPagetVChevillardSToxicity and genotoxicity of nano-SiO2 on human epithelial intestinal HT-29 cell lineAnn Occup Hyg201256562263022378843
- PassagneIMorilleMRoussetMPujalteIL’AzouBImplication of oxidative stress in size-dependent toxicity of silica nanoparticles in kidney cellsToxicology20122992–311212422627296
- NabeshiHYoshikawaTMatsuyamaKAmorphous nanosilica induce endocytosis-dependent ROS generation and DNA damage in human keratinocytesPart Fibre Toxicol20118121235812
- LandsiedelRMa-HockLVan RavenzwaayBGene toxicity studies on titanium dioxide and zinc oxide nanomaterials used for UV- protection in cosmetic formulationsNanotoxicology2010436438120925445
- LiCHShenCCChengYWOrgan biodistribution, clearance, and genotoxicity of orally administered zinc oxide nanoparticles in miceNanotoxicology20126774675621950449
- Monteiro-RiviereNAWienchKLandsiedelRSchulteSInmanAORiviereJESafety evaluation of sunscreen formulations containing titanium dioxide and zinc oxide nanoparticles in UVB sunburned skin: an in vitro and in vivo studyToxicol Sci2011123126428021642632
- GopalanROsmanIAmaniAMatasMAndersonDThe effect of zinc oxide and titanium dioxide nanoparticles in the comet assay with UVA photoactivation of human sperm and lymphocytesNanotoxicology2009313339
- GerloffKAlbrechtCBootsAWFörsterISchinsRPFCytotoxicity and oxidative DNA damage by nanoparticles in human intestinal Caco-2 cellsNanotoxicology200934355364
- YangHLiuCYangDZhangHXiZComparative study of cytotoxicity, oxidative stress and genotoxicity induced by four typical nanomaterials: the role of particle size, shape and compositionJ Appl Toxicol2009291697818756589
- YoshidaRKitamuraDMaenosonoSMutagenicity of water-soluble ZnO nanoparticles in Ames testJ Toxicol Sci200934111912219182441
- OsmanIFBaumgartnerACemeliEFletcherJNAndersonDGenotoxicity and cytotoxicity of zinc oxide and titanium dioxide in HEp-2 cellsNanomedicine (Lond)2010581193120321039197
- YinHCaseyPSMcCallMJFenechMEffects of surface chemistry on cytotoxicity, genotoxicity, and the generation of reactive oxygen species induced by ZnO nanoparticlesLangmuir20102619153991540820809599
- SharmaVAndersonDDhawanAZinc oxide nanoparticles induce oxidative stress and genotoxicity in human liver cells (HepG2)J Biomed Nanotechnol201171989921485822
- SharmaVSinghSKAndersonDTobinDJDhawanAZinc oxide nanoparticle induced genotoxicity in primary human epidermal keratinocytesJ Nanosci Nanotechnol20111153782378821780369
- HackenbergSZimmermannFZScherzedARepetitive exposure to zinc oxide nanoparticles induces dna damage in human nasal mucosa mini organ culturesEnviron Mol Mutagen201152758258921786336
- YinHCaseyPSMcCallMJSurface modifications of ZnO nanoparticles and their cytotoxicityJ Nanosci Nanotechnol201010117565757021137983
- ZhangXQYinLHTangMPuYPZnO, TiO(2), SiO(2,) and Al(2) O(3) nanoparticles-induced toxic effects on human fetal lung fibroblastsBiomed Environ Sci201124666166922365403
- XiaTKovochichMLiongMComparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress propertiesACS Nano20082102121213419206459
- De BerardisBCivitelliGCondelloMExposure to ZnO nanoparticles induces oxidative stress and cytotoxicity in human colon carcinoma cellsToxicol Appl Pharmacol Epub April 29, 2010
- SharmaVAndersonDDhawanAZinc oxide nanoparticles induce oxidative DNA damage and ROS-triggered mitochondria mediated apoptosis in human liver cells (HepG2)Apoptosis201217885287022395444
- AlarifiSAliDAlkahtaniSInduction of oxidative stress, DNA damage, and apoptosis in a malignant human skin melanoma cell line after exposure to zinc oxide nanoparticlesInt J Nanomedicine2013898399323493450
- GuoDBiHLiuBWuQWangDCuiYReactive oxygen species-induced cytotoxic effects of zinc oxide nanoparticles in rat retinal ganglion cellsToxicol In Vitro201327273173823232460
- JohnstonCJDriscollKEFinkelsteinJNPulmonary chemokine and mutagenic responses in rats after subchronic inhalation of amorphous and crystalline silicaToxicol Sci200056240541310911000
- SayesCMReedKLGloverKPChanging the dose metric for inhalation toxicity studies: short-term study in rats with engineered aerosolized amorphous silica nanoparticlesInhal Toxicol201022434835420001567
- LiuXKeaneMJZhongBZOngTMWallaceWEMicronucleus formation in V79 cells treated with respirable silica dispersed in medium and in simulated pulmonary surfactantMutat Res19963612–389948980693
- ECETOCEuropean Centre for Ecotoxicology and Toxicology of Chemicals. Synthetic amorphous silica (CAS No 7631-86-9)Brussels, BelgiumJACC Report2006237
- EPA. US Environmental Protection AgencyScreening-level hazard characterization of silane, dichlorodimethyl-, reaction product with silica (CASRN 68611-44-9)2011
- OECDOrganisation for Economic Co-operation and Development. SIDS dossier on synthetic amorphous silica and silicates2004
- YangKLinDXingBInteractions of humic acid with nanosized inorganic oxidesLangmuir20092563571357619708146
- GonzalezLThomassenLCPlasGExploring the aneugenic and clastogenic potential in the nanosize range: A549 human lung carcinoma cells and amorphous monodisperse silica nanoparticles as modelsNanotoxicology2010438239520925446
- ParkMVVerharenHWZwartEGenotoxicity evaluation of amorphous silica nanoparticles of different sizes using the micronucleus and the plasmid lacZ gene mutation assayNanotoxicology20115216818120735203
- ParkYHKimJNJeongSHAssessment of dermal toxicity of nanosilica using cultured keratinocytes, a human skin equivalent model and an in vivo modelToxicology20102671–317818119850098
- WangFGaoFLanMYuanHHuangYLiuJOxidative stress contributes to silica nanoparticle-induced cytotoxicity in human embryonic kidney cellsToxicol In Vitro200923580881519401228
- YeYLiuJXuJSunLChenMLanMNano-SiO2 induces apoptosis via activation of p53 and Bax mediated by oxidative stress in human hepatic cell lineToxicol In Vitro201024375175820060462
- YeYLiuJChenMSunLLanMIn vitro toxicity of silica nanoparticles in myocardial cellsEnviron Toxicol Pharmacol201029213113721787594
- ChenQXueYSunJKupffer cell-mediated hepatic injury induced by silica nanoparticles in vitro and in vivoInt J Nanomedicine201381129114023515466
- ParkY-HBaeHCJangYEffect of the size and surface charge of silica nanoparticles on cutaneous toxicityMol Cell Toxicol2013916774
- MagdolenovaZCollinsAKumarADhawanAStoneVDusinskaMMechanisms of genotoxicity. A review of in vitro and in vivo studies with engineered nanoparticlesNanotoxicology2014823327823379603
- GeiserMRothen-RutishauserBKappNUltrafine particles cross cellular membranes by nonphagocytic mechanisms in lungs and in cultured cellsEnviron Health Perspect2005113111555156016263511
- LiuLTakenakaTZinchenlcoAACationic silica nanoparticles are efficiently transferred into mammalian cellsMicro- NanoMechatronics and Human ScienceInternational Symposium on MHS ’072007281285
- YeoMKParkHGGene expression in zebrafish embryos following exposure to Cu-doped TiO2 and pure TiO2 nanometer-sized photocatalystsMol Cell Toxicol201282127137
- YeoMKKangMThe biological toxicities of two crystalline phases and differential sizes of TiO2 nanoparticles during zebrafish embryogenesis developmentMol Cell Toxicol201284317326
- NabievIMitchellSDaviesANonfunctionalized nanocrystals can exploit a cell’s active transport machinery delivering them to specific nuclear and cytoplasmic compartmentsNano Lett20077113452346117949046
- ToyokuniSOxidative stress and cancer: the role of redox regulationBiotherapy1998112–31471549677046
- RimKTKimSJSongSWParkJSEffect of cerium oxide nanoparticles to inflammation and oxidative DNA damages in H9c2 cellsMol Cell Toxicol201283271280
- ZastawnyTHAltmanSARanders-EichhornLDNA base modifications and membrane damage in cultured mammalian cells treated with iron ionsFree Radic Biol Med1995186101310227628727
- BrownDMWilsonMRMacNeeWStoneVDonaldsonKSize-dependent proinflammatory effects of ultrafine polystyrene particles: a role for surface area and oxidative stress in the enhanced activity of ultrafinesToxicol Appl Pharmacol2001175319119911559017
- KnaapenAMBormPJAlbrechtCSchinsRPInhaled particles and lung cancer. Part A: mechanismsInt J Cancer2004109679980915027112
- KarlssonHLCronholmPGustafssonJMollerLCopper oxide nanoparticles are highly toxic: a comparison between metal oxide nanoparticles and carbon nanotubesChem Res Toxicol20082191726173218710264
- LeeBCKimKTChoJGOxidative stress in juvenile common carp (Cyprinus carpio) exposed to TiO2 nanoparticlesMol Cell Toxicol201284357366
- ParkH-GYeoM-KComparison of gene expression changes induced by exposure to Ag, Cu-TiO2, and TiO2 nanoparticles in zebrafish embryosMol Cell Toxicol201392129139
- ParkEJYiJChungKHRyuDYChoiJParkKOxidative stress and apoptosis induced by titanium dioxide nanoparticles in cultured BEAS-2B cellsToxicol Lett2008180322222918662754
- PapageorgiouIBrownCSchinsRThe effect of nano- and micron-sized particles of cobalt-chromium alloy on human fibroblasts in vitroBiomaterials200728192946295817379299
- GurrJRWangASChenCHJanKYUltrafine titanium dioxide particles in the absence of photoactivation can induce oxidative damage to human bronchial epithelial cellsToxicology20052131–2667315970370
- BonvallotVBaeza-SquibanABauligAOrganic compounds from diesel exhaust particles elicit a proinflammatory response in human airway epithelial cells and induce cytochrome p450 1A1 expressionAm J Respir Cell Mol Biol200125451552111694458
- AbeSTakizawaHSugawaraIKudohSDiesel exhaust (DE)-induced cytokine expression in human bronchial epithelial cells: a study with a new cell exposure system to freshly generated DE in vitroAm J Respir Cell Mol Biol200022329630310696066
- DonaldsonKTranCLAn introduction to the short-term toxicology of respirable industrial fibresMutat Res20045531–25915288528
- WaldmanWJKristovichRKnightDADuttaPKInflammatory properties of iron-containing carbon nanoparticlesChem Res Toxicol20072081149115417672513
- BlancoDVicentSFragaMFMolecular analysis of a multistep lung cancer model induced by chronic inflammation reveals epigenetic regulation of p16 and activation of the DNA damage response pathwayNeoplasia200791084085217971904
- O’ByrneKJDalgleishAGChronic immune activation and inflammation as the cause of malignancyBr J Cancer200185447348311506482
- OberdorsterGGeleinRJohnstonCJMercerPCorsonNFinkelsteinJNAmbient ultrafine particles: inducers of acute lung injury?MohrUDungworthDLBrainJDDriscollKEGrafstromRCHarrisCCRelationships between respiratory disease and exposure to air pollutionWashingtonILSI Press1998216229
- LiXYGilmourPSDonaldsonKMacNeeWFree radical activity and pro-inflammatory effects of particulate air pollution (PM10) in vivo and in vitroThorax19965112121612228994518
- GojovaAGuoBKotaRSRutledgeJCKennedyIMBarakatAIInduction of inflammation in vascular endothelial cells by metal oxide nanoparticles: effect of particle compositionEnviron Health Perspect2007115340340917431490
- PetersKUngerREKirkpatrickCJGattiAMMonariEEffects of nano-scaled particles on endothelial cell function in vitro: studies on viability, proliferation and inflammationJ Mater Sci Mater Med200415432132515332593
- LaneDPCancer. p53, guardian of the genomeNature1992358638115161614522
- GeorgeSPokhrelSXiaTUse of a rapid cytotoxicity screening approach to engineer a safer zinc oxide nanoparticle through iron dopingACS Nano201041152920043640
- FujitaKMorimotoYOgamiAGene expression profiles in rat lung after inhalation exposure to C60 fullerene particlesToxicology20092581475519167457
- LiuXSunJEndothelial cells dysfunction induced by silica nanoparticles through oxidative stress via JNK/P53 and NF-kappaB pathwaysBiomaterials201031328198820920727582
- XiaTKovochichMBrantJComparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigmNano Lett2006681794180716895376
- MossmanBTBignonJCornMSeatonAGeeJBAsbestos: scientific developments and implications for public policyScience199024749402943012153315
- MossmanBTChurgAMechanisms in the pathogenesis of asbestosis and silicosisAm J Respir Crit Care Med19981575 Pt 1166616809603153
- RobinsonBWLakeRAAdvances in malignant mesotheliomaN Engl J Med2005353151591160316221782
- MossmanBTGeeJBAsbestos-related diseasesN Engl J Med198932026172117302659987
- GuthrieGDJrMossmanBTHealth effects of mineral dustsWashington, DCMineralogical Society of America199328
- HeintzNHJanssen-HeiningerYMMossmanBTAsbestos, lung cancers, and mesotheliomas: from molecular approaches to targeting tumor survival pathwaysAm J Respir Cell Mol Biol201042213313920068227
- ToyokuniSMechanisms of asbestos-induced carcinogenesisNagoya J Med Sci2009711–211019358470
- MagnaniCAgudoAGonzalezCAMulticentric study on malignant pleural mesothelioma and non-occupational exposure to asbestosBr J Cancer200083110411110883677
- SenyigitABayramHBabayigitCMalignant pleural mesothelioma caused by environmental exposure to asbestos in the southeast of Turkey: CT findings in 117 patientsRespiration200067661562211124643
- Biomarkers Definitions Working GroupBiomarkers and surrogate endpoints: preferred definitions and conceptual frameworkClin Pharmacol Ther2001693899511240971
- XiaTLiNNelAEPotential health impact of nanoparticlesAnnu Rev Public Health20093013715019705557
- LuoJCHsuKHShenWSInflammatory responses and oxidative stress from metal fume exposure in automobile weldersJ Occup Environ Med20095119510319136878
- AntoniniJMStoneSRobertsJREffect of short-term stainless steel welding fume inhalation exposure on lung inflammation, injury, and defense responses in ratsToxicol Appl Pharmacol2007223323424517706736
- BydashJKasmaniRNaraharisettyKMetal fume-induced diffuse alveolar damageJ Thorac Imaging2010252W27W2920351582
- CooperRGZinc toxicology following particulate inhalationIndian J Occup Environ Med2008121101320040991
- XiaoGGWangMLiNLooJANelAEUse of proteomics to demonstrate a hierarchical oxidative stress response to diesel exhaust particle chemicals in a macrophage cell lineJ Biol Chem200327850507815079014522998
- WangMXiaoGGLiNXieYLooJANelAEUse of a fluorescent phosphoprotein dye to characterize oxidative stress-induced signaling pathway components in macrophage and epithelial cultures exposed to diesel exhaust particle chemicalsElectrophoresis200526112092210815880549
- JungEJAvliyakulovNKBoontheungPLooJANelAEPro-oxidative DEP chemicals induce heat shock proteins and an unfolding protein response in a bronchial epithelial cell line as determined by DIGE analysisProteomics20077213906391817922515
- ZhangLWangMKangXOxidative stress and asthma: proteome analysis of chitinase-like proteins and FIZZ1 in lung tissue and bronchoalveolar lavage fluidJ Proteome Res2009841631163819714806
- KangXLiNWangMAdjuvant effects of ambient particulate matter monitored by proteomics of bronchoalveolar lavage fluidProteomics201010352053120029843
- LiNWangMBrambleLAThe adjuvant effect of ambient particulate matter is closely reflected by the particulate oxidant potentialEnviron Health Perspect200911771116112319654922
- LiNHarkemaJRLewandowskiRPAmbient ultrafine particles provide a strong adjuvant effect in the secondary immune response: implication for traffic-related asthma flaresAm J Physiol Lung Cell Mol Physiol20102993L374L38320562226
- OhshimaHTazawaHSyllaBSSawaTPrevention of human cancer by modulation of chronic inflammatory processesMutat Res20055911–211012216083916
- BaggsRBFerinJOberdorsterGRegression of pulmonary lesions produced by inhaled titanium dioxide in ratsVet Pathol19973465925979396140
- OberdorsterGPulmonary effects of inhaled ultrafine particlesInt Arch Occup Environ Health20017411811196075
- WangBFengWYWangTCAcute toxicity of nano- and micro-scale zinc powder in healthy adult miceToxicol Lett2006161211512316165331
- MullerJHuauxFFonsecaAStructural defects play a major role in the acute lung toxicity of multiwall carbon nanotubes: toxicological aspectsChem Res Toxicol20082191698170518636756
- ChithraniBDChanWCElucidating the mechanism of cellular uptake and removal of protein-coated gold nanoparticles of different sizes and shapesNano Lett2007761542155017465586
- GrattonSERoppPAPohlhausPDThe effect of particle design on cellular internalization pathwaysProc Natl Acad Sci U S A200810533116131161818697944
- PacurariMYinXJZhaoJRaw single-wall carbon nanotubes induce oxidative stress and activate MAPKs, AP-1, NF-kappaB, and Akt in normal and malignant human mesothelial cellsEnviron Health Perspect200811691211121718795165
- MaplesKRJohnsonNFFiber-induced hydroxyl radical formation: correlation with mesothelioma induction in rats and humansCarcinogenesis19921311203520391330349
- DonaldsonKBeswickPHGilmourPSFree radical activity associated with the surface of particles: a unifying factor in determining biological activity?Toxicol Lett1996881–32932988920751
- GhioAJStonehuernerJDaileyLACarterJDMetals associated with both the water-soluble and insoluble fractions of an ambient air pollution particle catalyze an oxidative stressInhal Toxicol1999111374910380158
- KaganVETyurinaYYTyurinVADirect and indirect effects of single walled carbon nanotubes on RAW 264.7 macrophages: role of ironToxicol Lett200616518810016527436
- LamCWJamesJTMcCluskeyRHunterRLPulmonary toxicity of single-wall carbon nanotubes in mice 7 and 90 days after intratracheal instillationToxicol Sci200477112613414514958
- WickPManserPLimbachLKThe degree and kind of agglomeration affect carbon nanotube cytotoxicityToxicol Lett2007168212113117169512
- FubiniBGhiazzaMFenoglioIPhysico-chemical features of engineered nanoparticles relevant to their toxicityNanotoxicology20104434736320858045
- MaynardADBaronPAFoleyMShvedovaAAKisinERCastranovaVExposure to carbon nanotube material: aerosol release during the handling of unrefined single-walled carbon nanotube materialJ Toxicol Environ Health A20046718710714668113
- ShvedovaAAKisinERMercerRUnusual inflammatory and fibrogenic pulmonary responses to single-walled carbon nanotubes in miceAm J Physiol Lung Cell Mol Physiol20052895L698L70815951334
- ThessALeeRNikolaevPCrystalline ropes of metallic carbon nanotubesScience199627352744834878662534
- KimYHParkHWRyooZYLeeHSKimDHLeeSAbnormal effects of unpurified and purified multi-walled carbon nanotubes in A549, Jurkat and THP-1 cell linesMol Cell Toxicol201281103112
- ChandraSBarickKCBahadurDOxide and hybrid nanostructures for therapeutic applicationsAdv Drug Deliv Rev20116314–151267128121729727
- MagdolenovaZBilanicovaDPojanaGImpact of agglomeration and different dispersions of titanium dioxide nanoparticles on the human related in vitro cytotoxicity and genotoxicityJ Environ Monit201214245546422277962
- FuYSDuXWKulinichSAStable aqueous dispersion of ZnO quantum dots with strong blue emission via simple solution routeJ Am Chem Soc200712951160291603318044896
- MunnierECohen-JonathanSLinassierCNovel method of doxorubicin-SPION reversible association for magnetic drug targetingInt J Pharm20083631–217017618687392
- ValoisCRBrazJMNunesESThe effect of DMSA-functionalized magnetic nanoparticles on transendothelial migration of monocytes in the murine lung via a beta2 integrin-dependent pathwayBiomaterials201031236637419822361
- MauriziLBishtHBouyerFMillotNEasy route to functionalize iron oxide nanoparticles via long-term stable thiol groupsLangmuir200925168857885919572525
- LimJKMajetichSATiltonRDStabilization of superparamagnetic iron oxide core-gold shell nanoparticles in high ionic strength mediaLangmuir20092523133841339319928938
- XieJXuCKohelrNHouYSunSControlled PEGylation of monodisperse Fe3O4 nanoparticles for reduced non-specific uptake by macrophage cellsAdv Mater2007192031633166
- SoenenSJHHodeniusMSchmitz-RodeTDe CuyperMProtein-stabilized magnetic fluidsJ Magn Magn Mater20083205634641
- NasongklaNBeyERenJMultifunctional polymeric micelles as cancer-targeted, MRI-ultrasensitive drug delivery systemsNano Lett20066112427243017090068
- PradhanPGiriJRiekenFTargeted temperature sensitive magnetic liposomes for thermo-chemotherapyJ Control Release2010142110812119819275
- SayesCMLiangFHudsonJLFunctionalization density dependence of single-walled carbon nanotubes cytotoxicity in vitroToxicol Lett2006161213514216229976
- SinghRPantarottoDLacerdaLTissue biodistribution and blood clearance rates of intravenously administered carbon nanotube radiotracersProc Natl Acad Sci U S A200610393357336216492781
- ChenPKuoCWLaiJJLCytotoxicity of the functionalized gold and silver nanorodsMat Res Soc2007951105110
- YimWCLeeB-MKwonYCross-experimental analysis of microarray gene expression datasets for in silico risk assessment of TiO2 nano-particlesMol Cell Toxicol201283229239
- ParkS-KKimJ-JYuA-RLeeM-YParkS-KElectrolyzed-reduced water confers increased resistance to environmental stressesMol Cell Toxicol201283241247
- JiangJOberdörsterGBiswasPCharacterisation of size, surface charge and agglomeration state of nanoparticle dispersions for toxicological studiesJ Nanopart Res2009117789
- Harush-FrenkelODebottonNBenitaSAltschulerYTargeting of nanoparticles to the clathrin-mediated endocytic pathwayBiochem Biophys Res Commun20073531263217184736
- LockmanPRKoziaraJMMumperRJAllenDDNanoparticle surface charges alter blood-brain barrier integrity and permeabilityJ Drug Target2004129–1063564115621689
- NanABaiXSonSJLeeSBGhandehariHCellular uptake and cytotoxicity of silica nanotubesNano Lett2008882150215418624386
- CliftMJRothen-RutishauserBBrownDMThe impact of different nanoparticle surface chemistry and size on uptake and toxicity in a murine macrophage cell lineToxicol Appl Pharmacol2008232341842718708083
- GonzalezLLisonDKirsch-VoldersMGenotoxicity of engineered nanomaterials:a critical reviewNanotoxicology20082252273
- LandsiedelRKappMDSchulzMWienchKOeschFGentoxicity investigations on nanomaterials: methods, preparation and characterization of test material, potential artifacts and limitations- many questions, some answersMutat Res200968124125819041420
