ABSTRACT
Antioxidants are important ingredients that are present in fruits and vegetables (FAV). With increased consumption of FAV in its raw and processed form, a predominantly plant-based diet rich in FAV could reduce the risk of the development of chronic human diseases. This review highlights the potentials of the various types of antioxidants containing FAV; their impact on human health as nutraceuticals, pharmaceuticals, and phytoceuticals; as well as prospects in tackling some chronic human diseases. The structures and activity relationship of the antioxidant compounds, as well as their mechanism of action, are examined from current scientific investigations. Information provided herein will give more insight into the roles of antioxidant ingredients present in FAV.
Introduction
Among the various food plants, fruits and vegetables (FAV) are reported to have health-improving benefits.[1–Citation3] Some FAV such as the citrus fruits (orange, grapefruit, lime, lemon), grapes, pomegranates, apples, dates, green and yellow vegetables (peppers), cabbage, strawberries, carrots, dark leafy greens, and banana,[Citation4–6] have been known worldwide to contain antioxidants. A characteristic of antioxidants is that they exert both additive and synergistic effects in reducing the risk of chronic diseases.[Citation7–9] Fruits and vegetables, therefore, exert protective roles against chronic diseases such as cardio- and cerebrovascular, ocular and neurological diseases, strokes, cancer, diabetes, hypertension, and blood-related diseases.[Citation10–15] Several epidemiological studies have presented compelling evidence that the potential of FAV to combat the majority of these health conditions are associated with the natural compounds found in them.[Citation1,Citation4] Low intake of FAV was estimated to be responsible for 31% of ischemic heart disease and 11% of stroke worldwide.[Citation2] In the report of the joint FAO/WHO consultation on diet, nutrition, and prevention of chronic diseases, a minimum daily intake of 400–500 g per day of FAV was recommended for the prevention of high blood pressure, stroke, cardiovascular diseases, and other micronutrient related deficiencies.[Citation2,Citation7] Inadequate intake of FAV is therefore a significant risk-factor that causes several nutritionally related non-communicable diseases (NCDs).[Citation2,Citation7,Citation13,Citation16]
The protective effect of FAV has generally been attributed to their antioxidant constituents (natural radical terminators) such as vitamins A, C (ascorbic acid), E (α-tocopherol), β- and α-carotene and glutathione.[Citation17,Citation18] Alkaloids, trepenoids, sulfur-containing compounds, phenolic and polyphenolic compounds () are other antioxidants present in FAV,[Citation15] which reduces oxidative damage by neutralizing the activities of free-radicals.[Citation13] These bioactive, non-nutritive plant compounds generally called phytochemicals further engage in the termination of chain reactions by removing the free radical intermediates.[Citation18] Niki and Noguchi[Citation20] noted that carotenoids, a significant bioactive present in FAV, are particularly effective at inhibiting the oxidation caused by singlet oxygen. Polyphenolic flavonoids, another group of bioactives, are plant metabolites that exert numerous biological and pharmacological properties.[Citation21,Citation22] Flavonoids consisting of anthocyanins, anthocyanidins, flavonols, flavones, and flavanones have been shown to possess antioxidant,[Citation23] anti-inflammatory,[Citation24] antimutagenic,[Citation25,Citation26] and anticarcinogenic properties.[Citation27,Citation28] Trombino et al.[Citation29] and Marinova et al.[Citation30] further reported a synergistic interaction between phenolic acid, β-carotene, and ascorbic acid, as well as between flavonoids and tocopherols inherent in FAV.
Figure 1. Groupings of bioactive compounds present in fruits and vegetables. Source: Desjardins.[Citation19] β-carotene α-carotene
![Figure 1. Groupings of bioactive compounds present in fruits and vegetables. Source: Desjardins.[Citation19] β-carotene α-carotene](/cms/asset/501286ad-0f8c-424a-8375-cbccb209f532/ljfp_a_1866597_f0001_b.gif)
Fruits and vegetables when consumed separately provide known bioactive that exert beneficial health effects. However, processing which is one of the means of improving the consumption of FAV in combination will further synergistically enhance the effects of bioactives on molecular mechanisms that are important for disease preventions.[Citation31] Combined foods such as salads, ready-to-eat meals, and mixtures of fresh vegetables or fruit ingredients[Citation32] will enhance the synergistic action of the different phytochemicals. Lichtenthaler and Marx[Citation33] postulated that processed products from FAV, such as juice, are rich sources of bio-accessible antioxidants, though their bioavailability is said to be relatively low. Septembre-Malaterre et al.[Citation34] investigated FAV as a source of nutritional compounds and phytochemicals as well as their changes during fermentation. It was observed that though FAV serves as a source of phytochemicals in their raw and unprocessed state, fermentation ensures changes in the forms and profiles of these bioactive, thereby leading to modifications of their health-related properties. This review thus highlights bioactive non-nutrient compounds in FAV in their raw and processed forms, to elucidate their roles in disease prevention and health.
High antioxidant fruits and vegetables
Fruits and vegetables are edible plant parts including roots, stalks, tubers, bulbs, leaves, stems, fruits, and flowers.[Citation35,Citation36] Fruits irrespective of their level of consumption contain varying antioxidant constituents (). However, the distinct antioxidants that can be derived from FAV include vitamins C and E, carotenoids, and phenolic compounds. Research has further shown that FAV acts as a major source of vitamins, polyphenols, carotenoids, glucosinolates, saponins, and sterols when consumed in sufficient amounts.[Citation60–63] Some tropical and subtropical fruits such as guava, mango, passion fruit, pineapple, banana, litchi, papaya, passiflora, kiwano, carambola, feijoa, star fruit, lime, and longan are utilized as ingredients in the diets of food consumed in North America and Europe.[Citation64–66] Burton-Freeman[Citation67] stated that consumption of FAV, apart from increasing the antioxidant capacity of the human blood, counterbalances the negative effects of high fat and carbohydrate meals.
Table 1. Comparative list of selected fruit and vegetables (FAVs) phytochemicals and their antioxidant-richness
From a taxonomical viewpoint and as shown in the works of Carlsen et al.,[Citation68] FAV and its plant parts contain variant levels of bioactive compounds that often result in some FAV recording higher concentration of antioxidants than others. Antioxidants present in FAV include the primary and secondary metabolites of plants.[Citation8] Secondary metabolites constitute most of the antioxidant compounds and they are classed into terpenoids, phenolics, alkaloids, and sulfur-containing compounds[Citation8] based on their chemical structures.[Citation69] Primary metabolites with antioxidant activity include vitamin B complex, protein and nonprotein amino acids, fatty acids, and organic acids. Although substantive information has been provided about the occurrence and content of different chemical compounds in FAV, it is yet to be effectively systemized.[Citation8,Citation9]
shows the antioxidant contents of FAV in mg/100 g of fresh food. The citrus family consists of fruits that are made up of high nutritional and antioxidant properties.[Citation60] Rich in ascorbic acid, citrus fruits also contain flavanone glycosides (hesperidin, narirutin, and naringenin), limonoids, flavones (sinensetin and nobiletin), and phenylpropanoids such as hydrocinnamates.[Citation13,Citation81,Citation82] Lako et al.[Citation80] who investigated the phytochemical flavonols, carotenoids, and the antioxidant properties of a wide selection of Fijian fruit, vegetables, and other readily available foods, showed that green leafy vegetables had the highest antioxidant capacity followed by fruits and root crops. Anyasi et al.[Citation83] also reported that banana (‘Muomva-red cultivar’) contains a high content of total polyphenols which is an indication that the banana could be a source of bio-nutrients with great medicinal and health functions. A different class of flavonoids has been reported in Opuntia cactus with types and concentration varying according to variety, tissue type, and maturation.[Citation84] Other reports have indicated that plants in the Cactaceae family produce flavonol 3-O-glycosides (quercetin, kaempferol, and isorhamnetin), dihydroflavonols, flavanones, and flavanonols.[Citation85] Nearly all reports on flavonoids found in Opuntia cacti have dealt with extraction from the floral tissue.
Table 2. Antioxidant contents of some fruits and vegetables in mg/100 g fresh food
High antioxidant-rich fruits
Fruits with a documented high concentration of antioxidants belong to the plant members of Rosaceae (dogrose, sour cherry, blackberry, strawberry, and raspberry), Empetraceae (crowberry), Ricaceae (blueberry), Grossulariaceae (blackcurrant), Juglandaceae (walnut), Asteraceae (sunflowerseed), Punicaceae (pomegranate), and Zingiberaceae (ginger).[Citation86] Pomegranate, grape, orange, plum, pineapple, lemon, date, kiwi, clementine, and grapefruit have been identified with high antioxidant properties.[Citation35,Citation86] Other fruits associated with a high amount of antioxidants include dog rose, sour cherry, blackberry, strawberry, raspberry, cloudberry, and rowanberry. Among these fruits, berries account for the highest antioxidant content and dog rose has the highest compared to others such as crowberry, wild berry, black currant, sour cherry, wild blackberry, wild strawberry, cultivated blackberry, and cowberry/cranberry. Berries have a high content of phytochemicals such as flavonoids, tannins, stilbenoids, phenolic acids, and lignans.[Citation87]
High antioxidant-rich vegetables
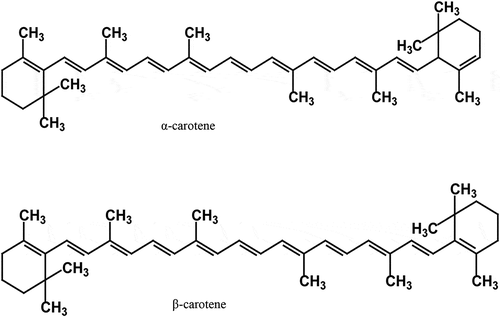
Broccoli, brussels sprout, green cabbage, tomato, cauliflower, spinach, leek, lettuce, and sweet pepper have been reported with different antioxidant levels.[Citation8,Citation88] Hounsome and Hounsome[Citation8] showed that the phytochemical α-carotene and antioxidant β-carotene () are richly found in broccoli (1 and 779 mg/100 FW), carrot (4.6 and 8.8 mg/100 FW), tomato (112 and 393 mg/100 FW), pea (19 and 485 mg/100 FW), and sweet pepper (59 mg/100 FW, β-carotene only). These phytochemicals were reported to vary in structure and function from vegetable to vegetable and from cultivar to cultivar, with the level of maturity, postharvest handling, and processing among other factors having a significant impact on their variability.[Citation89,Citation90] Vegetables rich in ascorbic acid include beans, broccoli, cabbage, cauliflower, cress, pea, spinach, spring onion, and sweet peppers.[Citation49] Asparagus, brussels sprout, cabbage, carrot, cauliflower, kale, lettuce, spinach, sweet potato, and turnip are rich sources of vitamin E.[Citation8] Red pepper has also been reported to have a high content of vitamin C (144 mg/100 g).[Citation91]
Fruits and vegetable phytochemicals and human health
There is an array of evidence that consumption of FAV is important for human health as they are richly endowed with health-improving nutrients.[Citation6,Citation92–95] Burton-Freeman[Citation67] postulated that antioxidant-rich FAV increases the antioxidant capacity of the blood, thus decreasing the risk of developing diseases such as cancer, diarrhea, coronary atherosclerosis, and gastrointestinal tract diseases. Results from epidemiological and laboratory studies conducted by Eckert,[Citation96] Harding et al.[Citation97] and Rouanet[Citation98] supported the hypothesis that consumption of FAV will prevent and significantly reduce cancer, Alzheimer’s diseases, diabetes, and heart diseases.
Further epidemiological studies have proposed an inverse correlation between high intake of FAV and many degenerative and aging diseases.[Citation16,Citation99–101] Improvement in diabetes mellitus, digestive problems, immune disorder, cataract, bronchitis, asthma, and other respiratory syndromes have all been reported upon regular intake of FAV. Butt and Sultan[Citation95] also suggested that humans relying on FAV have a 10–15% lower risk of developing cataracts than those who consumed lower proportions of FAV. Jaganath and Crosier[Citation7] indicated that a proper balance between oxidants and antioxidants is necessary to maintain health as alterations to this balance often leads to pathological responses that result in functional disorders and diseases. It has also been postulated that a non-vegetarian diet may alter hormone production, metabolism, or action at the cellular level thereby increasing the incidence of breast, colorectal, and prostate cancer.[Citation102] The health functions of FAV are therefore based on their phytochemical content, which in their various chemical forms, possess antioxidative, antiviral, anti-inflammatory, antimicrobial, anti-carcinogenic, antiangiogenic, antibiotic, and antithrombotic properties.[Citation8,Citation103,Citation104]
Baskar et al.[Citation105] demonstrated that regularly, free radicals are synthesized in the body either naturally or through external factors such as environmental stress, thus resulting in various degenerative diseases that harm the body. Though the body has an inbuilt defense mechanism to scavenge these radicals, there is a need for the utilization of additional antioxidants available in FAV.[Citation106] Diseases such as cancer, Alzheimer’s disease, Parkinson’s disease, arthritis, atherosclerosis, and aging are formed in the body as a result of these radicals.[Citation105] Antioxidants especially those synthesized by FAV therefore play a vital role in ensuring that these free radicals are scavenged, thereby preventing them from causing harm.[Citation106] This is especially more important due to the increased emphasis on the consumption of antioxidants from natural sources as used for preventive and therapeutic medicine.[Citation107]
Dietary antioxidants are defined as food compounds that significantly reduce the deleterious effects of ROS, RNS, or both, on the normal physiological function in humans.[Citation94,Citation108] ROS (oxygen ions, free radicals, and peroxides) and RNS (nitrous anhydride, peroxynitrite, and nitrogen dioxide radicals) cause oxidation, nitration, halogenation, and deamination of biomolecules of all types, including lipids, proteins, carbohydrates, and nucleic acids, with the formation of toxic and mutagenic products.[Citation109,Citation110] Pathogenesis of non-communicable and non-nutritionally related human diseases such as brain stroke, diabetes mellitus, rheumatoid arthritis, Parkinson’s disease, Alzheimer’s disease and cancer have been associated with the oxidative damage of these oxidants to cells[Citation9,Citation111] of which FAV phytochemicals are counter-intuitive. Examples include lycopene: the principal carotenoid in tomatoes,[Citation112] a very efficient quencher of singlet oxygen in the biological system. Another example is vitamin E, a major lipid-soluble chain-breaking antioxidant in humans, that protects the DNA, low-density lipoproteins, and polyunsaturated fatty acids from free radical-induced oxidation.[Citation8]
Vitamin C
Bruno et al.[Citation113] stated that the roles of vitamin C include the regulation of cell growth, cell signaling, apoptosis, antioxidants, and as cofactors for enzymes. Vitamin C occurs mainly in FAV and it is reduced by heat during processing; hence, its nutrient density in raw FAV is higher than in processed forms.[Citation114] Vitamin C scavenges reactive oxygen species (ROS) and reactive nitrogen species (RNS) and also regenerates α-tocopherol and coenzyme Q from α-tocopherol and coenzyme Q radical. This resultant action helps in maintaining the antioxidant activities of α-tocopherol and coenzyme Q.[Citation114] The studies by Lee et al.[Citation115] and Chen et al.[Citation116] postulated that ascorbate induces lipid hydroperoxide decomposition to genotoxins in the absence of redox-active metal ions and also leads to a reduction in the growth of aggressive tumor xenografts. As documented in studies conducted on animal species, consumption of FAV rich in vitamin C will greatly help protect the body against cardiovascular disorders, gastrointestinal disorders, cancer, skin infections, and diabetes through the reduction of insulin glycation and an increase in glucose homeostasis.[Citation117]
Vitamin E
Consisting of eight different types: α-, β-, γ- and δ-tocopherols and the α-, β-, γ- and δ-tocotrienols, vitamin E can be obtained from vegetable oils, nuts, and seeds of different fruits.[Citation114] Experimental model studies in vitro and in vivo have shown the antioxidative, pro-antioxidative, anti-inflammatory, modulation of cell signaling, antiproliferation, antiangiogenesis, and apoptosis induction effects of vitamin E. Other works have also shown that α-tocopherol is the major form in human tissues. Accounting for more than 90% of the literature on vitamin E studies, α-tocopherol is the most studied vitamin E isoform.[Citation118] α-tocopherols, and other forms of vitamin E play crucial roles in protection against lipid peroxidation, scavenging of peroxyl radicals,[Citation119] reaction with ROS and RNS and reduction in the synthesis of ROS from NADPH oxidase.[Citation120] The pro-oxidant effect of α-tocopherol also includes its ability to reduce redox-active metals such as copper and iron.
When present in the human skin, vitamin E serves as a vital line of dermal antioxidant protection. Its isoforms such as tocopherols and tocotrienols can protect the skin against disease conditions such as dermatitis, UV-irradiation induced skin injury, and chemically induced oxidative stress in animal models.[Citation121] Thus, vitamin E obtained from FAV has found application as cosmetics due to its protective dermal effects.[Citation122]
Carotenoids
Carotenoids consist of a group of lipophilic pigmented compounds that is made up of over six hundred fat-soluble plant pigments. They are chiefly responsible for colors such as yellow, red, and orange present in FAV and from which the compounds are derived. Major carotenoids present in human diets are α-, β-carotene, lutein, lycopene, zeaxanthin, astaxanthin, and β-cryptoxanthin, with these compounds playing an active role in the protection of plants from the damaging and scourging effects of exposure to sunlight.[Citation114,Citation123] Carotenoids function in the body as a precursor of vitamin A, thus preventing vitamin A deficiency in the body.[Citation124] Carotenoids further undertake antioxidant activities by scavenging reactive species, oxides, and radicals,[Citation125] suppressing inflammatory responses in both in vivo and in vitro systems, as well as assisting in the modulation of cell signaling and induction of apoptosis.[Citation126,Citation127] This is apart from their roles in cardiovascular protective activities, obesity, cancer, and gastrointestinal disorders.[Citation128–135]
Phenolic compounds
Stalikas[Citation136] showed that phenolic compounds are secondary metabolites of plant origin that carry one (phenols) or several (polyphenols) hydroxyl moieties in their aromatic ring. Among the major dietary sources are FAV, with an average daily consumption of approximately 1 g/day.[Citation137,Citation138] Phenolic compounds consist of approximately 8,000 naturally occurring metabolites which are divided into two main groups; flavonoids and non-flavonoids.
The flavonoids, a member of the polyphenols, are planar molecules whose varied structure arises partially due to methoxylation, prenylation, glycosylation, or hydroxylation. Their heterocyclic six-member ring with oxygen is encased by two aromatic rings (). Uppu and Parinandi[Citation139] reported that flavonoids are derived from two biosynthetic pathways: the shikimate and the acetate pathways. As natural pigments in FAV, they occur in colors such as yellow in flavonols and flavones; orange, blue, purple, red, and violet in anthocyanins.[Citation140] Flavonoids can be classified as flavones, flavonols, flavan-3-ols, flavanones, anthocyanidins, proanthocyanidins, isoflavones, and dihydroflavanols. The nonflavonoids () comprise simple phenols, hydrolyzable tannins, cinnamic acid, coumarins, xanthones, stilbenes, lignans, secoiridoids, benzophenones, acetophenones, phenylacetic, and benzoic acids. Classification of the nonflavonoids is dependent on the number of carbon frameworks present in the organic acids.[Citation136]
Figure 3. Basic structure of flavonoids. R = OH: flavonols; R = H: flavones.Source: Stalikas[Citation136]
![Figure 3. Basic structure of flavonoids. R = OH: flavonols; R = H: flavones.Source: Stalikas[Citation136]](/cms/asset/b09fb503-7dd3-46f3-a5b2-e58277272a06/ljfp_a_1866597_f0003_b.gif)
Figure 4. Structures of naturally occurring phenolic acids. A, Hydroxybenzoic; and B, hydroxycinnamic structures. R1, R2, and R3 represents H, OH, and OCH3. Source: Stalikas[Citation136]
![Figure 4. Structures of naturally occurring phenolic acids. A, Hydroxybenzoic; and B, hydroxycinnamic structures. R1, R2, and R3 represents H, OH, and OCH3. Source: Stalikas[Citation136]](/cms/asset/cc2985e6-4f46-428e-80d2-e8c6ee6b2bfb/ljfp_a_1866597_f0004_b.gif)
Phenolic compounds undertake antioxidant, pro-oxidant, anti-inflammatory activities and also exert great influence on the bioavailability of nitric oxide in humans.[Citation114] Stoclet et al.[Citation141] and Mann et al.[Citation142] demonstrated that bioavailability of nitric oxide involves a reduction in nitric oxide bioavailability by phenols, a step which is vital in the reduction of cardiovascular diseases. Its antioxidant activities involve ROS, RNS, and other reactive species scavenging activities; the inhibition of ROS synthesis from cells; and the induction of endogenous cellular antioxidant enzymes.[Citation143–146] The scavenging activities have been attributed largely to their redox properties which enable them to function as reducing agents, singlet oxygen quenchers, and hydrogen donors.[Citation147]
Determination of the antioxidant capacity of fruits and vegetables
The concentration of antioxidants in FAV varies in their degree of activity and mode of action, hence the difficulty in their analysis. Various methods exist for the determination of the antioxidant capacity of FAV.[Citation23,Citation80,Citation148,Citation149] However, there are known factors that affect the determination of the antioxidant capacity in fruits and such factors include geographical location, agricultural and farming practices, the season of cultivation, particle size, amount of extraction steps, sample to solvent ratio, and temperature of the process. [Citation140,Citation150,Citation151] Sample preparation is therefore a critical step in the total determination of antioxidants in FAV.[Citation136] Established protocols for the analysis and determination of antioxidant capacity in FAV () include oxygen radical absorbance capacity (ORAC), Trolox equivalent antioxidant capacity (TEAC), 2,2-azino-bis-3-ethylbenzthiazoline-6-sulfonic acid (ABTS), 1,1-diphenyl-2-picrylhydrazyl (DPPH), ferric reducing ability of plasma (FRAP), and vitamin C equivalent antioxidant capacity (VCEAC). The ORAC method is one of the most widely used methods for evaluating antioxidant capacity (AOC) due to known and unknown antioxidants present in tested foods.[Citation157] Peroxyl radical-scavenging activities of water-soluble and lipid-soluble antioxidants in samples are evaluated by the hydrophilic ORAC (H-ORAC) and lipophilic ORAC (L-ORAC) methods, respectively.
Table 3. Analytical methods for determination of antioxidant properties of fruits and vegetables
Mechanism of action of fruits and vegetable phytochemicals
The protective effect of FAV has generally been attributed to their antioxidant constituents (natural radical terminators) from known sources as well as from other unidentified compounds.[Citation17,Citation18] Niki and Noguchi[Citation20] stated that carotenoids are particularly effective at inhibiting the oxidation caused by singlet oxygen. Gil et al.[Citation23] postulated that flavonoids possess antioxidant properties, anti-inflammatory,[Citation24] antimutagenic[Citation25,Citation26] and anticarcinogenic properties.[Citation27,Citation28] Antioxidants delay the onset of free radical formation by their ability to donate hydrogen atom (or electron/proton) or chelation of metals involved in the formation of ROS.[Citation7,Citation8] The operational mode of antioxidants in negating the impact of free radicals to cells involves a combination of different mechanisms which include direct termination of the free radicals,[Citation158] and post-modification of the active compounds during metabolism.[Citation111,Citation159,Citation160] The latter mechanism often results in metabolites that are chemically, biologically, and in several instances, functionally distinct from the dietary form and such features underpin their bioactivity.[Citation161] Williams and Spencer[Citation162] hypothesized that cells respond to phytochemicals mainly through direct interactions with receptors, enzymes involved in signal transduction, or through modifying gene expressions which may result in modification of the redox status of the cell that may induce series of redox-dependent reactions.
Williams and Spencer[Citation162] also postulated that there is now emerging evidence that phytochemicals such as flavonoids may play an important role as modulators of intracellular signaling cascades, which are pivotal to the cell machinery. Intracellular signaling cascades are major routes of communication between the plasma membrane and regulatory targets in various intracellular compartments.[Citation7] These signaling cascades seem to consist of up to five tiers of protein kinases that sequentially activate each other by phosphorylation, which consequently affects the activity of transcription factors that regulates gene expression.[Citation163] Jaganath and Crosier[Citation7] showed that the presence of these cascades enables the cells to respond to variant stresses and signals which in turn regulate numerous cell processes, including growth, proliferation, and death (apoptosis). Phytochemicals can exert modulatory effects in cells through selective actions on different components of the signaling cascades.[Citation164,Citation165] The intracellular concentration of phytochemicals required to affect cell signaling pathways is considerably lower than those required to have an impact on cellular anti-oxidant capacity and their metabolites may still retain the ability to interact with cell signaling proteins, even if their antioxidant activity is diminished.[Citation160,Citation166]
Flavonoids are abundant in plants and the majority of plant tissues possess the ability to synthesize flavonoids.[Citation167] In plants, the leaves, flowers, and fruits contain flavonoid glycosides; the woody tissue contains aglycone; and the seeds may contain either the flavonoid glycosides or aglycones. Apigenin, a flavone present in parsley and celery hinders proinflammatory cytokines and HIFR, VEGF, and COX-2 expression through the inhibition of nuclear factor-κB (NF-κB), PI3K/Akt, and ATF/cyclic AMP responsive element signaling pathways.[Citation168–170] Tangeretin, a polymethoxylated flavone that is abundant in peels of citrus fruits serves as an anticarcinogenic agent by suppressing IL-1β-induced COX-2 expression through the inhibition of p38 MAPK, c-Jun N-terminal kinase (JNK), and Akt activation.[Citation171] Kaempferol, a flavonol in broccoli and tea diminishes the activity of inflammation-related genes such as iNOS and COX-2 by blocking signaling of STAT-1, NF-κB, and AP-1 in activated macrophages[Citation172] and human endothelial cells.[Citation173] Quercetin, another flavonol present in leafy green vegetables, onions, broccoli, apples, and grapes acts as a potent antioxidant and anti-inflammatory agent. Quercetin inhibits the expression of pro-inflammatory cytokines in mast cells[Citation174] and suppresses TNF-induced NF-κB and CBP/p300 recruitment to pro-inflammatory gene promoters.[Citation175] Furthermore, quercetin diminishes total cholesterol, triglycerides, and low-density lipoprotein and reduces glycemia as well as high-density lipoprotein levels through the inhibition of 11β-hydroxysteroid dehydrogenase type 1.[Citation176,Citation177]
Hamalainen et al.[Citation172] stated that naringenin, a flavanone occurring in oranges inhibits iNOS protein and gene expression by blocking the activation of NF-κB. Naringenin-7-O-glucoside is reported to stop cardiomyocytes from doxorubicin-induced toxicity through the induction of endogenous antioxidant enzymes by phosphorylation of extracellular signal-regulated kinases 1 and 2 (ERK1/2) and nuclear translocation of Nrf2[Citation178] and through the stabilization of the cell membrane as well as the reduction of reactive oxygen species generation.[Citation179] Cyanidin, an anthocyanidin inherent in cherries and strawberries was shown to inhibit tumor promoter-induced carcinogenesis and tumor metastasis in vivo through the modulation of the expression of COX-2 and TNF-R.[Citation180] Cyanidin-3-O-rutinoside retards in vivo absorption of carbohydrates through the inhibition of α-glucosidase.[Citation181] Delphinidin, another anthocyanidin present in dark fruits has been found to contribute antiangiogenic activity through the inhibition of the PDGF-BB/PDGF receptor (PDGFR)-β in smooth muscle cells.[Citation182] Anthocyanins suppress lipid peroxidation in caco-2 cells[Citation183] and reduce ethanol-induced migration of breast cancer cells through blockage of ethanol-induced activation of ErB2/cSrc/FAK pathway essential for cell migration.[Citation184] Furthermore, anthocyanins suppress benzo[a]pyrene-7,8-diol-9,10-epoxide-induced cyclooxygenase-2 (COX-2) expression mostly by hindering the activation of the Fyn signaling pathway;[Citation185] prevent nitric oxide synthase and COX-2 and reduction in nitric oxide and prostaglandin E2 production (PGE2);[Citation186] as well as inhibit IκBα phosphorylation, thus suppressing NF-κB activity in cell and in vivo models,[Citation187–189] thereby contributing to its chemopreventive ability.
Carotenoids such as lycopene present in papaya, tomatoes, watermelon, oranges, and pink grapefruit reduce inflammatory response through the lowering of iNOS and COX-2 gene expression[Citation190] as well as IL-12 production through blocking MAPK signaling and the activation of NF-κB in murine dendritic cells.[Citation191] Similarly, β-carotene inherent in fruits such as carrots, palm fruits, mangoes, papayas, and green leafy vegetables inhibits LPS-induced iNOS, COX-2, and TNF-α expression by decreasing phosphorylation and degradation of IκBR and nuclear translocation of NF-κB in macrophages.[Citation192] Lutein, a yellow pigment present in leafy vegetables such as spinach and kale was reported to inhibit LPS- and H2O2-induced pro-inflammatory gene expression by decreasing the activity of PI3K and NF-κB inducing kinase (NIK) and phosphorylation of Akt in RAW264.7 cells.[Citation193]
Fruits and vegetable phytochemicals and cancer
Cancer, a degenerative disease across the world and one of the leading causes of death,[Citation95,Citation101,Citation194] has been associated with lifestyle, environmental and dietary factors. The pathogenesis is attributable to genetic mutation, smoking, heavy metal ingestion, and lack of proper diet.[Citation95] Studies conducted by Lee and Smith[Citation195] and Wolfe et al.[Citation196] have shown that about one-third of all cancer cases and one-half of hypertension and cardiovascular infections are diet-related. However, Willett[Citation197] stated that appropriate dietary intake can prevent more than 30% of cancerous cell growth. Similarly, Amiot and Lairon[Citation198] and Butt and Sultan[Citation95] reported that about 30–40% chances of cancer can be prevented by adopting a proper physical and dietary lifestyle. Consumption of vegetables rich in dietary antioxidants has been linked to a lower risk of different types of cancer,[Citation199] especially the mouth, pharyngeal, esophageal, lung, stomach, and colon cancer.[Citation95,Citation200] Fahey[Citation57] explained that the anti-carcinogenic effect of glucosinolates is due to the activation of enzymes involved in the detoxification of carcinogens, inhibition of enzymes modifying steroid hormone metabolism, and protection against oxidative damage. Isothiocyanates resulting from glucosinolate hydrolysis possess antioxidative and anti-proliferative activities.[Citation201] Sulforaphane, an isothiocyanate found in broccoli, cauliflower, collard green, and turnips, is effective in reducing the risk of certain types of cancer such as breast and colon cancer.[Citation11,Citation95] Garlic, onions, chives, leek, and scallions are good sources of sulfur-containing compounds and are effective in reducing the risk of certain cancers.[Citation95]
Hollman and Kitan[Citation202] demonstrated that vegetables rich in flavonoids such as bean, broccoli, endive, leek, onion, and tomato were able to impact positively on cancerous conditions as they stimulate enzymes involved in detoxification of carcinogenic substances and inhibit inflammation associated with local production of free radicals. Similarly, tomatoes and their lycopene-rich products have the potential for minimizing certain forms of cancer. Sterols which are essential for the synthesis of prostaglandins and leukotriene are important components of the immune system.[Citation8] They are richly found in broccoli, brussels sprout, carrot, cauliflower, celery, tomato, soy, and spinach.[Citation203] Due to the structural similarity of sterols to cholesterol, sterols are also reported to inhibit cholesterol absorption.[Citation8] Also, of their cholesterol-lowering effects, plant sterols may possess anticancer, antiatherosclerosis, anti-inflammation, and antioxidant properties.[Citation103,Citation104] Although dietary phytoestrogens have been reported to help reduce the risk of certain hormone-stimulated malignancy such as breast and prostate cancers,[Citation204] their anti-carcinogenic properties still await further confirmation.[Citation95]
In humans, lignans possess antioxidant and (anti) estrogenic properties and may reduce the risk of certain cancers.[Citation8] Reduced cancer risks have also been associated with higher urinary lignan excretion.[Citation205] Lignans formed by oxidative dimerization of two phenylpropane units are present in nature in the free form, while their glycoside derivatives exist in minor forms.[Citation206] Lignans are secondary plant metabolites that serve as an important source of phytoestrogens.[Citation207] These secondary plant metabolites are present in fruits such as pears, plums, raspberries, strawberries, grapes, kiwi, and apricot and in vegetables such as asparagus, carrot, cabbage, broccoli, and garlic.[Citation34,Citation207] The different types of lignans () that occur in foods include medioresinol, syringaresinol, sesamin, and the lignan precursor sesamolin.[Citation207,Citation209,Citation210] Heinonen et al.[Citation208] postulated that lignans are considered phytoestrogens due to their being metabolized into enterodiol and enterolactone in the intestinal microflora. Phytoestrogens which are present in apples and vegetables have shown protective potential against cancer, CVD, menopausal symptoms, and osteoporosis.[Citation34,Citation211] Prasad[Citation212] and Saleem et al.[Citation213] reported that epidemiological and pharmacological studies on lignan presented it to be beneficial in the prevention of cancer, atherosclerosis, reduction of inflammation, and risk factors for oxidative stress and stroke. In a study that examined the relationship between plasma lignan concentration and the incidence of colon cancer in over 57,000 participants between the age of 50–64, higher concentrations of lignans were seen to lower the risk of colon cancer in women.[Citation207,Citation214]
Figure 5. Structures of plant lignans present in fruits and vegetables. Source: Heinonen et al.[Citation208]
![Figure 5. Structures of plant lignans present in fruits and vegetables. Source: Heinonen et al.[Citation208]](/cms/asset/d51cc6d0-32cc-4225-b12e-f8904accea8f/ljfp_a_1866597_f0005_b.gif)
Fruits and vegetables and cardiovascular diseases
Cardiovascular disease (CVD) is one of the leading causes of death globally and has other health contributing risk factors such as obesity and type II diabetes mellitus. Experimental studies conducted by Brigelius-Flohe and Frebar,[Citation215] and Butt and Sultan[Citation95] showed that greater intake of FAV proffers great coronary care and regulation of blood cholesterol levels, which helps in the prevention of CVDs. Dietary fibers in artichoke, sweet potato, and turnip,[Citation95] flavonoids in bean, broccoli, leek, endive, and tomatoes,[Citation8] all have potential in the suppression of CVDs. FAV has been shown () to contain lots of phytonutrients and in amounts that contribute to the prevention of degenerative diseases in the body.[Citation13]
Table 4. Fruits and vegetables and their health functions
Antioxidant-fortified fruit and vegetable products and human health
In both developed and developing countries, there is increasing interest in “natural” products, their content of phytochemicals and antioxidants[Citation250] in the fight against human ailments. Some of these antioxidant-rich natural products include tea, shrubs, wine, and fruit juice.[Citation92,Citation250,Citation251] There are many novel high antioxidant fruit ingredients, for example, acai berries and wolfberries, concentrations, and extracts of pomegranate, cranberry, and blueberry. Acai berries are similar to other dark-colored fruits because they are rich in anthocyanins, while wolfberries are rich in carotenoids, lutein, and zeaxanthin. Other emerging functional ingredients of beverages include sea buckthorn and baobab fruit powder. Gao et al.[Citation252] showed that sea buckthorn contains significant amounts of tocopherols, tocotrienols, and carotenoids. Processed vegetable products do contain these nutrients in portions from which the body can derive immense benefits when consumed.
Vitamin C has been found useful in the biofortification of fruit juice and drinks. Related works conducted by Biesalski[Citation253] and Mann et al.[Citation142] have elaborated the beneficial role of carotenoids in cardiovascular pathophysiology, when used as dietary supplements. Ascorbate can also be obtained through supplements especially in the juice where it is often added, as its deficiency in the body has been known to result in scurvy, anemia, blotchy bruises, poor wound healing, and susceptibility to diseases.[Citation114] However, natural antioxidants obtained from plants and dependent on the dose of consumption are said to be more safe and healthy than artificially synthesized antioxidants. Fan et al.[Citation254] hypothesized that fruits such as blue honeysuckle, blueberry, and lingberry which contain high amounts of anthocyanins can be utilized in replacing the red, blue, purple, and black artificially synthesized colorants of FAV products in the food industry.
Natural-antioxidant fruit and vegetable products
Preference for the use of natural products is due largely to their minimal side effects and the increasing preference for natural product used in preventive and therapeutic medicine. There is increasing advocacy and the use of natural vitamins C and E as protection from ultraviolet radiation in the skin and other cosmetic products. Processed food drinks such as fruit juice and wine are presently supplemented with ascorbic acid that is derived from fruits. Natural carotenoid-derived colorants obtained from FAV are increasingly in use to replace artificially synthesized colorants in the manufacture of food products by food processors. Berries and their products are potentially excellent antioxidant sources. However, during the processing of berries to jams, total phenol content is reduced resulting in lower antioxidant values in processed berry products than in fresh berries.[Citation80]
Future direction on natural antioxidant-rich fruits and vegetables
The health claims, in vitro, from antioxidants present in fruits are still being conducted in experimental models, with many of the claims on their therapeutic effects yet to be verified. Research on phenolic compounds is of growing interest because of the vital biological and pharmacological characteristics which these antioxidants have shown in human health.[Citation136] Hence, bioactives are the topic of discussion at most food and health-related conferences. Further research is therefore needed to verify these health claims and to ascertain antioxidant contents of most pre-packed fresh-cut versus whole FAV,[Citation255] as well as dried FAV that is incorporated into other food products. Recent research revealed that fruit peels and seeds, such as grape seeds and peels, pomegranate peel, wampee peel, and mango seed kernel may potentially possess antioxidant properties. Valorization of the entire plant parts of FAV including peels, rind, seeds, core, rag, stones, pods, vine, skin, pomace, shell, and stem to ensure extraction and utilization of these antioxidants in different food systems are currently ongoing.[Citation256,Citation257] With the increasing preference and advocacy for natural and minimally processed food products, more studies are ongoing on the antioxidant compounds and their functional, nutraceutical, and prebiotic roles in humans to push forward against cancer, neurodegenerative, and cardiovascular diseases.[Citation258] One of such is the postulation by Suntres[Citation259] that antioxidant liposomes hold an important role in future research on antioxidants. Genetic engineering has also been suggested as one of the research areas that should aim at breeding genetically modified plants that can produce higher quantities of specific compounds, yielding higher quantities of antioxidants. Traditional and exotic fruits as well as ornamental plants are receiving attention more than ever before, all in an attempt to maximize their benefits and significance on human health. Instrumental methods of assessing antioxidant activity are likely to become more important in the future as methods that do not require the use of chemical reagents or solvents reduce waste disposal problems. Thus, different mechanisms of extraction of the antioxidants inherent in FAV aside from the known conventional methods are presently being explored in the food processing industry. These unconventional extraction techniques such as supercritical fluid, pulsed electric field, microwave-assisted, ultrasound-assisted, and enzyme-assisted extraction methods are advantageous due to their high yield, use of organic solvent, low process time, reduced use of energy, and less waste generation.[Citation257,Citation260]
Conclusion
Though some FAV serves as food mostly in tropical countries, several studies have shown that the consumption of FAV worldwide is insufficient to meet the daily nutritional needs for human health and wellness. With the many health claims attributed to the antioxidant content of FAV, sustained and increased production will lead to increased consumption by humans and an effect of a decrease in degenerative disorders. However, with the increasing world population, there would continually be a reliance on synthetic antioxidant supplements. Therefore, scaling up of production and increased consumption in both scenarios may lead to a reduction of terminal and degenerative diseases.
Additional information
Funding
References
- Eastwood, M. A. Interaction of Dietary Antioxidants in Vivo: How Fruit and Vegetables Prevent Disease? Q. J. Med.. 1999, 92, 527–530.
- WHO. Fruit and Vegetables for Health; Report of a Joint FAO/WHO Workshop; Geneva, Switzerland: World Health Organization, 2004; pp pp. 7–9.
- Serna-Saldivar, S. O. Cereal Grains: Properties, Processing and Nutritional Attributes; Taylor and Francis Group: Boca Raton, FL, 2010; pp pp. 606–609.
- Rice-Evans, C.; Miller, N. J. Antioxidants – The Case for Fruits and Vegetable in Diet. Brit. Food J. 1995, 19(9), 35–40. DOI: https://doi.org/10.1108/00070709510100163.
- FSA. Eatwell: 8 tips for making healthier choices. http://www.food.gov.uk/multimedia/pdfs/publication/eatwell0708.pdf. 2010 (accessed 26, May 2013).
- Radovich, T. J. K. Biology and Classification of Vegetables. In Handbook of Vegetables and Vegetable Processing; Sinha, N. K., Hui, Y. H., Evranuz, E. O., Siddiq, M., Ahmed, J., Eds.; Blackwell Publishing: Iowa, 2011; pp pp. 43–47.
- Jaganath, I. B.; Crozier, A. Overview of Health-promoting Compounds in Fruit and Vegetables. In Improving the Health-promoting Properties of Fruit and Vegetable Products; Tomas-Barberan, F. A., Gil, M. I., Eds.; Woodhead Publishing: Cambridge, 2008; pp pp. 3–4.
- Hounsome, N.; Hounsome, B. Biochemistry of Vegetables: Major Classes of Primary (Carbohydrates, Amino Acids, Fatty Acids, Vitamins and Organic Acids) and Secondary Metabolites (Terpenoids, Phenolics, Alkaloids, and Sulphur-containing Compounds), in Vegetables. In Handbook of Vegetables and Vegetable Processing; Sinha, N. K., Hui, Y. H., Evranuz, E. O., Siddiq, M., Ahmed, J., Eds.; Blackwell Publishing: Iowa, USA, 2011; pp pp. 23–58.
- Pisoschi, A. M.; Negulescu, G. P. Methods for Total Antioxidant Activity Determination: A Review. Biochem, Anal. Biochem. 2011, 1, 106. DOI: https://doi.org/10.4172/2161-1009.1000106..
- Block, G.; Patterson, B.; Subhar, A. Fruit, Vegetables and Cancer Prevention: A Review of Epidemiological Evidence. Nutr. Cancer. 1992, 18, 1–29. DOI: https://doi.org/10.1080/01635589209514201.
- Steinmetz, K. A.; Potter, J. D. Vegetables, Fruit and Cancer Prevention: A Review. J. Am. Diet. Assoc. 1996, 96, 1027–1039. DOI: https://doi.org/10.1016/S0002-8223(96)00273-8.
- Ness, A. R.; Powles, J. W. Fruit and Vegetables, and Cardiovascular Disease: A Review. Int. J. Epidemiol. 1997, 26, 1–13. DOI: https://doi.org/10.1093/ije/26.1.1.
- Kaur, C.; Kapoor, H. C. Antioxidants in Fruits and Vegetables-the Millennium’s Health. Int. J. Food Sci. Technol.. 2001, 36, 703–725. https://doi.org/10.1046/j.1365-2621.2001.00513.x
- Hung, H. C.; Joshipura, K. J.; Jiang, R.; Hu, F. B.; Hunter, D.; Smith-Warner, S. A.; Colditz, G. A.; Rosner, B.; Spiegelman, D.; Willett, W. C. Fruit and Vegetable Intake and Risk of Major Chronic Disease. J. Nat. Cancer Inst. 2004, 96(21), 1577–1584.
- Barret, D. M.; Somogyi, L.; Ramaswamy, H. Processing Fruits Science Technology; CRC Press: Florida, 2005; pp 5–6.
- Liu, R. H. Health Benefits of Fruit and Vegetables are from Additive and Synergistic Combination of Phytochemicals. Am. J. Clin. Nutr.. 2003, 78, 517S–520S. DOI: https://doi.org/10.1093/ajcn/78.3.517S.
- Sies, H.; Stahl, W. Vitamins E and C, β-carotene and Carotenoids as Antioxidants. Am. J. Clin. Nutr.. 1995, 62, 1315s–1321s. DOI: https://doi.org/10.1093/ajcn/62.6.1315S.
- Landete, J. M. Dietary Intake of Natural Antioxidants: Vitamins and Polyphenols. Crit. Rev. Food. Sci. Nutr. 2013, 53(7), 706–721. DOI: https://doi.org/10.1080/10408398.2011.555018.
- Desjardins, Y. Fruit and Vegetables and Health: An Overview. In Horticulture: Plants for People and Places; Dixon, G. R., Aldous, D. E., Eds.; Springer Science+Business Media Dordrecht, 2014; Vol. 3, pp 965–1000.
- Niki, E.; Noguchi, N. Evaluation of Antioxidant Capacity. What Capacity Is Being Measured by Which Method?. IUBMB Life. 2000, 50(4–5), 323–329. DOI: https://doi.org/10.1080/15216540051081119.
- Cook, N. C.; Samman, S. Flavonoids—chemistry, Metabolism, Cardioprotective Effects, and Dietary Sources. Nutr Biochem. 1996, 7, 66–76. DOI: https://doi.org/10.1016/0955-2863(95)00168-9.
- Hollman, P. C. H.; Hertog, M. G. L.; Katan, M. B. Analysis and Health Benefits of Flavonoids. Food Chem.. 1996, 57, 43–46. DOI: https://doi.org/10.1016/0308-8146(96)00065-9.
- Gil, M. I.; Tomas-Barberan, F. A.; Hess-Pierce, B.; Holcroft, D. M.; Kader, A. A. Antioxidant Activity of Pomegranate Juice and Its Relationship with Phenolic Composition and Processing. J. Agr. Food Chem.. 2000, 48, 4581–4589. DOI: https://doi.org/10.1021/jf000404a.
- Middleton, E.; Kandaswani, C. Effects of Flavonoids on Immune and Inflammatory Cell Functions. Biochem. Pharmacol.. 1992, 43, 1167–1179. DOI: https://doi.org/10.1016/0006-2952(92)90489-6.
- Namiki, M. Antioxidant/antimutagens in Food. Crit. Rev. Food Sci. Nutr.. 1990, 29, 273–300.
- Edenharder, R.; Keller, G.; Platt, K. L.; Unger, K. K. Isolation and Characterization of Structurally Novel Antimutagenic Flavonoids from Spinach (Spinacia Oleracea). J Agr. Food Chem. 2001, 49, 2767–2773. DOI: https://doi.org/10.1021/jf0013712.
- Yoshida, M.; Sakai, T.; Hosokawa, N.; Marui, N.; Matsumoto, K.; Fugioka, A.; Nishino, H.; Aoike, A. The Effect of Quercetin in Cell Cycle Progression and Growth of Human Gastric Cancer Cells. Fed. Eur. Bio. Sci. Lett. 1990, 260, 10–13. DOI: https://doi.org/10.1016/0014-5793(90)80053-L.
- Dragsted, L. O.; Strube, M.; Larsen, J. C. Cancer-protective Factors in Fruits and Vegetables: Biochemical and Biological Background. Pharmacol. Toxicol. 1993, 72(S1), 116–135. DOI: https://doi.org/10.1111/j.1600-0773.1993.tb01679.x.
- Trombino, S.; Serini, S.; Di Nicuolo, F.; Celleno, L.; Ando, S.; Picci, N.; Calviello, G.; Palozza, P. Antioxidant Effect of Ferulic Acid in Isolated Membranes and Intact Cells: Synergistic Interactions with Alpha-tocopherol, Beta-carotene, and Ascorbic Acid. J. Agr. Food Chem.. 2004, 52(4), 2411–2420. DOI: https://doi.org/10.1021/jf0303924.
- Marinova, E.; Toneva, A.; Yanishilieva, N. Synergistic Antioxidant Effect of α-tocopherol and Myricetin on the Autoxidation of Triacylglycerols of Sunflower Oils. Food Chem.. 2008, 106(2), 628–633. DOI: https://doi.org/10.1016/j.foodchem.2007.06.022.
- van Breda, S. G. J.; de Kok, T. M. C. M. Smart Combinations of Bioactive Compounds in Fruits and Vegetables May Guide New Strategies for Personalized Prevention of Chronic Diseases. Mol. Nutr. Food Res.. 2018, 62, 1700597. DOI: https://doi.org/10.1002/mnfr.201700597.
- Yildiz, F. Initial Preparation, Handling, and Distribution of Minimally Processed Refrigerated Fruits and Vegetables. In Minimally Processed Refrigerated Fruits and Vegetables, Second Edition. Food Engineering Series; Yildiz, F., Wiley, R. C., Eds.; Springer Nature: New York, USA, 2017; pp pp. 53–92.
- Lichtenthaler, R.; Marx, F. Total Oxidant Scavenging Capacities of Common European Fruit and Vegetable Juices. J. Agr. Food. Chem. 2005, 53(1), 103–110. DOI: https://doi.org/10.1021/jf0307550.
- Septembre-Malaterre, A.; Remize, F.; Poucheret, P. Fruits and Vegetables, as a Source of Nutritional Compounds and Phytochemicals: Changes in Bioactive Compounds during Lactic Fermentation. Food Res. Int.. 2018, 104, 86–99. DOI: https://doi.org/10.1016/j.foodres.2017.09.031.
- Berrino, F.; Villarini, A. Fruit and Vegetables and Cancer. In Improving the Health-promoting Properties of Fruit and Vegetable Products; Tomas-Barberan, F. A., Gil, M. I., Eds.; Woodhead Publishing: Cambridge, 2008; pp pp. 76–94.
- Moyo, M.; Amoo, S. O.; Ncube, B.; Ndhlala, A. R.; Finnie, J. F.; Van Staden, J. Phytochemical and Antioxidant Properties of Unconventional Leafy Vegetables Consumed in Southern Africa. S. Afr. J. Bot. 2013, 84, 65–71. DOI: https://doi.org/10.1016/j.sajb.2012.09.010.
- Arts, I. C. W.; van de Putte, B.; Hollman, P. C. H. Catechin Contents of Foods Commonly Consumed in the Netherlands. 1. Fruits, Vegetables, Staple Foods and Processed Foods. J. Agr. Food Chem.. 2000, 48, 1748–1751.
- Pascual-teresa de, S.; Santos-Buelga, C.; Rivas-Gonzalo, J. C. Quantitative Analysis of Flavan-3-ols in Spanish Foodstuff and Beverages. J. Agr. Food Chem.. 2000, 48, 5331–5337. https://doi.org/10.1021/jf000549h
- Del Verde-Mendez, C. M.; Forster, M. P.; Rodriguez-Delgado, M. A.; Rodriguez-Rodriguez, E. M.; Diaz-Romero, C. Content of Free Phenolic Compounds in Banana from Tenerife (Canary Islands) and Ecuador. Eur. Food Res. Technol.. 2003, 217, 287–290. DOI: https://doi.org/10.1007/s00217-003-0762-8.
- Harnly, J. M.; Doherty, R. F.; Beecher, G. R.; Holden, J. M.; Haytowitz, D. B.; Bhagwat, S.; Gebhardt, S. Flavonoid Content of U.S. Fruits, Vegetables, and Nuts. J. Agr. Food Chem.. 2006, 54(26), 9966–9977. DOI: https://doi.org/10.1021/jf061478a.
- Bennet, R. N.; Shiga, T. M.; Hassimotto, N. M. A.; Rosa, E. A. S.; Lajolo, F. M.; Cordenunsi, B. R. Phenolics and Antioxidant Properties of Fruit Pulp and Cell Wall Fractions of Postharvest Banana (Musa Acuminata Juss.) Cultivars. J. Agr. Food Chem.. 2010, 58, 7991–8003. DOI: https://doi.org/10.1021/jf1008692.
- Anyasi, T. A.; Jideani, A. I. O.; Mchau, G. R. A. Functional Properties and Postharvest Utilization of Commercial and Noncommercial Banana Cultivars. Compr. Rev. Food Sci. F. 2013, 12(5), 509–522. DOI: https://doi.org/10.1111/1541-4337.12025.
- Kaur, C.; Kapoor, H. C. Antioxidants in Fruits and Vegetables – The Millennium’s Health. Int. J. Food Sci. Technol.. 2001, 36, 703–725. https://doi.org/10.1046/j.1365-2621.2001.00513.x
- Teixeira, A.; Baenas, N.; Dominguez-Perles, R.; Barros, A.; Rosa, E.; Moreno, D. A.; Natural Bioactive, G.-V. C. Compounds from Winery By-Products as Health Promoters: A Review. Int. J. Mol. Sci.. 2014, 15, 15638–15678. DOI: https://doi.org/10.3390/ijms150915638.
- Otero, P.; Viana, M.; Herrera, E.; Bonet, B. Antioxidant and Prooxidant Effects of Ascorbic Acid, Dehydroascorbic Acid and Flavonoids on LDL Submitted to Different Degrees of Oxidation. Free Radical Res.. 1997, 27(6), 619–626. DOI: https://doi.org/10.3109/10715769709097865.
- Bohm, V.; Puspitasari-Nienaber, N. L.; Ferruzi, M. G.; Schwartz, S. J. Trolox Equivalent Antioxidant Capacity of Different Geometrical Isomers of Alpha-carotene, Beta-carotene, Lycopene, and Zeaxanthin. J. Agr. Food Chem.. 2002, 50(1), 221–226. DOI: https://doi.org/10.1021/jf010888q.
- Brown, J. E.; Khodr, H.; Hider, R. C.; Rice-Evans, C. A. Structural Dependence of Flavonoid Interactions with Cu2+ Ions: Implications for Their Antioxidant Properties. Biochem. J.. 1998, 330(30), 1173–1178. DOI: https://doi.org/10.1042/bj3301173.
- Harris, G. K.; Qian, Y.; Leonard, S. S.; Sbarra, D. C.; Shi, X. Luteolin and Chrysin Differentially Inhibit Cyclooxygenase-2 Expression and Scavenge Reactive Oxygen Species but Similarly Inhibit prostaglandin-E2 Formation in RAW. 264.7 Cells. J. Nutr.. 2006, 136(6), 517–521. DOI: https://doi.org/10.1093/jn/136.6.1517.
- USDA. (2005) United States Department of Agriculture, Agricultural Research Service. USDA National Nutrient Database for Standard Reference Release http//www.nal.usda.gov/fnic/foodcomp/Data/2005(18) (accessed 18 August 2013).
- Rhodes, C. J.; Dintinger, T. C.; Moynihan, H. A.; Reid, I. D. Radio Labelling Studies of Free Radical Reactions Using Muonium (The Second Hydrogen Radioisotope): Evidence of a Direct Antioxidant Role for Vitamin K in Repair of Oxidative Damage to Lipids. Magn. Reson. Chem. 2000, 38(8), 646–649. DOI: https://doi.org/10.1002/1097-458X(200008)38:8<646::AID-MRC699>3.0.CO;2-W.
- Damon, M.; Zhang, N. Z.; Haytowitz, D. B.; Booth, S. L. Phylloquinone (Vitamin K1) Content of Vegetables. J. Food Compound Anal. 2005, 18(8), 751–758. DOI: https://doi.org/10.1016/j.jfca.2004.07.004.
- Chen, W. J.; Song, J. R.; Guo, P.; Wen, Z. Y. Butein, a More Effective Antioxidant than α-tocopherol. J. Mol. Struct. 2006, 736(1–3), 161–164. DOI: https://doi.org/10.1016/j.theochem.2005.12.035.
- Normen, L.; Johnsson, M.; Anderson, H.; van Gameren, Y.; Dutta, P. Plant Sterol in Vegetables and Fruits Commonly Consumed in Sweden. Eur. J. Nutr.. 1999, 38(2), 84–89. DOI: https://doi.org/10.1007/s003940050048.
- Yoshida, Y.; Niki, E. Antioxidant Effects of Phytosterol and Its Component. J. Nutr. Sci. Vitaminol.. 2003, 49(4), 277–280. DOI: https://doi.org/10.3177/jnsv.49.277.
- Meyer, A. S.; Heinonen, M.; Frankel, E. N. Antioxidant Interactions of Catechin, Cyaniding, Caffeic Acid, Quercitin, and Ellagic Acid on Human LDL Oxidation. Food Chem.. 1998, 61(1–2), 71–75. DOI: https://doi.org/10.1016/S0308-8146(97)00100-3.
- Pietta, P. G. Flavonoids as Antioxidants. J. National Produc. 2000, 63(7), 1035–1042. DOI: https://doi.org/10.1021/np9904509.
- Fahey, J. W.; Zalcmann, A. T.; Talalay, P. The Chemical Diversity and Distribution of Glucosinolates and Isothiocyanates among Plants. Phytochem. 2001, 56(1), 5–51. DOI: https://doi.org/10.1016/S0031-9422(00)00316-2.
- Johnson, I. T. Glucosinolates in the Human Diet. Bioavailability and Implication for Health. Phytochem. Rev.. 2002b, 1(2), 183–188. DOI: https://doi.org/10.1023/A:1022507300374.
- Gürbüz, N.; Uluişik, S.; Frary, A.; Doğanlar, S. Health Benefits and Bioactive Compounds of Eggplant. Food Chem.. 2018, 268, 602–610. DOI: https://doi.org/10.1016/j.foodchem.2018.06.093.
- Ye, X.; Chen, J.; Liu, D.; Jiang, P.; Shi, J.; Xue, S.; Wu, D.; Xu, J.; Kakuda, Y. Identification of Bioactive Composition and Antioxidant Activity in Young Mandarin Fruits. Food Chem.. 2011, 124, 1561–1566. DOI: https://doi.org/10.1016/j.foodchem.2010.08.013.
- Cassileth, B. Complementary Therapies, Herbs, and Other OTC Agents. Oncology-NY. 2008, 22, 1202.
- Alothman, M.; Bhat, R.; Karim, A. A. Antioxidant Capacity and Phenolic Content of Selected Tropical Fruits from Malaysia, Extracted with Different Solvents. Food Chem.. 2009, 115, 785–788. DOI: https://doi.org/10.1016/j.foodchem.2008.12.005.
- Kalpna, R.; Mital, K.; Sumitra, C. Vegetable and Fruit Peels as a Novel Source of Antioxidants. J. Med. Plant Res.. 2011, 5(1), 63–71.
- Dube, M.; Zunker, K.; Neidhart, S.; Carle, R.; Steinhart, H.; Paschke, A. Effect of Technological Processing on the Allergenicity of Mangoes (Mangifera Indica L. J. Agr. Food Chem.. 2004, 52, 3938–3945. DOI: https://doi.org/10.1021/jf030792r.
- Wu, L. C.; Hsu, H. W.; Chen, Y. C.; Chiu, C. C.; Lin, Y. I.; Ho, J. A. Antioxidant and Antiproliferative Activities of Red Pitaya. Food Chem.. 2005, 95, 319–327. DOI: https://doi.org/10.1016/j.foodchem.2005.01.002.
- De Assis, S. A.; Vellosa, J. C. R.; Brunetti, I. L.; Khalil, N. M.; Leite, K. M. D. C.; Martins, A. B. G. Antioxidant Activity, Ascorbic Acid and Total Phenol of Exotic Fruits Occurring in Brazil. Int. J. Food Sci. Nutr. 2009, 60, 439–448. DOI: https://doi.org/10.1186/1475-2891-9-3.
- Burton-Freeman, B. Postprandial Metabolic Events and Fruit-derived Phenolics: A Review of the Science. Brit. J. Nutr. 2010, 104(Suppl. 3), S1–S14. DOI: https://doi.org/10.1017/S0007114510003909.
- Carlsen, M. H.; Halvorsen, B. L.; Holte, K.; Bøhn, S. K.; Dragland, S.; Sampson, L.; Willey, C.; Senoo, H.; Umezono, Y.; Sanada, C.; et al.. The Total Antioxidant Content of More than 3100 Foods, Beverages, Spices, Herbs and Supplements Used Worldwide. Nutr. J. 2010, 9(3), 1–11.
- Shahidi, F.; Chandrasekara, A.; Zhong, Y. Bioactive Phytochemicals in Vegetables. In Handbook of Vegetables and Vegetable Processing; N. K, S., Hui, Y. H., Evranuz, E. O., M, S., J, A., Eds.; Blackwell Publishing: Iowa, USA, 2011; pp pp. 125–150.
- Hertog, M. G. L.; Hollman, P. C. H.; Venema, D. P. Optimization of a Quantitative HPLC Determination of Potentially Anticarcinogenic Flavonoids in Vegetables and Fruits. J. Agr. Food Chem.. 1992a, 40, 1591. DOI: https://doi.org/10.1021/jf00021a023.
- De Pascual-Teresa, S.; Santos-Buelga, C.; Rivas-Gonzalo, J. C. Quantitative Analysis of Flavan-3-ols in Spanish Foodstuffs and Beverages. J. Agr. Food Chem.. 2000, 48, 5331. https://doi.org/10.1021/jf000549h
- Aherne, S. A.; O’Brien, N. M. Dietary Flavonols: Chemistry, Food Content and Metabolism. Nutr. 2002, 18, 75. DOI: https://doi.org/10.1016/S0899-9007(01)00695-5.
- Gu, L.; Kelm, M.; Hammerstone, J. F.; Beecher, G.; Cunningham, D.; Vannozzi, S.; Prior, R. L. Fractionation of Polymeric Procyanidins from Lowbush Blueberry and Quantification of Procyanidins in Selected Foods with an Optimized Normal-phase HPLC MS Fluorescent Detection Method. J. Agr. Food Chem.. 2002, 50, 4852. DOI: https://doi.org/10.1021/jf020214v.
- Charoensiri, R.; Kongkachuichai, R.; Suknicom, S.; Sungpuag, P. Beta-carotene, Lycopene, and Alpha-tocopherol Contents of Selected Thai Fruits. Food Chem.. 2009, 113, 202–207. DOI: https://doi.org/10.1016/j.foodchem.2008.07.074.
- da Silva, L. M. R.; de Figueiredo, E. A. T.; Ricardo, N. M. P. S.; Vieira, I. G. P.; de Figueiredo, R. W.; Brasil, I. M.; Gomes, C. L. Quantification of Bioactive Compounds in Pulps and By-products of Tropical Fruits from Brazil. Food Chem.. 2014, 143, 398–404. DOI: https://doi.org/10.1016/j.foodchem.2013.08.001.
- Wu, X.; Beecher, G. R.; Holden, J. M.; Haytowitz, D. B.; Gebhardt, S. E.; Prior, R. L. Concentrations of Anthocyanins in Common Foods in the United States and Estimation of Normal Consumption. J. Agr. Food Chem.. 2006, 54, 4069. DOI: https://doi.org/10.1021/jf060300l.
- Liggins, J.; Bluck, L.; Runswick, S.; Atkinson, C.; Coward, W. A.; Bingham, S. A. Daidzein and Genistein Contents of Vegetables. Brit. J. Nutr. 2000, 84, 717. DOI: https://doi.org/10.1017/S0007114500002075.
- Crozier, A.; Burns, J.; Aziz, A. A.; Stewart, A. J.; Rabiasz, H. S.; Jenkins, G. I.; Edwards, C. A.; Lean, M. Antioxidant Flavonols from Fruits, Vegetables and Beverages: Measurements and Bioavailability. Biol. Res. 2000, 33, 79. DOI: https://doi.org/10.4067/S0716-97602000000200007.
- Hertog, M. G. L.; Hollman, P. C. H.; Katan, M. B. Content of Potentially Anticarcinogenic Flavonoids of 28 Vegetables and 9 Fruits Commonly Consumed in the Netherlands. J. Agr. Food Chem.. 1992b, 40, 2379. DOI: https://doi.org/10.1021/jf00024a011.
- Lako, J.; Trenerry, V. C.; Wahlqvist, M.; Wattanapenpaiboon, N.; Sotheeswaran, S.; Premier, R. Phytochemical Flavonols, Carotenoids and the Antioxidant Properties of a Wide Selection of Fijian Fruit, Vegetables and Other Readily Available Foods. Food Chem.. 2007, 101, 1727–1741. DOI: https://doi.org/10.1016/j.foodchem.2006.01.031.
- Gil-Izquierdo, A.; Gil, M. I.; Ferreres, F.; Tomas-Barberan, F. In Vitro Availability of Flavanoids and Other Phenolics in Orange Juice. J. Agr. Food Chem.. 2001, 49, 1035–1041. DOI: https://doi.org/10.1021/jf0000528.
- Del Caro, A.; Piga, A.; Vacca, V.; Agabbio, M. Changes of Flavonoids, Vitamin C and Antioxidant Capacity in Minimally Processed Citrus Segments and Juices during Storage. Food Chem.. 2004, 84, 99–105. DOI: https://doi.org/10.1016/S0308-8146(03)00180-8.
- Anyasi, T. A.; Jideani, A. I. O.; Mchau, G. A. Morphological, Physicochemical, and Antioxidant Profile of Noncommercial Banana Cultivars. Food Sci. Nutri. 2015, 3(3), 221–232. DOI: https://doi.org/10.1002/fsn3.208.
- Wallace, R. S. Biochemical Taxonomy and the Cactaceae. Cactus and Succulent J.. 1986, 58, 35–38.
- Burret, F.; Lebreton, P.; Voirin, B. Les aglycones flavoniques des Cactees: Distribution, signification. J. Nat. Prod.. 1982, 45, 687–693. DOI: https://doi.org/10.1021/np50024a006.
- Halvorsen, B. L.; Holte, K.; Myhrstad, M. C. W.; Barikmo, I.; Hvattum, E.; Remberg, S. F.; Wold, A. B.; Haffner, K.; Baugerød, H.; Andersen, L. F.; et al.. R. A Systematic Screening of Total Antioxidants in Dietary Plants. J. Nutr.. 2002, 132, 461–471. DOI: https://doi.org/10.1093/jn/132.3.461.
- Amakura, Y.; Umino, Y.; Tsuji, S.; Tonogai, Y. Influence of Jam Processing on the Radical Scavenging Activity and Phenolic Content in Berries. J. Agr. Food Chem.. 2000, 48, 6292–6297. DOI: https://doi.org/10.1021/jf000849z.
- Horbowicz, M.; Saniewski, M. Biosynthesis, Occurrence and Biological Properties Lycopene. Post Nauk Roln. 2000, 1, 29–46.
- Sariburun, E.; Şahin, S.; Demir, C.; Türkben, C.; Uylaşer, V. Phenolic Content and Antioxidant Activity of Raspberry and Blackberry Cultivars. J. Food Sci.. 2010, 75(4), C328–C335. DOI: https://doi.org/10.1111/j.1750-3841.2010.01571.x.
- Tiwari, U.; Cummins, E. Factors Influencing Levels of Phytochemicals in Selected Fruit and Vegetables during Pre-and Post-harvest Food Processing Operations. Food Res. Int.. 2013, 50, 497–506. DOI: https://doi.org/10.1016/j.foodres.2011.09.007.
- Lugasi, A.; Biro, L.; Hovarie, J.; Sagi, K. V.; Brand, S.; Barna, E. Lycopene Content of Foods and Lycopene Intake in Two Groups of the Hungarian Population. J. Nutr. Respiration. 2003, 23(8), 1035–1044.
- Liu, R. H. Potential Synergy of Phytochemicals in Cancer Prevention: Mechanism of Action. J. Nutr.. 2004, 134, 3479S–3485S. DOI: https://doi.org/10.1093/jn/134.12.3479S.
- Tapsell, L. C.; Hemphill, I.; Cobiac, L.; Patch, C. S.; Sullivan, D. R.; Fenesh, M.; Roodenrys, S.; Keogh, J. B.; Clifton, P. M.; Williams, P. G.; et al.. Health Benefits of Herbs and Spices the Past, the Present, the Future. Med. J. Australia. 2006, 185(2), 4–24.
- Wootton-Beard, P. C.; Ryan, L. Improving Public Health: The Role of Antioxidant-rich Fruit and Vegetable Beverages. Food Res. Int.. 2011, 44, 3133–3148. DOI: https://doi.org/10.1016/j.foodres.2011.09.015.
- Butt, M. S.; Sultan, M. T. In Nutritional Profile of Vegetable and Its Significance to Human Health. In Handbook of Vegetables and Vegetable Processing; Sinha, N. K., Hui, Y. H., Evranuz, E. O., Siddiq, M., Ahmed, J., Eds.; Blackwell Publishing: Iowa, 2011; pp pp. 107–123.
- Eckert, G. P. The Mediterranean Diet to Prevent Alzheimer’s Disease. Ernahrungs-Umschau. 2008, 55(8), 480–485.
- Harding, A. H.; Wareham, N. J.; Bingham, S. A.; Khaw, K.; Luben, R.; Welch, A. Plasma Vitamin C Level, Fruit and Vegetable Consumption, and the Risk of New Onset Type 2 Diabetes Mellitus: The European Prospective Investigation of Cancer – Norfolk Prospective Study. Arch. Internal. Med. 2008, 168(14), 1493–1499. DOI: https://doi.org/10.1001/archinte.168.14.1493.
- Rouanet, J. M.; Decorde, K.; Del Rio, D.; Auger, C.; Borges, G.; Cristol, J. P. Berry Juices, Teas, Antioxidants and the Prevention of Atherosclerosis in Hamsters. Food Chem.. 2010, 118(2), 266–271.
- Riboli, E.; Norat, T. Epidemiological Evidence of the Protective Effect of Fruit and Vegetables on Cancer Risk. Am. J. Clin. Nutr.. 2003, 78, 559S–769S. DOI: https://doi.org/10.1093/ajcn/78.3.559S.
- Schwager, J.; Mohajeri, M. H.; Fowler, A.; Weber, P. Challenges in Discovering Bioactives for the Food Industry. Curr. Opin. Biotechnol.. 2008, 19, 66–72. DOI: https://doi.org/10.1016/j.copbio.2008.02.016.
- Ares, G.; Gimenez, A.; Gambaro, A. Consumer Perceived Healthiness and Willingness to Try Functional Milk Dessert, Influence of Ingredient, Ingredient Name and Health Claim. Food Qual. Prefer.. 2009, 20(1), 50–56. DOI: https://doi.org/10.1016/j.foodqual.2008.07.002.
- Adlercreutz, H.; Phyto-oestrogens, M. W. Western Diseases. Annu. Med. 1997, 29(2), 95–120.
- Ostlund, R. E. Phytosterols in Human Nutrition. Annu. Rev. Nutr. 2002, 22, 533–549. DOI: https://doi.org/10.1146/annurev.nutr.22.020702.075220.
- Dutta, P. C. Phytosterols as Functional Food Components and Nutraceuticals; Mercel Dekker: New York, 2003; pp pp. 450.
- Baskar, R.; Shrisakthi, S.; Sathyapriya, B.; Shyampriya, R.; Nithya, R.; Poongodi, P. Antioxidant Potential of Peel Extracts of Banana Varieties (Musa Sapientum). Food Nutr. Sci.. 2011, 2, 1128–1133.
- Silva, B. A.; Ferreres, F.; Malva, J. O.; Dias, A. C. P. Phytochemical and Antioxidant Characterization of Hypericum Perforatum Alcoholic Extracts. Food Chem.. 2005, 90, 157–167. DOI: https://doi.org/10.1016/j.foodchem.2004.03.049.
- Jo, K. J.; Cha, M. R.; Lee, M. R.; Yoon, M. Y.; Park, H. R. Methanolic Extracts of Uncaria Rhynchophylla Induce Cytotoxicity and Apoptosis in HT-29 Human Colon Carcinoma Cells. Plant Food. Hum. Nutr. 2008, 63(2), 77–82. DOI: https://doi.org/10.1007/s11130-008-0074-z.
- SEDRI Dietary Reference Intakes: Proposed Definition and Plan for Review of Dietary Antioxidants and Related Compounds. In Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. National Academic Press: Washington DC; 1998. pp. pp. 24
- Patel, R. P.; McAndrew, J.; Sellak, H.; White, C. R.; Jo, H.; Freeman, B. A.; Darley-Usmar, V. M. Biological Aspects of Reactive Nitrogen Species. BBA Bioenergetics. 1999, 1411(2–3), 385–400. DOI: https://doi.org/10.1016/S0005-2728(99)00028-6.
- Castrol, L.; Freeman, B. A. Reactive Oxygen Species in Human Health and Disease. Nutr. 2001, 17(2), 161–165. DOI: https://doi.org/10.1016/S0899-9007(00)00570-0.
- Jaganath, I. B.; Mullen, W.; Edwards, C. A.; Crozier, A. The Relative Contribution of the Small and Large Intestine to the Absorption and Metabolism of Rutin in Man. Free Radical Res.. 2006, 40(10), 1035–1046. DOI: https://doi.org/10.1080/10715760600771400.
- Gerster, H. The Potential Role of Lycopene for Human Health. J. Am. Coll. Nutr.. 1997, 16, 109–126. DOI: https://doi.org/10.1080/07315724.1997.10718661.
- Bruno, E.J.; Ziegenfuss, T.N.; Landis, J. Vitamin C: Research Update. Curr. Sport Med. Rep. 2006, 5, 177–181.
- Li, Y. Antioxidants in Biology and Medicine: Essentials, Advances and Clinical Applications; Nova Science Publishers: New York, 2011; pp pp. 266–335.
- Lee, S. H.; Oe, T.; Blair, I. A. Vitamin C-induced Decomposition of Lipid Hydroperoxides to Endogenous Genotoxins. Science. 2001, 292, 2083–2086. DOI: https://doi.org/10.1126/science.1059501.
- Chen, Q.; Espey, M. G.; Sun, A. Y.; Pooput, C.; Kirk, K. L.; Krishna, M. C. Pharmacologic Doses of Ascorbate Act as a Prooxidant and Decrease Growth of Aggressive Tumor Xenografts in Mice. P. Natl. Acad. Sci. 2008, 105, 11105–11109. DOI: https://doi.org/10.1073/pnas.0804226105.
- Abdel-Wahab, Y. H.; O’Harte, F. P.; Mooney, M. H.; Barnett, C. R.; Flatt, P. R. Vitamin C Supplementation Decreases Insulin Glycation and Improves Glucose Homeostasis in Obese Hyperglycemic (Ob/ob) Mice. Metabolism. 2002, 51, 514–517. DOI: https://doi.org/10.1053/meta.2002.30528.
- Sen, C. K.; Khanna, S.; Roy, S. Tocotrienols in Health and Disease: The Other Half of the Natural Vitamin E Family. Mol. Aspects Med.. 2007, 28, 692–728. DOI: https://doi.org/10.1016/j.mam.2007.03.001.
- Cachia, O.; Benna, J. E.; Pedruzzi, E.; Descomps, B.; Gougerot-Pocidalo, M. A.; Leger, C. L. Alpha-tocopherol Inhibits the Respiratory Burst in Human Monocytes; Attenuation of P47 (Phox) Membrane Translocation and Phosphorylation. J. Biol. Chem.. 1998, 273, 32801–32805. DOI: https://doi.org/10.1074/jbc.273.49.32801.
- Traber, M. G.; Atkinson, J. Vitamin E Antioxidant and Nothing More. Free Radical Bio. Med. 2007, 43, 4–15. DOI: https://doi.org/10.1016/j.freeradbiomed.2007.03.024.
- McVean, M.; Liebler, D. C. Inhibition of UVB Induced DNA Photo Damage in Mouse Epidermis by Topically Applied Alpha-tocopherol. Carcinogenesis. 1997, 18, 1617–1622. DOI: https://doi.org/10.1093/carcin/18.8.1617.
- Manela-Azulay, M.; Bagatin, E. Cosmeceuticals Vitamins. Clin. Dermatol.. 2009, 7, 469–474. DOI: https://doi.org/10.1016/j.clindermatol.2009.05.010.
- Krinsky, N. I.; Johnson, E. J. Carotenoid Actions and Their Relation to Health and Disease. Mol. Aspects Med.. 2005, 26, 459–516. DOI: https://doi.org/10.1016/j.mam.2005.10.001.
- Stahl, W.; Sies, H. Bioactivity and Protective Effects of Natural Carotenoids. BBA-Mol. Basis Dis. 2005, 1740, 101–107. DOI: https://doi.org/10.1016/j.bbadis.2004.12.006.
- Ben-Dor, A.; Steiner, M.; Gheber, L.; Danilenko, M.; Dubi, N.; Linnewiel, K. Carotenoids Activate the Antioxidant Response Element Transcription System. Mol. Cancer Ther. 2005, 4, 177–186.
- Chew, B. P.; Brown, C. M.; Park, J. S.; Mixter, P. F. Dietary Lutein Inhibits Mouse Mammary Tumor Growth by Regulating Angiogenesis and Apoptosis. Anticancer Res.. 2003, 23, 3333–3339.
- Rao, A. V.; Rao, L. G. Carotenoids and Human Health. Pharmacol. Res. 2007, 55, 207–216. DOI: https://doi.org/10.1016/j.phrs.2007.01.012.
- Bennedsen, M.; Wang, X.; Willen, R.; Wadstrom, T.; Andersen, L. P. Treatment of H. Pylori Infected Mice with Antioxidant Astaxanthin Reduces Gastric Inflammation, Bacterial Load and Modulates Cytokine Release by Splenocytes. Immunol. Lett. 1999, 70, 185–189. DOI: https://doi.org/10.1016/S0165-2478(99)00145-5.
- Reifen, R.; Nur, T.; Matas, Z.; Halpern, Z. Lycopene Supplementation Attenuates the Inflammatory Status of Colitis in a Rat Model. Int. J. Vitamin Nutr. Res. 2001, 71, 347–351. DOI: https://doi.org/10.1024/0300-9831.71.6.347.
- Tapiero, H.; Townsend, D. M.; Tew, K. D. The Role of Carotenoids in the Prevention of Human Pathologies. Biomed. Pharmacother.. 2004, 58, 100–110. DOI: https://doi.org/10.1016/j.biopha.2003.12.006.
- Kim, J. H.; Kim, Y. S.; Song, G. G.; Park, J. J.; Chang, H. I. Protective Effect of Astaxanthin on Naproxen-induced Gastric Antral Ulceration in Rats. Eur. J. Pharmacol.. 2005, 514, 53–59. DOI: https://doi.org/10.1016/j.ejphar.2005.03.034.
- Pashkow, F. J.; Watumull, D. G.; Campbell, C. L. Astaxanthin: A Novel Potential Treatment for Oxidative Stress and Inflammation in Cardiovascular Disease. Am. J. Cardiol. 2008, 101, 58D–68D. DOI: https://doi.org/10.1016/j.amjcard.2008.02.010.
- Erdman, J. W.; Ford, N. A.; Lindshield, B. L. Are the Health Attributes of Lycopene Related to Its Antioxidant Function?. Arch Biochem. Biophys.. 2009, 483, 229–235. DOI: https://doi.org/10.1016/j.abb.2008.10.022.
- Fassett, R. G.; Coombes, J. S. Astaxanthin, Oxidative Stress, Inflammation and Cardiovascular Disease. Future Cardiol.. 2009, 5, 333–342. DOI: https://doi.org/10.2217/fca.09.19.
- Nishino, H.; Murakoshi, M.; Tokuda, H.; Satomi, Y. Cancer Prevention by Carotenoids. Arch. Biochem. Biophys. 2009, 483, 165–168. DOI: https://doi.org/10.1016/j.abb.2008.09.011.
- Stalikas, C. D. Phenolic Acids and Flavonoids: Occurrence and Analytical Methods. In Free Radicals and Antioxidant Protocols, Methods in Molecular Biology; Uppu, R. M., Murthy, S. N., Pryor, W. A., Parinandi, N. L., Eds.; Humana Press: New York, 2010; pp pp. 74.
- Scalbert, A.; Williamson, G. Dietary Intake and Bioavailability of Polyphenols. J. Nutr.. 2000, 130, 2073S–2085S. DOI: https://doi.org/10.1093/jn/130.8.2073S.
- Stevenson, D. E.; Hurst, R. D. Polyphenolic Phytochemicals-just Antioxidants or Much More?. Cell Mol. Life Sci. 2007, 64, 2900–2916. DOI: https://doi.org/10.1007/s00018-007-7237-1.
- Uppu, R. M.; Murthy, S. N.; Pryor, W. A.; Parinandi, N. L. Free Radicals and Antioxidant Protocols; Humana Press: New York, 2010; pp pp. 65.
- Bravo, L.; Mateos, R. Analysis of Flavonoids in Functional Foods and Nutraceuticals. In Methods of Analysis for Functional Foods and Nutraceuticals; Hurst, W. F., Ed.; CRC Press: Boca Raton, FL, 2008; pp pp. 149.
- Stoclet, J. C.; Chataigneau, T.; Ndiaye, M.; Oak, M. H.; El Bedoui, J.; Chataigneau, M. Vascular Protection by Dietary Polyphenols. Eur. J. Pharmacol.. 2004, 500, 299–313. DOI: https://doi.org/10.1016/j.ejphar.2004.07.034.
- Mann, G. E.; Rowlands, D. J.; Li, F. Y.; de Winter, P.; Siow, R. C. Activation of Endothelial Nitric Oxide Synthase by Dietary Isoflavones: Role of NO in Nrf2-mediated Antioxidant Gene Expression. Cardiovasc. Res. 2007, 75, 261–274. DOI: https://doi.org/10.1016/j.cardiores.2007.04.004.
- Andreadi, C. K.; Howells, L. M.; Atherfold, P. A.; Manson, M. M. Involvement of Nrf2, P38, B-Raf, and Nuclear factor-kappaB, but Not Phosphatidylinositol 3-kinase, in Induction of Hemeoxygenase-1 by Dietary Polyphenols. Mol. Pharmacol. 2006, 69, 1033–1040. DOI: https://doi.org/10.1124/mol.105.018374.
- Sang, S.; Shao, X.; Bai, N.; Lo, C. Y.; Yang, C. S.; Ho, C. T. Tea Polyphenol (-)-epigallocatechin-3-gallate: A New Trapping Agent of Reactive Dicarbonyl Species. Chem. Res. Toxicol.. 2007, 20, 1862–1870. DOI: https://doi.org/10.1021/tx700190s.
- Patel, R.; Maru, G. Polymeric Black Tea Polyphenols Induce Phase II Enzymes via Nrf2 in Mouse Liver and Lungs. Free Radical Bio. Med 2008, 44, 1897–1911. DOI: https://doi.org/10.1016/j.freeradbiomed.2008.02.006.
- Steffen, Y.; Gruber, C.; Schewe, T.; Sies, H. Mono-O-methylated Flavanols and Other Flavonoids as Inhibitors of Endothelial NADPH Oxidase. Arch. Biochem. Biophys. 2008, 469, 209–219. DOI: https://doi.org/10.1016/j.abb.2007.10.012.
- Babbar, N.; Oberoi, H. S.; Uppal, D. S.; Patil, R. T. Total Phenolic Content and Antioxidant Capacity of Extracts Obtained from Six Important Fruit Residues. Food Res. Int.. 2011, 44, 391–396. DOI: https://doi.org/10.1016/j.foodres.2010.10.001.
- Ghiselli, A.; Serafini, M.; Natella, F.; Scaccini, C. Total Antioxidant Capacity as a Tool to Assess Redox Status: Critical View and Experimental Data. Free Radical Bio. Med. 2000, 29(1), 1106–1114. DOI: https://doi.org/10.1016/S0891-5849(00)00394-4.
- Pulido, R.; Bravo, L.; Saura-Calixto, F. Antioxidant Activity of Dietary Polyphenols as Determined by a Modified Ferric Reducing/antioxidant Power Assay. J. Agr. Food Chem.. 2000, 48(8), 3396–3402. DOI: https://doi.org/10.1021/jf9913458.
- Chun, O. K.; Kim, D. O.; Smith, N.; Schroeder, D.; Han, J. T.; Lee, C. Y. Daily Consumption of Phenolics and Total Antioxidant Capacity from Fruits and Vegetables in the American Diet. J. Sci. Food Agr.. 2005, 85, 1715–1724. DOI: https://doi.org/10.1002/jsfa.2176.
- Floegel, A.; Kim, D.; Chung, S.; Koo, S. I.; Chun, O. K. Comparison of ABTS/DPPH Assays to Measure Antioxidant Capacity in Popular Antioxidant-rich US Foods. J. Food Compos. Anal.. 2011, 24, 1043–1048. DOI: https://doi.org/10.1016/j.jfca.2011.01.008.
- Cetkovic, G.; Canadanovic-Brunet, J.; Djilas, S.; Savatovic, S.; Mandic, A.; Tumbas, V. Assessment of Polyphenolic Content and in Vitro Antiradical Characteristics of Apple Pomace. Food Chem.. 2008, 109, 340–347. DOI: https://doi.org/10.1016/j.foodchem.2007.12.046.
- Almeida, M. M. B.; de Sousa, P. H., M.; Arriaga, A. M. C.; Do Prado, G. M.; Magalhaes, C. E.; Maia, G. A.; de Lemos, T. L. G. Bioactive Compounds and Antioxidant Activity of Fresh Exotic Fruits from Northeastern Brazil. Food Res. Int.. 2011, 44, 2155–2159.
- Dembitsky, V. M.; Poovarodom, S.; Leontowicz, H.; Leontowicz, M.; Vearasilp, S.; Trakhtenberg, S.; Gorinstein, S. The Multiple Nutrition Properties of Some Exotic Fruits: Biological Activity and Active Metabolites. Food Res. Int.. 2013, 44, 1671–1701. DOI: https://doi.org/10.1016/j.foodres.2011.03.003.
- de Souza, V. R.; Pereira, P. A. P.; da Silva, T. L. T.; Lima, L. C. O.; Pio, R.; Queiroz, F. Determination of the Bioactive Compounds, Antioxidant Activity and Chemical Composition of Brazilian Blackberry, Red Raspberry, Strawberry, Blueberry and Sweet Cherry Fruits. Food Chem.. 2014, 156, 362–368. DOI: https://doi.org/10.1016/j.foodchem.2014.01.125.
- Liu, R. H. Dietary Bioactive Compounds and Their Health Implications. J. Food Sci.. 2013, 78(S1), 18–25. DOI: https://doi.org/10.1111/1750-3841.12101.
- Haytowitz, D. B.; Bhagwat, S. USDA Database for the Oxygen Radical Absorbance Capacity (ORAC) of Selected Foods; U.S Department of Agriculture, 2010.
- Heim, K. E.; Tagliaferro, A. R.; Bobilya, D. J. Flavonoid Antioxidants: Chemistry Metabolism and Structure-activity Relationships. J. Nutr. Biochem.. 2002, 13(10), 572–584. DOI: https://doi.org/10.1016/S0955-2863(02)00208-5.
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food Sources and Bioavailability. Am. J. Clin. Nutr.. 2004, 79, 727–747.
- Mullen, W.; Boitier, A.; Stewart, A. J.; Crozier, A. Flavonoid Metabolites in Human Plasma and Urine after the Consumption of Red Onions: Analysis by Liquid Chromatography with Photodiode Array and Full Scan Tandem Mass Spectrometric Detection. J. Chromatogr. A. 2004, 1058, 163–168. DOI: https://doi.org/10.1016/S0021-9673(04)01476-1.
- Kroon, P. A.; Clifford, M. N.; Crozier, A.; Day, A. J.; Donovan, J. L.; Manach, C.; Williamson, G. How Should We Assess the Effects of Exposure to Dietary Polyphenols in Vitro?. Am.J. Clin. Nutr.. 2004, 80, 15–21.
- Williams, R. J.; Spencer, J. P. E.; Rice-Evans, C. Flavonoids: Antioxidants or Signalling Molecules?. Free Radical Biol. Med.. 2004, 36, 838–849. DOI: https://doi.org/10.1016/j.freeradbiomed.2004.01.001.
- Seger, R.; Krebs, E. G. The MAPK Signaling Cascade. J. Fed. Am. Soc. Exp. Biol. 1995, 9, 726–735.
- Kong, N.; Yu, R.; Chen, C.; Mandlekar, P. T. Signal Transduction Events Elicited by Natural Products: Role of MAPK and Caspase Pathways in Homeostatic Response and Induction of Apoptosis. Arch. Pharmacol. Res. 2000, 23, 1–16. DOI: https://doi.org/10.1007/BF02976458.
- Schroeter, H.; Spencer, J. P.; Rice-Evans, C.; Williams, R. J. Flavonoids Protect Neurons from Oxidized Low-density-lipoprotein-induced Apoptosis Involving c-Jun N-terminal Kinase (JNK), c-Jun and Caspase-3. Biochem. J.. 2001, 358, 547–557. DOI: https://doi.org/10.1042/bj3580547.
- Spencer, J. P.; Rice-Evans, C.; William, R. J. Modulation of Pro-survival Akt/protein Kinase B and ERK ½ Signalling Cascades by Quercetin and Its in Vivo Metabolites Underlie Their Action on Neuronal Viability. J. Biol. Chem.. 2003, 278, 34783–34793. DOI: https://doi.org/10.1074/jbc.M305063200.
- Pan, M.-H.; Lai, C.-S.; Dushenkov, S.; Ho, C.-T. Modulation of Inflammatory Genes by Natural Dietary Bioactive Compounds. J. Agric. Food Chem.. 2009, 57, 4467–4477. DOI: https://doi.org/10.1021/jf900612n.
- Fang, J.; Xia, C.; Cao, Z.; Zheng, J. Z.; Reed, E.; Jiang, B. H. Apigenin Inhibits VEGF and HIF-1 Expression via PI3K/AKT/p70S6K1 and HDM2/p53 Pathways. Faseb J.. 2005, 19(3), 342–353. DOI: https://doi.org/10.1096/fj.04-2175com.
- Nicholas, C.; Batra, S.; Vargo, M. A.; Voss, O. H.; Gavrilin, M. A.; Wewers, M. D.; Guttridge, D. C.; Grotewold, E.; Doseff, A. I. Apigenin Blocks Lipopolysaccharide-induced Lethality in Vivo and Proinflammatory Cytokines Expression by Inactivating NF-κB through the Suppression of P65 Phosphorylation. J. Immunol.. 2007, 179(10), 7121–7127. DOI: https://doi.org/10.4049/jimmunol.179.10.7121.
- Van Dross, R. T.; Hong, X.; Essengue, S.; Fischer, S. M.; Pelling, J. C. Modulation of UVB-induced and Basal Cyclooxygenase-2 (COX-2) Expression by Apigenin in Mouse Keratinocytes: Role of USF Transcription Factors. Mol. Carcinog. 2007, 46(4), 303–314. DOI: https://doi.org/10.1002/mc.20281.
- Chen, K. H.; Weng, M. S.; Lin, J. K. Tangeretin Suppresses IL-1β-induced Cyclooxygenase (COX)-2 Expression through Inhibition of P38 MAPK, JNK, and AKT Activation in Human Lung Carcinoma Cells. Biochem. Pharmacol.. 2007, 73(2), 215–227. DOI: https://doi.org/10.1016/j.bcp.2006.09.018.
- Hamalainen, M.; Nieminen, R.; Vuorela, P.; Heinonen, M.; Moilanen, E. Anti-inflammatory Effects of Flavonoids: Genistein, Kaempferol, Quercetin, and Daidzein Inhibit STAT-1 and NF-κB Activations, Whereas Flavone, Isorhamnetin, Naringenin, and Pelargonidin Inhibit Only NF-κB Activation along with Their Inhibitory Effect on iNOS Expression and NO Production in Activated Macrophages. Mediators Inflamm. 2007, 2007, 45673. DOI: https://doi.org/10.1155/2007/45673.
- Crespo, I.; Garcia-Mediavilla, M. V.; Gutierrez, B.; Sanchez-Campos, S.; Tunon, M. J.; Gonzalez-Gallego, J. A Comparison of the Effects of Kaempferol and Quercetin on Cytokine-induced Pro-inflammatory Status of Cultured Human Endothelial Cells. Br. J. Nutr.. 2008, 100(5), 968–976. DOI: https://doi.org/10.1017/S0007114508966083.
- Park, H. H.; Lee, S.; Son, H. Y.; Park, S. B.; Kim, M. S.; Choi, E. J.; Singh, T. S.; Ha, J. H.; Lee, M. G.; Kim, J. E.; et al.. Flavonoids Inhibit Histamine Release and Expression of Proinflammatory Cytokines in Mast Cells. Arch. Pharm. Res.. 2008, 31(10), 1303–1311.
- Ruiz, P. A.; Braune, A.; Holzlwimmer, G.; Quintanilla-Fend, L.; Haller, D. Quercetin Inhibits TNF-induced NF-κB Transcription Factor Recruitment to Proinflammatory Gene Promoters in Murine Intestinal Epithelial Cells. J. Nutr.. 2007, 137(5), 1208–1215. DOI: https://doi.org/10.1093/jn/137.5.1208.
- Torres-Piedra, M.; Ortiz-Andrade, R.; Villalobos-Molina, R.; et al.. A Comparative Study of Flavonoid Analogues on Streptozotocin-nicotinamide Induced Diabetic Rats: Quercetin as A Potential Antidiabetic Agent Acting via 11β-hydroxysteroid Dehydrogenase Type 1 Inhibition. Eur. J. Med. Chem.. 2010, 45(6), 2606–2612.
- Vinholes, J.; Gelain, D. P.; Vizzotto, M. Stone Fruits as a Source of Bioactive Compounds. In Natural Bioactive Compounds from Fruits and Vegetables as Health Promoters Part I; da Silva, L. R., Silva, B., Eds.; Bentham Science Publishers: Sharjah, UAE, 2016; pp pp110–142.
- Han, X.; Pan, J.; Ren, D.; Cheng, Y.; Fan, P.; Lou, H. Naringenin-7-O-glucoside Protects against Doxorubicin-induced Toxicity in H9c2 Cardiomyocytes by Induction of Endogenous Antioxidant Enzymes. Food Chem. Toxicol. 2008, 46(9), 3140–3146. DOI: https://doi.org/10.1016/j.fct.2008.06.086.
- Han, X. Z.; Gao, S.; Cheng, Y. N.; et al.. Protective Effect of naringenin-7-O-glucoside against Oxidative Stress Induced by Doxorubicin in H9c2 Cardiomyocytes. Biosci. Trends. 2012, 6(1), 19–25.
- Ding, M.; Feng, R.; Wang, S. Y.; Bowman, L.; Lu, Y.; Qian, Y.; Castranova, V.; Jiang, B. H.; Shi, X. Cyanidin-3-glucoside, a Natural Product Derived from Blackberry, Exhibits Chemopreventive and Chemotherapeutic Activity. J. Biol. Chem.. 2006, 281(25), 17359–17368. DOI: https://doi.org/10.1074/jbc.M600861200.
- Adisakwattana, S.; Yibchok-Anun, S.; Charoenlertkul, P.; Wongsasiripat, N. Cyanidin-3-rutinoside Alleviates Postprandial Hyperglycemia and Its Synergism with Acarbose by Inhibition of Intestinal α-glucosidase. J. Clin. Biochem. Nutr. 2011, 49(1), 36–41. DOI: https://doi.org/10.3164/jcbn.10-116.
- Lamy, S.; Beaulieu, E.; Labbe, D.; Bedard, V.; Moghrabi, A.; Barrette, S.; Gingras, D.; Beliveau, R. Delphinidin, a Dietary Anthocyanidin, Inhibits Platelet-derived Growth Factor Ligand/receptor (PDGF/PDGFR) Signaling. Carcinogenesis. 2008, 29(5), 1033–1041. DOI: https://doi.org/10.1093/carcin/bgn070.
- Elisia, I.; Kitts, D. D. Anthocyanins Inhibit Peroxyl Radical-induced Apoptosis in Caco-2 Cells. Mol. Cell Biochem. 2008, 312(1–2), 139–145. DOI: https://doi.org/10.1007/s11010-008-9729-1.
- Xu, M.; Bower, K. A.; Wang, S.; et al.. Cyanidin-3-glucoside Inhibits Ethanol-induced Invasion of Breast Cancer Cells Overexpressing ErbB2. Mol. Cancer. 2010, 9(1), 285.
- Lim, T.-G.; Kwon, J. Y.; Kim, J.; et al.. Cyanidin-3-glucoside Suppresses B[a]PDE-induced Cyclooxygenase-2 Expression by Directly Inhibiting Fyn Kinase Activity. Biochem. Pharmacol.. 2011, 82(2), 167–174.
- Wang, Q.; Xia, M.; Liu, C.; et al.. Cyanidin-3-O-β-glucoside Inhibits iNOS and COX-2 Expression by Inducing Liver X Receptor Alpha Activation in THP-1 Macrophages.. Life Sci.. 2008, 83(5–6), 176–184.
- Zhang, Y.; Lian, F.; Zhu, Y.; et al.. Cyanidin-3-O-β-glucoside Inhibits LPS-induced Expression of Inflammatory Mediators through Decreasing IkappaBalpha Phosphorylation in THP-1 Cells. Inflamm. Res.. 2010, 59(9), 723–730.
- Min, S.-W.; Ryu, S.-N.; Kim, D.-H. Anti-inflammatory Effects of Black Rice, cyanidin-3-O-β-D-glycoside, and Its Metabolites, Cyanidin and Protocatechuic Acid. Int. Immunopharmacol.. 2010, 10(8), 959–966. DOI: https://doi.org/10.1016/j.intimp.2010.05.009.
- Hassimotto, N. M. A.; Moreira, V.; G-d, N. N.; Souto, P.-C. M. D.-C.; Teixeira, C.; Lajolo, F. M. Inhibition of Carrageenan-induced Acute Inflammation in Mice by Oral Administration of Anthocyanin Mixture from Wild Mulberry and Cyanidin-3-glucoside. Bio. Med. Res. Int. 2013, 2013, 10.
- De, S. D.; Maiuri, M. C.; Simeon, V.; Grassia, G.; Soscia, A.; Cinelli, M. P.; Carnuccio, R. Lycopene, Quercetin and Tyrosol Prevent Macrophage Activation Induced by Gliadin and IFN-γ. Eur. J. Pharmacol.. 2007, 566(1–3), 192–199. DOI: https://doi.org/10.1016/j.ejphar.2007.03.051.
- Kim, G. Y.; Kim, J. H.; Ahn, S. C.; Lee, H. J.; Moon, D. O.; Lee, C. M.; Park, Y. M. Lycopene Suppresses the Lipopolysaccharide-induced Phenotypic and Functional Maturation of Murine Dendritic Cells through Inhibition of Mitogen-activated Protein Kinases and Nuclear factor-κB. Immunology. 2004, 113(2), 203–211. DOI: https://doi.org/10.1111/j.1365-2567.2004.01945.x.
- Bai, S. K.; Lee, S. J.; Na, H. J.; Ha, K. S.; Han, J. A.; Lee, H.; Kwon, Y. G.; Chung, C. K.; Kim, Y. M. β-Carotene Inhibits Inflammatory Gene Expression in Lipopolysaccharide-stimulated Macrophages by Suppressing Redox-based NF-κB Activation. Exp. Mol. Med. 2005, 37(4), 323–334. DOI: https://doi.org/10.1038/emm.2005.42.
- Kim, J. H.; Na, H. J.; Kim, C. K.; Kim, J. Y.; Ha, K. S.; Lee, H.; Chung, H. T.; Kwon, H. J.; Kwon, Y. G.; Kim, Y. M. The Nonprovitamin A Carotenoid, Lutein, Inhibits NF-κB-dependent Gene Expression through Redox-based Regulation of the Phosphatidylinositol 3-kinase/PTEN/Akt and NF-κB-inducing Kinase Pathways: Role of H(2)O(2) in NF-κB Activation. Free Radical Biol. Med.. 2008, 45(6), 885–896. DOI: https://doi.org/10.1016/j.freeradbiomed.2008.06.019.
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Thun, M. J. Cancer Statistics CA. Cancer J. Clin. 2009, 59(4), 225–249. DOI: https://doi.org/10.3322/caac.20006.
- Lee, C. Y.; Smith, N. L. Apples: An Important Source of Antioxidants in the American Diet. New York Fruit Quart. 2000, 8, 8–10.
- Wolfe, K.; Wu, X.; Liu, R. H. Antioxidant Activity of Apple Peels. J. Agr. Food Chem.. 2003, 51, 609–614. DOI: https://doi.org/10.1021/jf020782a.
- Willett, W. C. Balancing Life-style and Genomics Research for Disease Prevention. Sci. 2002, 296(5568), 695–698. DOI: https://doi.org/10.1126/science.1071055.
- Amiot, M. J.; Lairon, D. Fruit and Vegetables, Cardiovascular Diseases, Diabetes and Obesity. In Improving the Health-promoting Properties of Fruit and Vegetable Products; Tomas-Barberan, F. A., Gil, M. I., Eds.; Woodhead Publishing: Cambridge, 2008; pp pp. 96–118.
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M. T. D.; Mazura, M.; Telserd, J. Free Radicals and Antioxidants in Normal Physiological Functions and Human Disease. Int. J. Biochem. Cell Biol.. 2007, 39(1), 44–84. DOI: https://doi.org/10.1016/j.biocel.2006.07.001.
- Hsing, A. W.; Chokkalingam, A. P.; Gao, Y. T.; Madigan, M. P.; Deng, J.; Gridley, G.; Fraumeni, J. F. Allium Vegetables and Risk of Prostate Cancer: A Population-based Study. J. Nat. Cancer Inst. 2002, 94(21), 1648–1651. DOI: https://doi.org/10.1093/jnci/94.21.1648.
- Zhang, Y.; Yao, S.; Li, J. Vegetable-derived Isothiocyanates: Anti-proliferative Activity and Mechanism of Action. Process Nutr. Soc. 2006, 65(1), 68–75. DOI: https://doi.org/10.1079/PNS2005475.
- Hollman, P. C. H.; Kitan, M. B. Dietary Flavonoids: Intake Health Effects and Bioavailability. Food Chem. Toxicol. 1999, 37(9–10), 937–942. DOI: https://doi.org/10.1016/S0278-6915(99)00079-4.
- Piironen, V.; Toivo, J.; Puupponen-Pimia, R.; Lampi, A. M. Plant Sterols in Vegetables, Fruits and Berries. J. Sci. Food Agr.. 2003, 83(4), 330–337. DOI: https://doi.org/10.1002/jsfa.1316.
- Murphy, P. A.; Hendrich, S. Phytoestrogens in Foods. Adv. Food Nutr. Res. 2002, 44, 195–246.
- Webb, A. L.; McCullough, M. L. Dietary Lignans: Potential Role in Cancer Prevention. Nutr. Cancer. 2005, 51(20), 117–131. DOI: https://doi.org/10.1207/s15327914nc5102_1.
- Ignat, I.; Volf, I.; Popa, V. I. A Critical Review of Methods for Characterisation of Polyphenolic Compounds in Fruits and Vegetables. Food Chem. 2011, 126, 1821–1835. DOI: https://doi.org/10.1016/j.foodchem.2010.12.026.
- Yalcin, H.; Çapar, T. D. Bioactive Compounds of Fruits and Vegetables. In Minimally Processed Refrigerated Fruits and Vegetables, Second Edition. Food Engineering Series; Yildiz, F., Wiley, R. C., Eds.; Springer Nature: New York, USA, 2017; pp pp. 723–745.
- Heinonen, S.; Nurmi, T.; Liukkonen, K.; Poutanen, K.; Wähälä, K.; Deyama, T.; Adlercreutz, H. In Vitro Metabolism of Plant Lignans: New Precursors of Mammalian Lignans Enterolactone and Enterodiol. J. Agric. Food Chem.. 2001, 49, 3178–3186. DOI: https://doi.org/10.1021/jf010038a.
- Smeds, A. I.; Eklund, P. C.; Sjöholm, R. E.; Willför, S. M.; Nishibe, S.; Deyama, T.; Holmbom, B. R. Quantification of a Broad Spectrum of Lignans in Cereals, Oilseeds, and Nuts. J. Agric. Food Chem.. 2007, 55(4), 1337–1346. DOI: https://doi.org/10.1021/jf0629134.
- Peñalvo, J. L.; Heinonen, S.-M.; Aura, A.-M.; Adlercreutz, H. Dietary Sesamin Is Converted to Enterolactone in Humans. J. Nutr.. 2005, 135(5), 1056–1062. DOI: https://doi.org/10.1093/jn/135.5.1056.
- Bradford, P. G.; Awad, A. B. Phytosterols as Anticancer Compounds. Mol. Nutr. Food Res.. 2007, 51(2), 161–170. DOI: https://doi.org/10.1002/mnfr.200600164.
- Prasad, K. Hypocholesterolemic and Antiatherosclerotic Effect of Flax Lignan Complex Isolated from Flaxseed. Atherosclerosis. 2005, 179(2), 269–275. DOI: https://doi.org/10.1016/j.atherosclerosis.2004.11.012.
- Saleem, M.; Kim, H. J.; Ali, M. S.; Lee, Y. S. An Update on Bioactive Plant Lignans. Nat. Prod. Rep.. 2005, 22(6), 696–716. DOI: https://doi.org/10.1039/b514045p.
- Johnsen, N. F.; Olsen, A.; Thomsen, B. L. R.; Christensen, J.; Egeberg, R.; Knudsen, K. E. B.; Loft, S.; Overvad, K.; Tjønneland, A. Plasma Enterolactone and Risk of Colon and Rectal Cancer in a Case–cohort Study of Danish Men and Women. Cancer Causes Control. 2010, 21(1), 153–162. DOI: https://doi.org/10.1007/s10552-009-9445-5.
- Brigelius-Flohe, R.; Trebar, M. G. Vitamin E: Function and Metabolism. J. Am. Soc. Exp. Biol. 1999, 13(10), 1145–1155.
- Boyer, J.; Liu, R. H. Apple Phytochemicals and Their Health Benefits. Nutr. J.. 2004, 3(1), 5. DOI: https://doi.org/10.1186/1475-2891-3-5.
- Liu, R. H.; Liu, J.; Chen, B. Apples Prevent Mammary Tumors in Rats. J. Agr. Food Chem.. 2005, 53, 2341–2343. DOI: https://doi.org/10.1021/jf058010c.
- Serfaty, C. A.; Oliveira-Silva, P.; Melibeu, A. D. F.; Campello-Costa, P. Nutritional Tryptophan Restriction and the Role of Serotonin in Development and Plasticity of Central Visual Connections. Neuro. Immuno. Modulat. 2008, 15, 170–175.
- Meyers, K. J.; Watkins, C. B.; Pritts, M. P.; Liu, R. H. Antioxidant and Antiproliferative Activities of Strawberries. J. Agr. Food Chem.. 2003, 51, 6887–6892. DOI: https://doi.org/10.1021/jf034506n.
- Wang, S. Y.; Feng, R.; Lu, Y.; Bowman, L.; Ding, M. Inhibitory Effect on Activator Protein-1, Nuclear Factor-kappaB, and Cell Transformation by Extracts of Strawberries (Fragaria × Ananassa Duch. J. Agr. Food Chem.. 2005, 53, 4187–4193. DOI: https://doi.org/10.1021/jf0478049.
- Benavente-Garcia, O.; Castillo, J.; Marin, F. R.; Ortuno, A.; Del Rio, J. A. Uses and Properties of Citrus Flavonoids. J. Agr. Food Chem.. 1997, 45, 4505–4515. DOI: https://doi.org/10.1021/jf970373s.
- Li, S.; Pan, M. H.; Lo, C. Y.; Tan, D.; Wang, Y.; Shahidi, F.; Ho, C. T. Chemistry and Health Effects of Polymethoxyflavones and Hydroxylated Polymethoxyflavones. J. Funct. Foods. 2009, 1, 2–12. DOI: https://doi.org/10.1016/j.jff.2008.09.003.
- Frankel, E. N.; Bosanek, C. A.; Mayer, A. S.; Silliman, K.; Kirk, L. L. Commercial Grape Juices Inhibit the in Vitro Oxidation of Human Low-density Lipoproteins. J. Agr. Food Chem.. 1998, 46, 834–838. DOI: https://doi.org/10.1021/jf9707952.
- Downey, M. O.; Dokoozlian, N. K.; Krstic, M. P. Cultural Practice and Environmental Impacts on the Flavonoid Composition of Grapes and Wine: A Review of Recent Research. Am. J. Enol. Viticult. 2006, 57, 257–268.
- Ferrara, L. The Dietary Importance of a Tropical Fruit: The Kiwano. Ingredienti. Aliment. 2006, 5, 14–17.
- Pero, R. W. Medicinal Compositions of Salts, Chelates And/or Free Acids of α-hydroxy Organic Acids, Especially Quinic Acid. PCT Int. Appl. WO/2006/101922. publication date 28.Sep.2006.
- Kalandiya, A.; Vanidze, M.; Papunidze, S.; Chkhikvishvili, I.; Shalashvili, A. Flavonols from Chinese Actinidia (Actinidia Chinensis Planch.). Fruits. B. Georgian Acad. Sci. 2001, 163, 157–159.
- Gil, M. I.; Aguayo, E.; Kader, A. A. Quality Changes and Nutrient Retention in Fresh-cut versus Whole Fruits during Storage. J. Agr. Food Chem.. 2006, 54, 4284–4296. DOI: https://doi.org/10.1021/jf060303y.
- Masibo, M.; He, Q. Major Mango Polyphenols and Their Potential Significance to Human Health. Compr. Rev. Food Sci. F. 2008, 7, 309–319. DOI: https://doi.org/10.1111/j.1541-4337.2008.00047.x.
- Giordani, E.; Doumett, S.; Nin, S.; Del Bubba, M. Selected Primary and Secondary Metabolites in Fresh Persimmon (Diospyros Kaki Thunb.): A Review of Analytical Methods and Current Knowledge of Fruit Composition and Health Benefits. Food Res. Int.. 2011, 44, 1752–1767. DOI: https://doi.org/10.1016/j.foodres.2011.01.036.
- Yi, X.; Wei, B.; Teng, J.; Gao, C. Determination of the Phenolic Compounds in Pineapple by High-performance Liquid Chromatography. Shipin Yu Fajiao Gongye. 2006, 32, 99–101.
- Stintzing, F. C.; Schieber, A.; Carle, R. Identification of Betalains from Yellow Beet (Beta Vulgaris L.) And Cactus Pear (Opuntia Ficus-indica L. MilL.) By High-performance Liquid Chromatography–electrospray Ionization Mass Spectroscopy. J. Agr. Food Chem.. 2002, 50, 2302–2307.
- Castellanos-Santiago, E.; Yahia, E. M. Identification and Quantification of Betalains from the Fruits of 10 Mexican Prickly Pear Cultivars by High-performance Liquid Chromatography and Electron Spray Ionization Mass Spectrometry. J. Agr. Food Chem.. 2008, 56, 5758–5764. DOI: https://doi.org/10.1021/jf800362t.
- Shui, G.; Leong, L. P. Residue from Star Fruit as Valuable Source for Functional Food Ingredients and Antioxidant Nutraceuticals. Free Radical Biol. Med.. 2004a, 36, S132.
- Shui, G.; Leong, L. P. Analysis of Polyphenolic Antioxidants in Star Fruit Using Liquid Chromatography and Mass Spectrometry. J. Chromatogr. A. 2004b, 1022, 67–75. DOI: https://doi.org/10.1016/j.chroma.2003.09.055.
- Robards, K.; Prenzler, P. D.; Tucker, G.; Swatsitang, P.; Glover, W. Phenolic Compounds and Their Role in Oxidative Processes in Fruits. Food Chem.. 1999, 66, 401–436. DOI: https://doi.org/10.1016/S0308-8146(99)00093-X.
- Lin, X. Y.; Liu, J. Z.; Milner, J. A. Dietary Garlic Suppresses DNA Adducts Caused by N Nitroso Compounds. Carcinogenesis. 1994, 15, 349–352. DOI: https://doi.org/10.1093/carcin/15.2.349.
- Lee, E. S.; Steiner, M.; Lin, R. Thioallyl Compounds: Potent Inhibitors of Cell Proliferation. BBA-Mol. Cell Res. 1994, 1221, 73–77.
- Kahkonen, M. P.; Hopia, A. I.; Vurela, H. J.; Rauha, J.-P.; Pihlaja, K.; Kujala, T. S.; Heinon, M. Antioxidant Activity of Plant Extracts Containing Phenolic Compounds. J. Agr. Food Chem.. 1999, 47, 3954–3962. DOI: https://doi.org/10.1021/jf990146l.
- Yahia, E. M. The Contribution of Fruit and Vegetable Consumption to Human Health. In Fruit and Vegetable Phytochemicals: Chemistry, Nutritional Value, and Stability; de la Rosa, L. A., Alvarez-Parrilla, E., Gonzalez-Aguilar, G. A., Eds.; Wiley-Blackwell: Iowa, 2010; pp pp. 3–51.
- Plumb, G. W.; Chambers, S. J.; Lambert, N.; Wanigatunga, S.; Williamson, G. Influence of Fruit and Vegetable Extracts on Lipid Peroxidation in Microsomes Containing Specific Cytochrome P450s. Food Chem.. 1997a, 60, 161–164. DOI: https://doi.org/10.1016/S0308-8146(95)00256-1.
- Plumb, G. W.; Price, K. R.; Rhodes, M. J. C.; Williamson, G. Antioxidant Properties of the Major Polyphenolic Compounds in Broccoli. Free Radical Res.. 1997b, 27, 429–435. DOI: https://doi.org/10.3109/10715769709065782.
- Minguez-Mosquera, M. I.; Hornero-Mendez, D. Comparative Study of the Effect of Paprika Processing on the Carotenoids in Peppers (Capsicum Annuum) of the Bola and Agridulce Varieties. J. Agr. Food Chem.. 1994, 42, 1555–1560. DOI: https://doi.org/10.1021/jf00043a031.
- Markus, F.; Daood, H. G.; Kapitany, J.; Biacs, P. A. Changes in the Carotenoid and Antioxidant Content of Spice Red Pepper (Paprika) as a Function of Ripening and Some Technological Factors. J. Agr. Food Chem.. 1999, 47, 100–107. DOI: https://doi.org/10.1021/jf980485z.
- Al-Saikhan, M. S.; Howard, L. R.; Miller, J. C. Antioxidant Activity and Total Phenolics in Different Genotypes of Potato (Solanum Tuberosum L). J. Food Sci.. 1995, 60, 341–343. DOI: https://doi.org/10.1111/j.1365-2621.1995.tb05668.x.
- Velioglu, Y. S.; Mazza, G.; Gao, L.; Oomah, D. B. Antioxidant Activity and Total Phenolics in Selected Fruits, Vegetables, and Grain Products. J. Agr. Food Chem.. 1998, 46, 4113–4117. DOI: https://doi.org/10.1021/jf9801973.
- Dorais, M.; Papadopoulos, A. P.; Gosselin, A. Greenhouse Tomato Fruit Quality. Hort. Rev. 2001a, 26, 239–319.
- Dorais, M.; Papadopoulos, A. P.; Gosselin, A. Influence of Electric Conductivity Management on Greenhouse Tomato Yield and Fruit Quality. Agronomie. 2001b, 21(4), 367–383. DOI: https://doi.org/10.1051/agro:2001130.
- Yahia, E. M.; Brecht, J. K. Tomato. Chapter 2. In Crop Postharvest: Science and Technology; Rees, D., Farrell, G., Orchard, J. E., Eds.; Wiley-Blackwell: Oxford, 2009; Vol. ume 3, pp pp. 34.
- Sanjust, E.; Mocci, G.; Zucca, P.; Rescigno, A. Mediterranean Shrubs as Potential Antioxidant Sources. Nat. Prod. Res. 2008, 22(8), 689–708. DOI: https://doi.org/10.1080/14786410801997125.
- Toure, A.; Xu, X.; Michel, T.; Bangoura, M. In Vitro Antioxidant and Radical Scavenging of Guinean Kinkeliba Leaf (Combretum Micranthum G. Don) Extracts. Nat. Prod. Res. 2011, 25(11), 1025–1036.
- Gao, X.; Ohlander, M.; Jeppsson, N.; Bjork, L.; Trajkovski, V. Changes in Antioxidant Effects and Their Relationship to Phytonutrients in Fruits of Sea Buckthorn (Hippophae Rhamnoides L.) During Maturation. J. Agr. Food Chem.. 2000, 48(5), 1485–1490. DOI: https://doi.org/10.1021/jf991072g.
- Biesalski, H. K. Polyphenols and Inflammation: Basic Interactions. Curr. Opin. Clin. Nutr. Met. Care. 2007, 10, 724–728. DOI: https://doi.org/10.1097/MCO.0b013e3282f0cef2.
- Fan, Z.; Wang, Z.; Liu, J. Cold-field Fruit Extracts Exert Different Antioxidant and Antiproliferative Activities. In Vitro. Food Chem. 2011, 129, 402–407. DOI: https://doi.org/10.1016/j.foodchem.2011.04.091.
- Umezuruike, L. O.; Majeed, R. A. Antioxidant Contents of Pre-packed Fresh-cut versus Whole Fruit and Vegetables. Brit. Food J. 2010, 112(8), 797–810. DOI: https://doi.org/10.1108/00070701011067424.
- Sagar, N. A.; Pareek, S.; Sharma, S.; Yahia, E. M.; Lobo, M. G. Fruit and Vegetable Waste: Bioactive Compounds, Their Extraction, and Possible Utilization. Compr. Rev. Food Sci. Food Saf.. 2018, 17, 512–531.
- Saini, A.; Panesar, P. S.; Bera, M. B. Valorization of Fruits and Vegetables Waste through Green Extraction of Bioactive Compounds and Their Nanoemulsions‑based Delivery System. Bioresour. Bioprocess.. 2019, 6, 26. DOI: https://doi.org/10.1186/s40643-019-0261-9.
- Carocho, M.; Ferreira, C. F. R. A Review on Antioxidants, Prooxidants and Related Controversy: Natural and Synthetic Compounds, Screening and Analysis Methodologies and Future Perspectives. Food. Chem. Toxicol. 2013, 51, 15–25. DOI: https://doi.org/10.1016/j.fct.2012.09.021.
- Suntres, Z. E. Liposomal Antioxidants for Protection against Oxidant-induced Damage. J. Toxicol. 2011. DOI: https://doi.org/10.1155/2011/152474.
- Soquetta, M. B.; Terra, L. D.-M.; C. P, B. Green Technologies for the Extraction of Bioactive Compounds in Fruits and Vegetables. Cyta. J. Food. 2018, 16(1), 400–412.