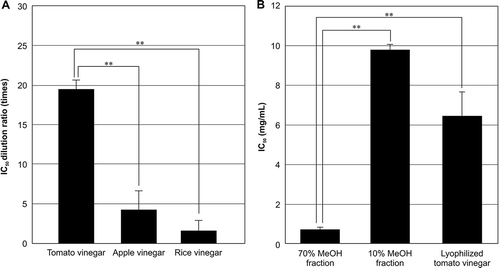
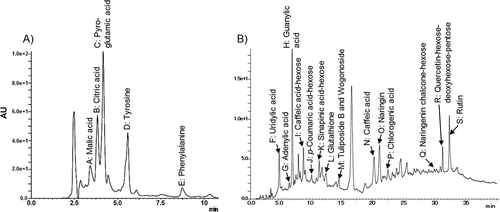
Figures & data
Table 1. Summary of investigated organic acid, amino acid, sugar, and mineral concentrations during the production process of tomato vinegar (mg/100 mL).
Table 2. Mass spectrometry ion data for compounds detected in the tomato vinegar fractions by liquid chromatography electrospray ionization mass spectrometry.