References
- Pople , J.A. , Schneider , W.G. and Bernstein , H.J. 1959 . High-resolution Nuclear Magnetic Resonance , McGraw Hill . Chaps 15 and 16
- Ronayne , J. and Williams , D.H. 1969 . Annual Rev. N.M.R. Spectroscopy , 2 : 83 – 83 .
- Raynes , W.T. , Buckingham , A.D. and Bernstein , H.J. 1962 . J. chem. Phys. , 36 : 3481 – 3481 .
- Jameson , A.K. , Jameson , C.J. and Gutowsky , H.S. 1970 . J. chem. Phys. , 53 : 2310 – 2310 .
- Jameson , C.J. and Jameson , A.K. 1971 . Molec. Phys. , 20 : 957 – 957 .
- Kromhout , R.A. and Linder , B. 1971 . J. chem. Phys. , 54 : 1834 – 1834 .
- Bloembergen , N. and Dickinson , W.C. 1950 . Phys. Rev. , 79 : 179 – 179 . McConnell, H. M., and Robertson, R. E., 1958, J. chem. Phys., 29, 1361.
- Van Vleck , J.H. 1932 . The Theory of Electric and Magnetic Susceptibilities , 272 – 272 . Oxford University Press .
- James , T.C. and Thibault , R.J. 1964 . J. chem. Phys. , 41 : 2806 – 2806 .
- The values of and used in these calculations were taken from Hirschfelder J.O. Curtis C.F. Bird R.B. Molecular Theory of Gases and Liquids 1954 (Wiley), Appendix I A; The combining ‘rules’ and were used to estimate the Lennard-Jones parameters for mixed pairs
- Watson , R.E. and Freeman , A.J. 1967 . Hyperfine Interactions , Edited by: Freeman , A.J. and Frankel , R.B. Academic Press . Chap. 2
- Moore , C.E. 1958 . Atomic Energy Levels, Circular 467 , Vol. III , National Bureau of Standards .
- Turner , D.W. 1968 . Proc. R. Soc. A , 307 : 15 – 15 . gives the ionization potential of O2 as 12·07 eV. From extrapolation of the energies of the processes Ba2+ + e- →Ba+ and and Cs+ + e-→Cs (see [12]) one can estimate the electron affinity of Xe as +2 eV. Thus, the energy for the Xe-O2 + resonance structure, after considering coulomb attraction at ∼4 Å, shows it unfavourable by at least 9 eV. (We thank Dr. J. F. Liebman for suggesting this energy estimate.)
- Brion , H. , Moser , C. and Yamazaki , M. 1959 . J. chem. Phys. , 30 : 673 – 673 .
-
Cade
,
P.E.
unpublished double-zeta M.O. wavefunction for O2 is of the following form:
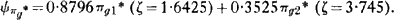 Note that Cade's ζ values for a 2p orbital correspond to Z
eff/2, and that where is the Slater orbital i centred on atom a.
Note that Cade's ζ values for a 2p orbital correspond to Z
eff/2, and that where is the Slater orbital i centred on atom a.
- Herman , F. and Skillman , S. 1963 . Atomic Structure Calculations , Prentice-Hall .
- Ray , S. and Das , T.P. 1968 . Phys. Rev. , 174 : 32 – 32 . Ray, S., Lyons, J. D., and Das, T. P., 1968, Phys. Rev., 174, 104. Lyons, J. D., Ray, S., and Das, T. P., 1968, Phys. Rev., 174, 112.
- Ray , S. , Das , G. , Maldonado , P. and Wahl , A.C. 1970 . Phys. Rev. A , 2 : 2196 – 2196 .
- Clarke , G.A. 1962 . J. chem. Phys. , 36 : 2211 – 2211 .
- Arditi , M. and Carver , T.R. 1958 . Phys. Rev. , 112 : 449 – 449 .
- Buckingham , A.D. and Pople , J.A. 1955 . Trans. Faraday Soc. , 51 : 1173 – 1173 .
- Abramowitz , M. and Stegun , I.A. 1964 . Handbook of Mathematical Functions , 917 – 917 . National Bureau of Standards . 923