Figures & data
Figure 1. Chromatogram of the standard solution of phenolic compounds (1) Gallic acid, (2) Catechin, (3) Chlorogenic acid, (4) Caffeic acid, (5) Vanillin, (6) p-coumaric acid, (7) Ferulic acid, (8) m-coumaric acid, (9) o-coumaric acid, (10) Rosmarinic acid, (11) Quercetin (12) Trans-cinnamic acid (4 x 10-5 mol L-1) (A) and methanol extracts of leaves of carrot (B), beet (C), sweet potato (D), yam (E), radish (F). Identified peaks: (B): 1) Gallic acid, (2) Catechin, (3) Chlorogenic acid, (4) Caffeic acid, (5) Vanillin, (6) p-coumaric acid, (7) Ferulic acid, (9) o-coumaric acid, (10) Rosmarinic acid, (11) Quercetin, (12) Trans-cinnamic acid. (C): 1) Gallic acid, (2) Catechin, (3) Chlorogenic acid, (4) Caffeic acid, (5) Vanillin, (6) p-coumaric acid, (7) Ferulic acid, (10) Rosmarinic acid, (11) Quercetin (12) Trans-cinnamic acid. (D): (1) Gallic acid, (2) Catechin, (3) Chlorogenic acid, (4) Caffeic acid, (5) Vanillin, (6) p-coumaric acid, (7) Ferulic acid (9) o-coumaric acid, (10) Rosmarinic acid, (11) Quercetin (12) Trans-cinnamic acid. (E): (1) Gallic acid, (2) Catechin, (3) Chlorogenic acid, (4) Caffeic acid, (5) Vanillin, (8) m-coumaric acid, (9) o-coumaric acid, (12) Trans-cinnamic acid. (F): 1) Gallic acid, (4) Caffeic acid, (3) Chlorogenic acid, (5) Vanillin, (6) p-coumaric acid, (7) Ferulic acid, (10) Rosmarinic acid, (11) Quercetin (12) Trans-cinnamic acid.
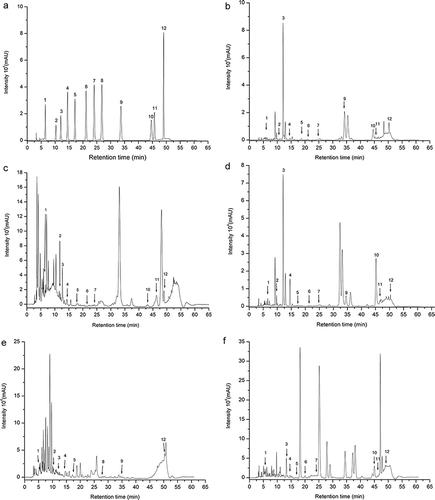
Table 1. Identification and quantification of phenolic compounds in leaf extracts of carrot, beet, sweet potato, yam, and radish.
Table 2. Iron reducing antioxidant power (FRAP), total compounds, phenolics, β-carotene, scavenging activity of free radicals (DPPH/IC50).
Table 3. Correlation coefficient (p value) between the analysis and the antioxidant capacity of the studied leaves.
