Abstract
The stability of camellia oil (saturated fatty acid: monounsaturated fatty acid: polyunsaturated fatty acid = 1:7:1) after frying potatoes was compared with palm oil (saturated fatty acid: monounsaturated fatty acid: polyunsaturated fatty acid = 4:4:1) and peanut oil (saturated fatty acid: monounsaturated fatty acid: polyunsaturated fatty acid = 2:4:4). Oil samples were evaluated for acid value, iodine value, peroxide value, p-anisidine value, total oxidation value, tocopherols content, and fatty acids composition. There was the least change in fatty acid composition in camellia oil among the three edible oils. The α-tocopherol was more vulnerable to heat degradation than γ-tocopherol and δ-tocopherol, and α-tocopherol was completely degraded before the whole frying process was done for palm and peanut oils. The oxidative stabilities of the three edible oils were in the order of camellia oil > palm oil > peanut oil. The oxidative stability was mainly determined by the calculated oxidizability value related to fatty acid composition, and when calculated oxidizability values were similar, the tocopherol contents of edible oils would be a key factor in affecting their oxidative stabilities.
INTRODUCTION
Frying is a fast and convenient technique for the production of foods with sensory properties that are favored by consumers, including a typical flavor, color, taste, and a crispy surface.[Citation1] However, it may result in a high rate of peroxides degradation because of high temperature, and contribute to radical-radical polymerization reactions and formation of non-polar dimmers and oligomers because of low availability of oxygen.[Citation2] These chemical reactions are mainly influenced by the type and quality of the oil, the food properties, and the food/oil ratio and the frying temperature.[Citation3,Citation4]
Changes occurred in oils after frying food have been extensively reported. Several studies have verified the variation tendency of different physical and chemical parameters with the increasing frying cycles. The free fatty acids, peroxide value (POV), p-anisidine value (p-AV), total polar compounds (TPC) of the oils increased gradually under the frying conditions.[Citation5,Citation6] The total phenols, tocopherols, and fatty acids composition can affect the stability of oils against oxidation and decomposition in the frying process.[Citation6,Citation7] Abdulkarim[Citation8] evaluated the performance of the high-oleic Moringa oleifera seed oil saturated fatty acid (SFA; SFA: monounsaturated fatty acid [MUFA]: polyunsaturated fatty acid [PUFA] = 2:7:0) in deep-frying by comparing its frying stability with other conventional frying oils (canola [SFA: MUFA: PUFA = 1:6:3], soybean [SFA: MUFA: PUFA = 1.5:2.5:6], and palm olein [SFA: MUFA: PUFA = 4:4:1]). The free fatty acids of these four edible oils increased 66.6, 71.4, 60.0, and 65.0%, respectively. And the changes in p-AV and TOTOX value of Moringa oleifera seed oil were found to be significantly lower than other oils, followed by palm olein oil, with the highest values obtained in canola oil and soybean oil. Juárez[Citation9] investigated the discontinuous deep frying of potatoes, milanesas, and churros in soybean oil, sunflower oil, and partially hydrogenated fats. After 80.5 h of deep-frying churros, all the oils exceeded 25% of TPC and the contents of polymeric and dimerized triacylglycerols were higher than 10% and the losses of tocopherols during frying reached 76.0%.
The oxidative stabilities of different edible oils during the frying cycles were dependent upon the fatty acids composition, particularly the unsaturated degree, and the content of antioxidants (such as tocopherols) in the oil.[Citation10] There were several articles reporting the flavor and storage stability of potato chips after frying in different oils.[Citation11,Citation12] The camellia oil, rich in oleic acid (over 70%) and tocopherol, has become more and more popular in China. However, little has been known regarding the oxidative stability of camellia oil compared with common edible oils with different fatty acids composition and tocopherol contents after frying potatoes. Therefore, the main objective of present work was to determine the oxidative stability of camellia oil (SFA: MUFA: PUFA = 1:7:1) after frying potatoes and make comparison with palm oil (SFA: MUFA: PUFA = 4:4:1) and peanut oil (SFA: MUFA: PUFA = 2:4:4) by the indicators of IV, POV, p-AV, and TOTOX, and to investigate effect of tocopherols content and fatty acids composition on the oils’ oxidative stabilities.
MATERIALS AND METHODS
Three edible oils, namely palm oil, peanut oil, camellia oil (with no antioxidants added) were purchased from l Edible Oil LTD Nanchang, China. Pre-fried potatoes approximately 10 cm long and 1 cm thick were purchased from a local supermarket.
Standard fatty acid methyl esters (FAME, #463) spiked with a mixture of four positional CLA isomers (#UC-59 M) were obtained from Nu-Chek Prep Inc. (Elysian, MN). n-hexane used in a gas chromatograph (GC) was purchased from Merck (Darmstadt, Germany), and other solvents were of analytical reagent grade. All experiments and analytical determinations were performed at least in triplicate.
Frying Procedure
Deep frying was carried out in an electrical fryer (Multi-function Fryer, Guangzhou, China). The temperature of the oil in the fryer was monitored by a thermo recorder. It was equipped with a thermostat and supplied with an insert cross-linked steel wire-mesh which allowed the food to be dipped into the oil, without coming in contact with the fryer’s inner surface. The ratio of potatoes to frying oil was set at 1/25 (w/v), in the first frying batch, 80 g potatoes were deeply fried for 3 min at 170°C in 2000 mL oil, and every frying batch was conducted for 5 min. After each of the five frying cycles, 50 mL of oil and all potatoes were taken out and stored at –20°C until analysis.[Citation13] Frying experimental series consisted of 75 frying batches conducted in three consecutive days. At the end of the experiment, there was 1002 mL oil left.
Determination of Chemical Parameters
Acid value (AV)
AV was determined according to AOCS Official Method Ca 5a–40. The titration of an accurate sample solution, dissolved in ethanol/water (95:5, v/v), with 0.1 M KOH solution, was used, with phenolphthalein as an indicator.
Iodine value (IV)
IV was measured according to the AOAC Official Method 920.158. The titration of an accurate sample solution, dissolved in cyclohexane/glacial acetic acid (1:1, v/v), with 0.2 M sodium thiosulfate pentahydrate, was used, with starch as an indicator. Before the titration, 25 mL Wijs reagent was added to the mixture and incubated in the darkness for 1–2 h, and then 20 mL potassium iodide solution (100 g/L) and 150 mL water were added immediately.
POV
POV was measured by AOCS Official Method Cd 8–53. The titration of an accurate sample solution, dissolved in acetic acid glacial/2, 2, 4-trimethylpentane (60:40, v/v), with 0.01 M sodium thiosulfate pentahydrate, was used, with starch as an indicator. Before the titration, 0.5 mL saturated solution of potassium iodide was added to react with the sample solution for 1 min, and 30 mL water added immediately.
p-AV
p-AV was determined according to AOCS Official Method Cd 18-90. This measurement is based on the absorbance increase per gram of oil, measured at 350 nm (722G visible spectrophotometer, China) of the three oil solutions in isooctane, before and after reaction with p-anisidine reagent in the darkness for 8 min.
Total oxidation value (TOTOX)
TOTOX measures oil deterioration and is calculated from the formula: TOTOX = 2POV + p-AV.[Citation14]
Tocopherols
The content of tocopherols was measured using a Agilent 1100 High Performance Liquid Chromatograph, coupled with fluorescence detector (excitation at 295 nm; emission at 325 nm), Hypersil ODS2-C18 (5 μm, 4.6 μm × 150 mm) column. The mobile phase was HPLC- grade mixture of methyl alcohol: water = 98:2, at a flow rate of 0.8 mL/min and the sample amount was 3 μL at the room temperature.[Citation15] The determinations of the samples were carried out in triplicate.
Analysis of Fatty Acids Composition
The FAME was analyzed by a GC equipped with a flame ionization detector and a fused silica capillary column (100 m × 0.25 mm × 0.2 μm) coated with 100% cyanopropyl polysiloxane (CP-Sil 88, Chrompack; Middelburg, The Netherlands). The temperature program (86 min in total) was as follows: The initial temperature of the oven was 45°C for 4 min, increased to 175°C at a rate of 13°C/min, maintained for 27 min, further raised to 215°C at a rate of 4°C/min, and finally kept at this temperature for 35 min.[Citation16] Analysis of all peaks was accomplished by comparison of their retention time with FAME standards. All samples were carried out in triplicate.
Statistical Analysis
All the values were analyzed with one-way analysis of variance, and mean values were compared using the least significant difference test. The significance level was set at p < 0.05. Pearson’s Correlation Coefficient was used for the correlation analyses. All statistical analyses were carried out using IBM SPSS Statistics 19 software for Windows.
RESULTS
Changes in Chemical Parameters during Frying Cycles
The AV indicates the amounts of free fatty acid produced in oil hydrolysis. The changes in AV during frying cycles were shown in . The AV was higher in peanut oil than in palm oil and camellia oil during the frying process. However, the change of AV was the largest in palm oil, with an increase from 0.11 mg KOH/g to 0.40 mg KOH/g (the growth rate was about 263.6%). The AV of peanut oil was very high before frying, and it rose gradually with a rate of 40.5% (from 0.74 to 1.04 mg KOH/g). The AV in camellia oil increased from 0.17 to 0.55 mg KOH/g with a rate of 223.5%. These results suggested that more hydrolysis reaction occurred in palm oil and camellia oil than peanut oil under the frying conditions.
TABLE 1 Changes in vitamin E contents during frying cycles
FIGURE 1 Changes of five chemical parameters during frying cycles; (a) changes in acid values; (b) changes in iodine values; (c) changes in peroxide values; (d) changes in p-anisidine values; (e) changes in total oxidation values; Mean values with standard deviations plotted as bars.
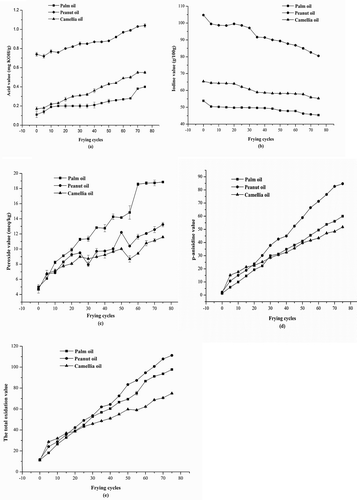
IV indicates the degree of unsaturation in oils. As shown in , IV in peanut oil was the highest among these three oils. It decreased from 104.74 to 80.52 g/100 g with the increasing frying cycles. Least changes occurred in palm oil: 53.83 to 45.36 g/100 g while the slight change of camellia oil was from 65.40 to 55.29 g/100 g. These results indicated that the decrease of unsaturated fatty acids in peanut oil was the largest among these three oils.
POV indicates the formation of peroxides due to oxidation during frying cycles. Therefore, it is widely used for the measurement of oxidative rancidity in oils and fats.[Citation17] The determination of POV can be used as oxidative index during the early stage of lipid oxidation as hydroperoxide is the primary product of lipid oxidation.[Citation18] The changes in POV during frying cycles are shown in . The overall variation trend of POV was increasing for three vegetable oils. The change of POV in palm oil was the largest, with an increase from 4.98 to 18.86 meq/kg (the growth rate was about 278.7%). The POV of peanut oil and camellia oil increased from 4.75 to 13.24 meq/kg with a rate of 178.7% and from 4.68 to 11.58 meq/kg with a rate of 147.4%, respectively.
p-AV reflected the amounts of aldehyde (principally 2-alkenals and 2,4-alka-dienals) in animal fats and vegetable oils.[Citation1] The changes in p-AV during frying cycles are shown in . There were substantial increases in p-AV for the three edible oils. The change of p-AV in camellia oil was the least (from 1.70 to 51.78) while the change in peanut oil was the largest (from 2.25 to 84.71) with the increasing frying cycles. Moderate change was observed in palm oil (from 1.36 to 60.00).
Generally speaking, the lower the TOTOX of oil, the better stability of this oil. Changes in TOTOX during frying cycles were shown in . The results showed that the change of TOTOX in camellia oil was the least (from 11.07 to 74.93), followed by palm oil (from 11.31 to 97.73), and peanut oil (from 11.76 to 111.20). Camellia oil had the lowest TOTOX as the oil contained high amounts of monounsaturated fatty acids.
Changes of Vitamin E Contents in Three Edible Oils during Frying Cycles
It is well known that vitamin E, especially α-tocopherol, is an important vegetable oil component contributing to the antioxidant properties. They are believed to protect PUFA against peroxidation.[Citation19] As shown in , palm oil was only characterized by α-tocopherol while both peanut oil and camellia oil contained γ and δ-tocopherol besides α-tocopherol. They exert their antioxidant effects by numerous biochemical and biophysical mechanisms, including scavenging active oxygen species and free radicals, and acting as efficient chain terminators in lipid autoxidation reactions.[Citation20]
Palm oil was only characterized by α-tocopherol while both peanut oil and camellia oil contained γ and δ-tocopherol besides α-tocopherol. In these three edible oils, camellia oil had the highest concentration of α-tocopherol, while peanut oil had the highest contents of γ and δ-tocopherol. Peanut oil contained higher amount of total vitamin E (509.52 mg/kg) than the other two oils. It decreased from 509.52 to 55.52 mg/kg during frying cycles. The change of α-tocopherol level was the largest in three types of tocopherols for the three edible oils. The amount of α-tocopherol decreased to 0 before the frying cycles ended in palm oil and peanut oil (palm oil: from 201.94 mg/kg to 0; peanut oil: from 327.24 mg/kg to 0) and it decreased from 439.22 to 4.36 mg/kg in camellia oil. Juárez[Citation9] noticed that there were significant losses of tocopherols, especially α-tocopherol after frying. The main reason is that α-tocopherol degrades more quickly at high temperatures than at room temperature.[Citation21] Furthermore, the effectiveness of an antioxidant mainly relies on its chemical reactivity, how it interacts with other food components and its concentration and especially its physical location in different homogeneous or heterogeneous food systems.[Citation17] Due to its largest biological activity, α-tocopherol is the most unstable among all the tocopherols.
Changes of Fatty Acids Composition during Frying Cycles
The main constituent of oils is fatty acids, and the unsaturation degree is the most important factor determining the oxidative stability among all the factors.[Citation7] As shown in , the content of SFA in palm oil increased (from 43.77 to 45.22%); the contents of MUFA, PUFA, and trans-fatty acids (TFAs) were decreased. For the peanut oil, the contents of SFA and MUFA were increased from 18.67 to 23.54% and 39.21 to 40.19%, respectively, while the contents of PUFA and TFAs were decreased from 39.23 to 34.53% and 2.77 to 1.74%, respectively. Minor changes occurred in camellia oil during frying cycles. The content of SFA increased (from 12.18 to 18.81%) and MUFA, PUFA, TFA decreased slightly.
TABLE 2 Changes in fatty acids composition during frying cycles for three edible oils (%)
On the whole, there was a clear reduction of unsaturated fatty acids with the consequent increase in SFA, compared to the relative percentage of fatty acids during the increasing frying cycles.[Citation22] The decrease was particularly noticeable in PUFA contributing to their multiple unsaturated bonds’ instability. In our study, TFAs decreased in frying oils with increased frying cycles although there was a slight increase of TFAs in the fried potatoes. The formation of TFAs is usually under severe conditions.[Citation23,Citation24] To our knowledge, frying process has not been reported to produce TFAs. However, most TFAs found in foods would come from the oil and not from the process itself.[Citation25] Furthermore, the fatty acids composition of fried potatoes was very similar to the fatty acid composition of the frying oil. This indicated that the changes of fatty acids composition include TFAs for fried potatoes were mainly dependent on the frying oils.
Correlation between Chemical Parameters and Fatty Acids Composition
shows the correlation between chemical parameters and fatty acids composition in three edible oils. There were significant correlations between fatty acids composition and the chemical parameters in three edible oils, except the ∑MUFA in peanut oil. Fatty acids composition was positively correlated with AV, POV, p-AV, and TOTOX, while it was negatively correlated with IV. ∑TFAs were highly correlated with AV, POV, IV, p-AV, and TOTOX (p < 0.05), the correlation coefficients were –0.426, –0.378, 0.360, –0.375, and –0.379, respectively. These results indicated that the changes of chemical parameters were closely related to the fatty acids composition.
TABLE 3 Pearson correlation coefficients between chemical parameters and fatty acids composition in three edible oils
Correlation between Chemical Parameters and Contents of Tocopherols
There were significant relationships between tocopherol contents and the chemical parameters in the three edible oils (). The tocopherol contents were negatively correlated with AV, POV, p-AV, and TOTOX (p < 0.01), but were positively correlated with IV (p < 0.01). The correlation coefficients between TOTOX and the total tocopherols contents were –0.987 for palm oil, –0.984 for peanut oil, and –0.950 for camellia oil (p < 0.01). These results indicated that the tocopherol content was also an important factor affecting the oxidative stability of oil.
TABLE 4 Pearson correlation coefficients between chemical parameters and contents of tocopherols in three edible oils
DISCUSSION
Fatty Acids
In this study, camellia oil (SFA: MUFA: PUFA = 1:7:1) was found to have the highest stability among three edible oils. Camellia oil has been consumed for 2000 years in China, and it contains over 70% of MUFA which is quite similar to olive oil. Wamer[Citation26] and Zenis[Citation27] also reported that potato chips fried in oil with 60–70% oleic acid, 20% linoleic acid and 3–10% linolenic acid showed higher flavor quality and stability than other oils. Lolos[Citation28] reported that the fatty acids composition of frying oils affected the oxidative deterioration during storage of potato chips. The higher C18:2 and C18:3 content of the oil resulted in a greater rate of increase in POV and TOTOX number while the olive kernel oil with higher C18:1 content contained low POV and TOTOX value. Dobarganes[Citation29] also indicated that the high-oleic sunflower oils showed an excellent behavior with regard to thermal-oxidation and frying process.
The fatty acids composition and content of oil did not change significantly during frying process. The SFA, MUFA, and PUFA of potatoes were very similar to the oil after frying process. The TFAs were detected in the three oils as well. Interestingly, the TFAs content in the three oils decreased, however, the TFAs level in potatoes increased from 0.80 to 1.30–1.74% after frying. Tsuzuki[Citation24] also reported that the TFAs were usually formed under severe condition, not by the frying process. The fatty acids composition and contents in the frying potatoes were mainly influenced by frying oil since the ratio of potato and frying oil was 1/25 (w/v).
Tocopherols
Apart from fatty acids composition, tocopherol content is another important factor to determine the oxidative stability of vegetable oils. Tocopherols play an important role in trapping the hydro-peroxide intermediates and stopping the autoxidation chain reaction and α-tocopherol is the most efficient on scavenging peroxyl radicals in homogenous tocopherols.[Citation30] In our study, the original α-tocopherol contents of palm oil, peanut oil, camellia oil were 201.94, 327.24, and 439.22 mg/kg, respectively. And the growth rates of POV were 278.7, 178.7, and 147.4%, respectively. It indicated that in the certain range, the lower concentration of α-tocopherol content, the faster the POV grew. Ruiz[Citation31] reported that the increase of POV in sunflower oil B (from 26 to 323 meq O2/kg) with 390 mg/kg α-tocopherol content was larger than sunflower oil A (from 12 to 120 meq O2/kg) with 545 mg/kg α-tocopherol content during the storage period. Fuster[Citation32] studied the antioxidant effects of α- and γ-tocopherols in a model system based on the auto-oxidation of high oleic sunflower oil and rapeseed oil. The POV of high oleic sunflower oil with 445 mg/kg α-tocopherol content increased from 0 to 300 meq O2/kg, while the POV of rapeseed oil with 224 mg/kg α-tocopherol content increased from 0 to 600 meq O2/kg after seven days of oxidation. These results agreed with ours.
Different patterns of tocopherols showed different roles: α-tocopherols affected human nutrition and health, γ-tocopherols in seed showed a strong activity at protecting compounds, such as fatty acids, while δ-tocopherols showed less active than other tocopherols.[Citation33] Generally, most oils contained three types of tocopherol, α-, γ-, and δ-tocopherol. In our study, all three oils contained α-tocopherol, peanut oil, and camellia oil also contained γ- and δ-tocopherol with much lower concentrations than α-tocopherol. During the frying process, the tocopherols showed degradation, and the decrease of tocopherols level in the three oils was in this order: α > γ > δ, after 75 frying cycles (almost 7.5 h). The results were consistent with those of Warner,[Citation21] who found that α-tocopherol was preferentially lost compared to γ- and δ-tocopherols. Sliva reported that the decrease of γ-tocopherol was faster than the decrease of α-tocopherol in corn oil and soy oil after frying at 180°C for 1 h.[Citation33] It was found that the γ-tocopherol levels in corn oil (393.9 mg/kg) and soy oil (348.8 mg/kg) were higher than α- and δ-tocopherol level of those two oils in Saliva’s study, which was the reverse case in our study. In addition, the change in tocopherols concentration showed a close relationship with fatty acids composition in oils. The α-tocopherol concentrations in palm oil with 11.83% PUFA, peanut oil with 39.23% PUFA, and camellia oil with 10.21% PUFA were decreased by 9.93, 42.46, and 5.96% at the 60th frying cycle, respectively, indicating that α-tocopherol was lost rapidly in more polyunsaturated oil during frying process. This result was consistent with a previous study.[Citation34] Casal pointed out that vitamin E and the highly monounsaturated nature of the olive oils played the main role for the reduced thermal degradation under frying conditions.[Citation7] Fatemi reported that the relative oxidation rates of methyl oleate, linoleate, and linolenate in mixtures were 1, 10.3, and 21.6.[Citation35] Therefore, the calculated oxidizability (COX) value was calculated by the percentage of unsaturated C18 fatty acids, namely COX = (1 [18:1%] + 10.3 [18:2%] + 21.6 [18:3%])/100. In this study, the COX values of fresh palm oil, peanut oil, camellia oil were 1.615, 4.631, and 1.772, respectively. And the total tocopherol contents in palm oil, peanut oil, and camellia oil were 201.94, 509.52, and 458.01 mg/kg, respectively. The oxidative stabilities of three edible oils were in the order: camellia oil > palm oil > peanut oil. Hence, it was concluded that fatty acids composition (COX value) determined the oils’ oxidative stabilities, but the tocopherol contents of the oils would play a critical role when COX values were similar.
CONCLUSIONS
The results of this work showed that the oxidative stability of camellia oil (SFA: MUFA: PUFA = 1:7:1)was the best in three edible oils, followed by palm oil and peanut oil. There was a least change of fatty acids composition in camellia oil among all the edible oils, with a lowest TOTOX. The fatty acids composition and tocopherol contents were two important factors of oils’ oxidative stabilities. The fatty acids composition (COX value) mainly determined the oils’ oxidative stabilities, and the tocopherol contents of edible oils would be an important factor to affect their oxidative stabilities when the COX values were similar.
FUNDING
We are grateful to National Natural Science Foundation of China (31071561), Research Program of State Key Laboratory of Food Science and Technology, Nanchang University (Project Nos. SKLF-ZZA-201303 and SKLF-QN-201109), PhD Subject Fund from Education Department (20113601120004), Natural Science Fund from Science and Technology Department of Jiangxi Province (20114BAB214016) for financial support.
REFERENCES
- Bou, R.; Navas, J.A.; Tres, A.; Codony, R.; Guardiola, F. Quality assessment of frying fats and fried snacks during continuous deep-fat frying at different large-scale producers. Food Control 2012, 27, 254–267.
- Nawar, W. Lipids. In: Food Chemistry; Fennema, O.R.; Eds.; Dekker M Inc.: New York, NY, 1996; 225–319.
- Saguy, I.S.; Dana, D. Integrated approach to deep fat frying: Engineering, nutrition, health, and consumers aspects. Journal of Food Engineering 2003, 56, 143–152.
- Tao, N.G.; Liu, Y.J. Chemical composition and antimicrobial activity of the essential oil from the peel of shatian pummelo (Citrus grandis Osbeck). International Journal of Food Properties 2012, 15, 709–716.
- Wang, F.; Jiang, L.Z.; Zhu, X.Q.; Hou, J.C. Effects of frying on polar material and free fatty acids in soybean oils. International Journal of Food Science and Technology 2013, 48, 1218–1223.
- Karakaya, S.; Simsek, S. Changes in total polar compounds, peroxide value, total phenols, and antioxidant activity of various oils used in deep fat frying. Journal of the American Oil Chemists’ Society 2011, 88, 1361–1366.
- Casal, S.; Malheiro, R.; Sendas, A.; Oliveira, B.P.P.; Pereira, J.A. Olive oil stability under deep-frying conditions. Food and Chemical Toxicology 2010, 48, 2972–2979.
- Abdulkarim, S.M.; Long, K.; Lai, O.M.; Muhammad, S.K.S.; Ghazali, H.M. Frying quality and stability of high-oleic Moringa oleiferaseed oil in comparison with other vegetable oils. Food Chemistry 2007, 105, 1382–1389.
- Juárez, M.D.; Osawa, C.C.; Acuña, M.E.; Sammán, N.; Gonçalves, L.A.G. Degradation in soybean oil, sunflower oil, and partially hydrogenated fats after food frying, monitored by conventional and unconventional methods. Food Control 2010, 22, 1920–1927.
- Rohman, A.; Che Man, Y.B. Authentication of extra virgin olive oil from sesame oil using FTIR spectroscopy and gas chromatography. International Journal of Food Properties 2012, 15, 1309–1318.
- Masson, L.; Robert, P.; Dobarganes, M.C.; Urra, C.; Romero, N.; Ortiz, J.; Goicoechea, E.; Pérez, P.; Salamé, M.T. Stability of potato chips fried in vegetable oils with different degree of unsaturation. Effect of ascorbyl palmitate during storage. Grasas Y Aceites 2002, 53, 190–198.
- Petukhow, I.; Malcolmson, L.J.; Przybylski, R. Storage stability of potato chips fried in genetically modified canola oils. Journal of the American oil Chemists’ Society 1999, 76, 889–896.
- Romero, A.; Bastida, S.; Sanchez-Muniz, F.J. Cyclic fatty acid monomer formation in domestic frying of frozen foods in sunflower oil and high oleic acid sunflower oil without oil replenishment. Food and Chemical Toxicology 2006, 44, 1674–1681.
- Shahidi, F.; Wanasundara, U.N. Methods for measuring oxidative rancidity in fats and oils. In: Food Lipids: Chemistry, Nutrition, and Biotechnology; Akoh, C.C.; Min, D.B.; Eds.; Marcel Dekker Inc.: New York, NY, 2002; 465–482.
- Wang, R.Y.; Tang, J.; Zhang, L.J.; Wang, Q. Determination of vitamin E contents of a variety of plant oils by high performance liquid chromatography (HPLC). Journal of Xinjiang University 2003, 20, 393–395.
- Cruz-Hernandez, C.; Deng, Z.Y.; Zhou, J.Q.; Hill, A.R.; Yurawecz, M.P.; Delmonte, P.; Mossoba, M.M.; Dugan, M.E.R.; Kramer, J.K.G. Methods for analysis of conjugated linoleic acids and trans-18:1 isomers in dairy fats by using a combination of gas chromatography, silver-ion thin-layer chromatography/gas chromatography, and silver-ion liquid chromatography. Journal of AOAC International 2004, 87, 545–562.
- Ramadan, M.F. Healthy blends of high linoleic sunflower oil with selected cold pressed oils: Functionality, stability, and anti-oxidative characteristics. Industrial Crops and Products 2013, 43, 65–72.
- Ramadan, M.F.; Moersel, J.T. Oxidative stability of black cumin (Nigella sativaL.), coriander (Coriandrum sativum L.), and niger (Guizotia abyssinica Cass.) upon stripping. European Journal of Lipid Science Technology 2004, 106, 35–43.
- Afaf, K.E.; Andersson, R. A multivariate study of the correlation between tocopherol content and fatty acid composition in vegetable oils. Journal of the American oil Chemists’ Society 1997, 74, 375–380.
- Afaf, K.E.; Lars-Åke, A. The chemistry and antioxidant properties of the tocopherols and tocotrienols. Lipids 1996, 31, 671–701.
- Warner, K.; Su, C.; White, P.J. Role of antioxidants and polymerization inhibitors in protecting frying oils. In: Frying Technology and Practices Champaign; Gupta, M.K.; Warner, K.; White, P.J.; Eds.; AOCS Press: 2004; 37–49.
- Bansal, G.; Zhou, W.B.; Tan, T.W.; Neo, F.L.; Lo, H.L. Analysis of trans-fatty acids in deep frying oils by three different approaches. Food Chemistry 2009, 116, 535–541.
- Liu, W.H.; Inbaraj, B.S.; Chen, B.H. Analysis and formation of trans-fatty acids in hydrogenated soybean oil during heating. Food Chemistry 2007, 104, 1740–1749.
- Tsuzuki, W.; Matsuoka, A.; Ushida, K. Formation of trans-fatty acids in edible oils during the frying and heating process. Food Chemistry 2010, 123, 976–982.
- Romero, A.; Cuesta, C.; Sanchez-Muniz, F.J. Trans fatty acid production in deep fat frying of frozen foods with different oils and frying modalities. Nutrition Research 2000, 20, 599–608.
- Warner, K.; Orr, P.; Parrott, L.; Glynn, M. Effects of frying oil composition on potato chip stability. Journal of the American oil Chemists’ Society 1994, 1, 1117–1121.
- Zenia, J.H.; Karen, E.; Sangsook, K.; Hardm, R.T. Quality and stability of potato chips fried in canola, partially hydrogenated canola, soybean, and cottonseed oils. Journal of Food Quality 1996, 19, 107–120.
- Lolos, M.; Oreopoulou, V.; Tzia, C. Oxidative stability of potato chips: Effect of frying oil type, temperature, and antioxidants. Journal of the Science of Food and Agriculture 1999, 79, 1524–1528.
- Dobarganes, M.C.; Máquez-Ruiz, G.; Pérez-Camino, M.C. Thermal stability and frying performance of genetically modified sunflower seed (Helianthus annuus L.) oils. Journal of Agricultural and Food Chemistry 1993, 41, 678–681.
- Kamal-Eldin, A. Effect of fatty acids and tocopherols on the oxidative stability of vegetable oils. European Journal of Lipid Science and Technology 2006, 108, 1051–1061.
- Ruiz, G.M.; Polvillo, M.M.; Jorge, N.; Ruiz Méndez, M.V.; Dobarganes, M.C. Influence of used frying oil quality and natural tocopherol content on oxidative stability of fried potatoes. Journal of the American Oil Chemists’ Society 1999, 76, 421–425.
- Fuster, M.D.; Lampi, A.M.; Hopia, A.; Kamal-Eldin, A. Effects of α-and γ-tocopherols on the autooxidation of purified sunflower triacylglycerols. Lipids 1998, 33, 715–722.
- Silva, L.; Pinto, J.; Carrola, J.; Paiva-Martins, F. Oxidative stability of olive oil after food processing and comparisons with other vegetable oils. Food Chemistry 2010, 121, 1177–1187.
- Yuki, E.; Ishikawa, Y. Tocopherol contents of nine vegetable frying oils and their changes under simulated deep fat frying conditions. Journal of the American oil Chemists’ Society 1976, 53, 673–676.
- Fatemi, S.H.; Hammond, E.G. Analysis of oleate, linoleate, and linolenate hydroperoxides in oxidized ester mixtures. Lipids 1980, 15, 379–385.
