ABSTRACT
The objective of this present study was to determine the structural, physicochemical, and in-vitro digestibility properties of cross-linked rice starch with citric acid (CARS). Native rice starch (NRS) was cross-linked with different concentrations of citric acid (0, 1, 10, and 30%, w/w) of starch dry basis. The SEM results showed that cross-linking with citric acid induced changes in the morphology of starch granules. Compared with NRS, a new peak at 1734 cm−1 appeared in FT-IR spectra of CARS samples, except CARS1. In the X-ray diffraction patterns, crystalline peaks of all CARS samples became much smaller or even disappeared, as compared to that of NRS. 13C NMR spectra of CARS10 and CARS30 showed new peaks at about 45.55 ppm; in particular, an additional peak at 178.94 ppm occurred in the spectrum of CARS30. The swelling factor, light transmittance, thermal properties, and pasting properties of CARS were significantly lower than those of NRS. Furthermore, resistant starch contents of NRS were significantly increased, while the rapidly digestible starch and slowly digestible starch contents of NRS were significantly decreased after cross-linking with citric acid. Therefore, cross-linked rice starch with citric acid showed potential for application as a dietary fibre source in the food industry.
Introduction
Rice (Oryza sativa L.) is composed of a significant amount of starch, accounting for approximately 90% of the milled rice by dry weight. The cooking properties of rice depend on starch and its interactions with other components of the rice endosperm such as lipids and proteins. Rice is regarded as non-allergenic because of the hypoallergenicity of its residual protein; hence, rice starch is widely used in infant foods, processed meats, puddings, and sauces as binder, thickening agent, and inflating agent. However, rice starch has some disadvantages that limit its applications in food products. Rice starch has a high glycaemic index similar to glucose and shows instability under various temperature, shear, and pH conditions.[Citation1] To overcome these limitations, native rice starch (NRS) is often used for chemical or physical modification.
From a nutritional point of view, starch is classified into rapidly digestible starch (RDS, the starch fractions digested within 20 min after ingestion), slowly digestible starch (SDS, the starch fractions digested between 20 and 120 min after ingestion), and resistant starch (RS, the starch fractions that cannot be digested in the small intestine after ingestion) based on the rate and extent of its digestion.[Citation2] RS yields short-chain fatty acids in the large intestine through fermentation, which is beneficial to human health similar to the functional properties of dietary fibre.[Citation3]
Of the various chemical modifications of starch, cross-linking has been generally used to modify native starches using various cross-linking agents such as phosphoryl chloride (POCl3), epichlorohydrin (EPI), and polycarboxylic acids.[Citation4] In the food industry, cross-linked starch is commonly used to provide stabilized granular structure and restricted swelling as well as nutritionally beneficial effects.[Citation4] Citric acid is considered as one of the least toxic and inexpensive cross-linking agents, as compared to many other cross-linking agents. The conventional dry cross-linking method is generally used for cross-linking between starch molecules and citric acid. For example, when citric acid is heated, citric acid is dehydrated to form an anhydride, which reacts with starch molecules to form a starch citrate adduct. Further heating can result in additional dehydration of citric acid and cause cross-linking between starch molecules and citric acid.[Citation5] Xie and Liu[Citation5] reported that 78.8% RS was obtained when citric acid (40% of starch dry weight) was reacted with normal corn starch at 140°C for 7 h. Jyothi et al.[Citation6] showed that citrate cassava starches synthesized by microwave technique were less susceptible to enzyme hydrolysis than native starch.
Although the functional and structural properties of corn and cassava starches cross-linked with citric acid have been investigated previously,[Citation5,Citation7] the structural and physicochemical of cross-linked rice starch with different concentrations of citric acid have not been reported. In addition, the in-vitro digestibility properties of cross-linked rice starch with citric acid including RDS, SDS, and RS have not been discussed. Therefore, the specific objectives of the present study were to (1) produce cross-linked rice starch with different concentrations of citric acid and (2) determine structural, physicochemical, and in-vitro digestibility properties of cross-linked rice starch with different concentrations of citric acid.
Materials and methods
Materials
Rice flour was purchased from Nongshim Co., Ltd. (Seoul, Korea). Citric acid and pancreatin from porcine pancreas (P7545, 8×USP) were purchased from Junsei Chemical Co. Inc. (Tokyo, Japan), and Sigma-Aldrich Chemical Co. (St. Louis, MO, USA), respectively. Amyloglucosidase (AMG 300 L, activity 300 AGU/ml) and α-amylase were purchased from Novozyme (Bagsvaerd, Denmark) and Megazyme International Ireland Ltd. Co. (Wicklow, Ireland), respectively. Other chemicals were analytical grade reagents.
Starch preparations
The alkaline steeping method,[Citation8] with slight modification, was used for the isolation of rice starch. Rice flour (500 g) was dispersed in 1500 mL of 0.2% NaOH with constant stirring at 30 °C for 1 h. Subsequently, the suspension was centrifuged at 1800 × g for 10 min (Combi 408, Hanil Science Industrial Co., Ltd., Incheon, Korea). The supernatant was discarded and the protein extract was removed from the precipitate, and the alkaline extraction was repeated twice. The starch precipitates were then mixed with 1000 mL of distilled water and neutralized at pH 7.0 with 0.1 M HCl. The starch dispersion was centrifuged and washed twice with distilled water followed by washing 95% ethanol. The purified starch was dried at 40°C for 24 h, ground, sieved (150 μm), and stored in a sealed container at 4°C before use.
Chemical composition
Standard AACCI-approved methods were used to determine the chemical composition of isolated NRS, including the contents of moisture (44-15A), ash (08–01), protein (46-12A), and lipid (30–10).[Citation9] Total starch content was analysed using the Total Starch Assay Kit (Megazyme International Ireland Ltd. Co., Wicklow, Ireland). Amylose content of NRS was determined by the method of Williams et al.[Citation10] The amylose content was measured from a standard curve prepared with mixtures of potato amylose and maize amylopectin (Sigma-Aldrich, St. Louis, MO, USA).
Preparation of cross-linked rice starch with citric acid
Cross-linked rice starch with citric acid was produced based on the method of Klaushofer et al. [Citation11] with some modifications. Citric acid (1, 10, and 30% of starch dry basis) was dissolved in distilled water; subsequently, the pH of the solution was adjusted to 3.5 with 10 M NaOH, and finally diluted to a final volume of 50 mL with distilled water. The citric acid solution (50 mL) was mixed with 50 g of NRS and the dispersion was conditioned at ambient temperature for 16 h. The mixture was dried in an air oven at 60°C for 6 h to a moisture level of 5–10% (w/w), ground, and dried in a dry oven for 4 h at 140°C. The dry mixture was rinsed three times with distilled water to remove unreacted citric acid. The washed starch was air-dried at 40°C for 24 h, ground into powder, and passed through a 150 μm sieve. The NRS cross-linked with the three concentrations of citric acid (1, 10, and 30%) were designated to as: CARS1, CARS10, and CARS30, respectively. The degree of substitution (DS) was determined based on the method of Kweon et al.[Citation12]
Scanning electron microscopy (SEM)
The morphology of NRS and CARS was determined using Scanning Electron Microscope (S-4700, Hitachi Co., Tokyo, Japan) according to the method of Atrous et al.[Citation13] with some modifications. Starch samples were placed on double-sided carbon tape attached to a specimen holder and then coated with platinum powder. The samples were examined at an accelerating voltage of 10 kV and magnification of ×10,000.
Fourier-transform infrared (FT-IR) spectroscopy
Fourier-Transform Infrared (FT-IR) spectroscopy of NRS and CARS was performed by the method of Chung et al. [Citation14] (2011) with some modifications. FT-IR spectra were measured using a Fourier-transform infrared spectrophotometer (Spectrum One System, Perkin-Elmer, Waltham, MA, USA). NRS and CARS were ground with potassium bromide (KBr) and pressed into a disc. The spectral scanning range was 4000 to 500 cm−1.
X-ray (XRD) diffraction
To determine the X-ray diffraction of CARS and NRS, the method of Won et al. [Citation15] was used with some modifications. X-ray diffraction patterns were obtained by an X-ray diffractometer (X′Pert PRO, PANalytical, Almelo, Netherlands). X-ray generator tension and current were 40 kV and 40 mA, respectively. The samples were scanned through the range of 2θ from 6 ° to 30°.
13C nuclear magnetic resonance (NMR) spectroscopy
The 13C NMR spectra were recorded on a JMM-ECZ 500R spectrometer (JEOL Co., Tokyo, Japan). The samples were prepared for NMR experiments based on the method of Xu and Seib [Citation16] with some modification. Each sample (2 g) with α-amylase (10 ul) was dispersed in 30 mL of distilled water; afterwards, sodium acetate (0.3 g) was added. The mixture was heated in a water bath at 85°C for 2 h under constant agitation to hydrolyse the starch, and then moved to a boiling water bath for 30 min to inactivate the enzyme. After cooling to room temperature, the mixture was centrifuged at 3000 rpm for 10 min and the supernatant obtained was freeze-dried (FD8508, Ilshin BioBase Co., Ltd., Yangju, Korea). The freeze-dried starch hydrolysate (0.2 g) was dissolved in 3 mL of D2O for 2 h at room temperature followed by freeze-drying, and the process was repeated once. The D2O-exchanged starch (0.05 g) was solubilized in 0.5 mL of D2O. 13C NMR spectra were recorded at 400.13 MHz. The 13C spectra were measured at 25°C for 128 scans with an acquisition time of 0.82 s and a delay time of 1 s.
Swelling factor and light transmittance
Swelling factor of NRS and CARS was determined by the method of Tester and Morrison.[Citation17] Light transmittance of NRS and CARS was measured based on the procedure of Reddy and Seib.[Citation18]
Thermal properties
Thermal properties of NRS and CARS were investigated by the method of Chung and Liu [Citation19] with some modifications. The thermal properties of NRS and CARS were analysed using a differential scanning calorimeter (DSC; A200, TA Instruments, New Castle, DE, USA). Starch (20 mg) and distilled water (1 mL) were added to an aluminium DSC pan. The pan was immediately sealed and equilibrated at room temperature for 1 h before heating in the DSC. The starch sample was heated from 30 to 130°C at a heating rate of 10°C/min. An empty aluminium pan was used as reference. The onset, peak, and final temperature (To, Tp, and Tc, respectively) and gelatinization enthalpy (ΔH) were calculated using Universal Analysis software provided by the manufacturer.
Pasting properties
The pasting properties of NRS and CARS were conducted using a Rapid Visco Analyser (RVA model 4, Newport Scientific Inc., Warriewood, Australia) with Standard Method 1 profile according to AACC Approved Method 76–21 (American Association of Cereal Chemists International, 2000). [Citation9] The starch suspensions (14%, wet basis) were held at 50°C, heated at 12°C/min from 50 to 95°C, held at 95°C for 2.5 min, cooled from 95 to 50°C at the same rate, and held at 50°C for 2 min. The RVA instrument provided the following parameters: pasting temperature, peak viscosity, breakdown, setback and final viscosity. The parameters from the RVA were processed by Thermocline version 1.2 software (Newport Scientific Inc.).
In-vitro digestibility
The in-vitro digestibility of NRS and CARS was determined based on the method described by Englyst et al.[Citation2] with some modifications. Pancreatin (1 g) was dispersed in 12 mL of distilled water for 10 min. The dispersion was then centrifuged at 1500 × g for 10 min. The supernatant (10 mL) was mixed with 0.2 mL of amyloglucosidase and 1.8 mL of distilled water. This enzyme solution was prepared freshly and equilibrated at 37°C for 10 min before each test.
The NRS and CARS (30 mg, db) with 2 glass balls (5 mm diameter) were added to each micro-tube (2 mL), followed by adding to 0.75 mL of sodium acetate buffer (0.1 M, pH 5.2). The micro-tubes were equilibrated in a water bath (37°C) for 10 min. The enzyme solution (0.75 mL) was then added to the sample tube and incubated in a water bath at 37°C with shaking (240 rpm). After hydrolysis for 20 or 120 min, the tubes were boiled for 10 min to deactivate the enzyme and terminate the reaction and centrifuged at 5000 × g for 10 min. The glucose content in the supernatant was measured by the dinitrosalicylic acid (DNS) method. The percentage of hydrolysed starch was calculated by multiplying a factor of 0.9 to change values for glucose to starch. The rapidly digestible starch (RDS), slowly digestible starch (SDS), and resistant starch (RS) contents in NRS and CARS were calculated using the equations (1, 2), and (3).
where G20 and G120 are glucose content released after 20 and 120 min, respectively; and W is the starch weight (mg) used for each test.
Statistical analysis
All statistical analyses were performed using SAS version 9.3 (SAS Institute Inc., Cary, NC, USA). Analysis of variance (ANOVA) was performed using the general linear models (GLM) procedure to determine significant differences among the samples. Means were compared by using Fisher’s least significant difference (LSD) procedure. Significance was defined at the 5% level.
Results and discussion
Chemical composition
The moisture, ash, protein, lipid, and total starch contents of NRS were 3.60%, 0.17%, 0.33%, 0.90%, and 93.32% (data are not shown), respectively, suggesting that protein, lipid, and ash in rice flour were removed almost entirely from the isolated starch. The amylose and amylopectin contents of NRS were 22.11 and 77.78%, respectively (data not shown). These findings were consistent with previous literatures. [Citation20,Citation21] You et al. [Citation21] reported that the amylose contents of various rice species ranged 22.4 to 25.5%.
Determination of DS
The DS of CARS varied from 0.01 to 0.27 and showed increase with increasing concentrations of citric acid to starch (). Mei et al.[Citation7] reported that the DS values of cassava starch cross-linked with citric acid ranged from 0.058 to 0.178 when the concentrations of citric acid were increased from 10 to 30%, respectively. In the case of hydroxylpropylated and oxidized normal potato starch films cross-linked with citric acid, the degree of di-esterification was increased from 0.008 to 0.054 with the increase of citric acid contents from 5 to 30%, respectively.[Citation22]
Table 1. Degree of substitution (DS), swelling factor, light transmittance and thermal properties of native and cross-linked rice starches prepared with different concentrations of citric acid (1–30%).a
Scanning electron microscopy (SEM)
The scanning electron microphotographs of NRS and CARS samples are presented in . NRS had polygonal, and irregular shape with a smooth surface, which was consistent with the previous publication.[Citation23] Besides, NRS showed some pores on the surface of the granules (depicted by arrows). However, the surface of CARS appeared rougher than that of NRS. As the concentrations of citric acid increased, this phenomenon was more pronounced, subsequently, causing the erosion on the core part of the starch granules. The morphological changes possibly indicate that the citric acid penetrated the pores on the surface of starch granules and approached the hilum through the channels that connect the pores to the hilum.[Citation3] This could result in the erosion on the central part of starch granules. This result indicated that the cross-linking reaction with citric acid could affect the granule morphology.
Fourier-transform infrared (FT-IR) spectroscopy
The FT-IR spectra of NRS, CARS, and pure citric acid are presented in . Both NRS and CARS showed a broad peak in the range of 3000–3600 cm−1 and 2932 cm−1, which correspond to OH and CH stretching, respectively.[Citation24,Citation25] The peaks around 1650 cm−1 and 1420 cm−1 were attributed to OH and CH bending, respectively.[Citation25] Three peaks observed near 1018–1155 cm−1 are the distinctive peaks of the pyranose form of glucose residues.[Citation26]
Figure 2. FT-IR spectra of citric acid, native, and cross-linked rice starches prepared with different concentrations of citric acid (1–30%).
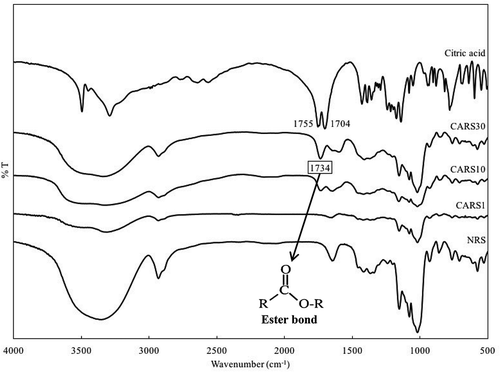
In particular, a new peak at 1734 cm−1 appeared in the FT-IR spectra of CARS samples except CARS1. The absorbance at 1734 cm−1 may be associated with the new characteristic ester group between the hydroxyl groups of cassava starch molecules and carboxyl groups of citric acids during cross-linking.[Citation27] Thus, a hydroxyl group of the starch can be reacted with a carboxyl group of the citric acid during cross-linking, consequently leading to the formation of the ester bond in the CARS structure.
In the FT-IR spectrum of pure citric acid (), the peak observed at 1704 cm−1 was due to the C = O stretching vibration in the carboxyl groups; and the peak found at 1755 cm−1 was the interference peak that occur when the carboxyl groups are very close to each other in diacid.[Citation28] However, these two peaks disappeared in the FT-IR spectra of CARS samples. According to Zhou et al.,[Citation29] the change of the peak’s position from 1709 cm−1 in pure citric acid to 1743 cm−1 in the modified starch nanocrystals confirmed that the crosslinking between starch and citric acid occurred during heating. These authors also noted that the unbound citric acid was thoroughly washed from the modified starch nanocrystal, leading to disappearance of the 1709 cm−1 peak. Thus, these results revealed successful cross-linking of NRS with citric acids during the cross-linking process.
X-ray (XRD) diffraction
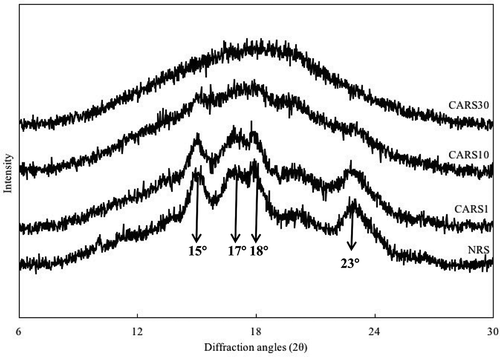
The X-ray diffraction patterns of NRS and CARS samples are shown in . NRS exhibited the typical A-type crystalline pattern with 2θ reflection intensities at 15, 17, 18 and 23°.[Citation1,Citation30] After cross-linking with citric acid, the crystalline patterns of CARS1 rarely change in comparison to NRS. However, higher DS of CARS samples is associated with lower intensity of these peaks; moreover, the crystalline peaks of CARS30 were not detected. This suggested that cross-linking of citric acid should occur both in the amorphous regions and crystalline regions. According to Xie et al.,[Citation3] when citric acid migrates into the channels and cavities of the starch granules, it could destroy the crystalline structures due to the concentrated solution of citric acid. Therefore, esterification of the citric acid group on the amorphous regions and crystalline regions of starch could form a highly cross-linked starch and restrict the mobility of the starch chain.
13C NMR spectroscopy
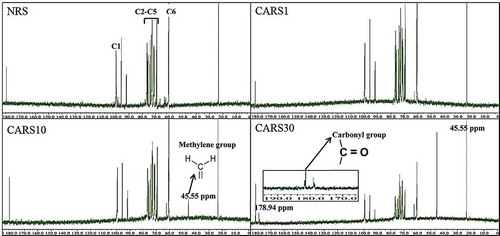
As shown by 13C NMR spectra of NRS and CARS (), the signals at around 100 ppm corresponded to the internal anomeric C-1.[Citation31] The multiplets between 68 and 78 ppm were assigned to C-2, C-3, C-4, and C-5.[Citation32] The signal at 60.44 ppm was from C-6.[Citation32] There were no significant differences between NRS and CARS1, probably because DS was too low to change the structure of NRS. However, 13C NMR spectra of CARS10 and CARS30 had an additional peak near 45.55 ppm, which might be attributed to the methylene group of citric acid.[Citation33] In case of the spectrum of CARS30, a new peak appeared at 178.94 ppm, as compared to other samples prepared in this study. This peak might be assigned to the carbonyl group of CARS for an ester bond, which confirmed the reaction between carboxylic groups from citric acid and hydroxyl groups from starch molecules.[Citation31] This result was confirmed by FT-IR spectra (). Therefore, in the present study, the results of the 13C NMR spectra confirmed the change of structure of NRS after cross-linking.
Swelling factor and light transmittance
The swelling factors of the CARS samples were significantly lower than that of the NRS samples (). In addition, the swelling factor of CARS samples significantly decreased with increasing concentrations of citric acid, in agreement with the results of Mei et al.,[Citation7] who reported that the swelling power of cross-linked cassava starch significantly decreased with the increase of citric acid. The findings indicated that cross-linking between starch molecules and citric acid could strengthen the starch network both chemically and physically, thus resulting in lower swelling factor.[Citation22] Dastidar et al.[Citation27] noted that the cross-linking of corn starch with malonic acid hindered the penetration of water into the starch network because of the compact structure of the cross-linked starch. Additionally, cross-linked tapioca starch with citric acid decreased the absorption of water.[Citation34] In the present study, we confirmed that the cross-linked structure between starch granules and citric acid inhibited the absorption of water, subsequently resulting in the reduction of swelling factor. The light transmittance of NRS and CARS pastes is shown in . The light transmittance values of CARS pastes were significantly lower than that of NRS. The light transmittance values of CARS were significantly decreased with increasing the concentrations of citric acid, which could be associated with new cross-linking between the starch granules. Jyothi et al.[Citation4] reported that cross-linked starch had denser granules than native starch and reflected rather than transmitted light.
Thermal properties
The gelatinization parameters of NRS and CARS are shown in . The onset temperature (To), peak temperature (Tp), conclusion temperature (Tc), and enthalpy of gelatinization (ΔH) of NRS were 59.56°C, 64.89°C, 71.53°C, and 1.59 J/g, respectively. The To, Tp, Tc, and ΔH values of CARS samples were significantly decreased for CARS 1 and not determined for CARS 10 and CARS 30, as compared with those of NRS. The new crosslinking between starch granules in CARS can prevent the water absorption and consequently decrease the swelling and gelatinization of starch granules. Kartha and Srivastas[Citation35] reported that highly cross-linked starches had lower gelatinization enthalpy than native starch due to less portion of starch granules that could be gelatinized by heat. Accordingly, in the present study, cross-linking with citric acid could restrict the hydration, swelling and gelatinization of CARS molecules, subsequently resulting in the reduction of gelatinization parameters for CARS samples.
Pasting properties
The pasting properties of NRS and CARS are presented in . The pasting properties of CARS30 were not detected due to too high degree of cross-linking. CARS1 and CARS10 showed a significant decrease in the pasting temperature and peak viscosity, as compared to NRS. The reinforcement of bonding between starch chains by cross-linking with citric acid can restrict water absorption and swelling of the CARS granules, leading to a decrease in the peak viscosity of CARS.[Citation36]
Table 2. Rapidly digestible starch (RDS), slowly digestible starch (SDS), resistant starch (RS), and pasting properties of native and cross-linked rice starches prepared with different concentrations of citric acid (1–30%).a
CARS1 and CARS10 exhibited lower breakdown viscosity (2.38–113.73 RVU) than NRS (227.75 RVU). Yussof et al.[Citation37] reported that cross-linked starches showed lower breakdown than native starch because of their compact granule structure. In the present study, we confirmed that cross-linking with citric acid could form the compact granule structures of CARS during crosslinking, consequently improving the resistance against the high heat and shear.
In-vitro digestibility
The starch fractions including rapidly digestible starch (RDS), slowly digestible starch (SDS), and resistant starch (RS) of NRS and all CARS are presented in . The RDS and SDS contents of NRS were significantly higher than those of CARS. The RDS contents of CARS were significantly decreased from 47.52 to 16.87%, when the citric acid concentrations were increased from 1 to 30%, respectively. However, the RS contents of all CARS were significantly higher than that of NRS. In particular, the RS content of CARS30 was about 5.6 times higher than that of NRS. These findings could be due to the stearic hindrance by the newly cross-linked structure in CARS. The presence of citric acid in CARS could sterically inhibit the proper positioning of the substrate into the active site of digestive enzymes such as α-amylase and amyloglucosidase, and consequently, restrict enzyme hydrolysis.[Citation38] These results corroborate the results of previous studies.[Citation6,Citation25]
Another possible explanation for the decrease in the digestibility of starch could be the decrease in the swelling factor of CARS (). Chung et al.[Citation39] reported that the swelling of starch granules could facilitate the access of digestive enzymes to the granule interior, leading to an increase in the RDS contents. In the present study, the swelling factor of CARS was decreased with increasing the concentrations of citric acid, which caused difficulty in access of the digestive enzymes to the starch granules and subsequently, increased the RS contents of CARS. Therefore, the cross-linking with citric acid could cause stearic hindrance and restrict the swelling in the CARS granules, thereby increasing the RS contents of CARS.
Conclusion
The structural, physicochemical, and in-vitro digestibility properties of CARS with different concentrations of citric acid were evaluated in the present study. Based on FT-IR, X-ray, SEM, and 13C NMR results, esterification of the citric acid group on the amorphous regions and crystalline regions of starch could cause formation of the compact structure of cross-linked starch. When the DS of CARS were increased, the swelling factor, light transmittance, thermal properties, and pasting properties of CARS were significantly decreased. These results confirmed that the structural changes induced by cross-linking with citric acid affected the physicochemical properties of NRS. Furthermore, the compact granule structure of CARS could sterically hinder the access of digestive enzymes to the surface of starch, leading to a decrease in digestibility of NRS. Cross-linking with citric acid improved the RS contents of rice starch. Thus, CARS can be used as an ingredient of functional food in the food industry.
Additional information
Funding
References
- Van Hung, P.; Chau, H. T.; Phi, N. T. L. In Vitro Digestibility and in Vivo Glucose Response of Native and Physically Modified Rice Starches Varying Amylose Contents. Food Chemistry 2016, 191, 74–80.
- Englyst, H. N.; Kingman, S. M.; Cummings, J. H. Classification and Measurement of Nutritionally Important Starch Fractions. European Journal of Clinical Nutrition 1992, 46, S33–50.
- Xie, X. S.; Liu, Q.; Cui, S. W. Studies on the Granular Structure of Resistant Starches (Type 4) from Normal, High Amylose and Waxy Corn Starch Citrates. Food Research International 2006, 39, 332–341.
- Jyothi, A. N.; Moorthy, S. N.; Rajasekharan, K. N. Effect of Cross‐Linking with Epichlorohydrin on the Properties of Cassava (Manihot esculenta Crantz) Starch. Starch‐Stärke 2006, 58, 292–299.
- Xie, X. S.; Liu, Q. Development and Physicochemical Characterization of New Resistant Citrate Starch from Different Corn Starches. Starch‐Stärke 2004, 56, 364–370.
- Jyothi, A. N.; Moorthy, S. N.; Sreekumar, J. N.; Rajasekharan, K. N. Studies on the Properties of Citrate Derivatives of Cassava (Manihot esculenta Crantz) Starch Synthesized by Microwave Technique. Journal of the Science of Food and Agriculture 2007, 87, 871–879.
- Mei, J.; Zhou, D.; Jin, Z.; Xu, X.; Chen, H. Effects of Citric Acid Esterification on Digestibility, Structural and Physicochemical Properties of Cassava Starch. Food Chemistry 2015, 187, 378–384.
- Yamamoto, K.; Sawada, S.; Onogaki, T. Properties of Rice Starch Prepared by Alkali Method with Various Conditions. Journal of the Japanese Society of Starch Science 1973, 20, 99–104.
- American Association of Cereal Chemists (AACC). Approved Method of the AACC, Methods 08-01, 44-15A, 46-12A, 30-10; AACC International: St. Paul, MN, USA, 2000; 10th ed.
- Williams, P.; Kuzina, F.; Hlynka, I. Rapid Colorimetric Procedure for Estimating the Amylose Content of Starches and Flours. Cereal Chemistry 1970, 47, 411–421.
- Klaushofer, H.; Berghofer, E.; Steyrer, W. Die Neuentwicklung Modifizierter Stärken Am Beispiel Von Citratstärke. Ernahrung/Nutrition 1978, 2, 51–55.
- Kweon, D.; Choi, J.; Kim, E.; Lim, S. Adsorption of Divalent Metal Ions by Succinylated and Oxidized Corn Starches. Carbohydrate Polymers 2001, 46, 171–177.
- Atrous, H.; Benbettaieb, N.; Chouaibi, M.; Attia, H.; Ghorbel, D. Changes in Wheat and Potato Starches Induced by Gamma Irradiation: A Comparative Macro and Microscopic Study. International Journal of Food Properties 2017, 20, 1532–1546.
- Chung, H. J.; Liu, Q.; Lee, L.; Wei, D. Relationship between the Structure, Physicochemical Properties and in Vitro Digestibility of Rice Starches with Different Amylose Contents. Food Hydrocolloids 2011, 25, 968–975.
- Won, C.; Jin, Y. I.; Kim, M.; Lee, Y.; Chang, Y. H. Structural and Rheological Properties of Potato Starch Affected by Degree of Substitution by Octenyl Succinic Anhydride. International Journal of Food Properties 2017 doi:10.1080/10942912.2016.1272610.
- Xu, A.; Seib, P. Determination of the Level and Position of Substitution in Hydroxypropylated Starch by High-Resolution 1H NMR Spectroscopy of Alpha-Limit Dextrins. Journal of Cereal Science 1997, 25, 17–26.
- Tester, R. F.; Morrison, W. R. Swelling and Gelatinization of Cereal Starches. I. Effects of Amylopectin, Amylose, and Lipids. Cereal Chemistry 1990, 67, 551–557.
- Reddy, I.; Seib, P. A. Paste Properties of Modified Starches from Partial Waxy Wheats. Cereal Chemistry 1999, 76, 341–349.
- Chung, H. J.; Liu, Q. Effect of Gamma Irradiation on Molecular Structure and Physicochemical Properties of Corn Starch. Journal of Food Science 2009, 74, C353–C361.
- Bao, J.; Shen, S.; Sun, M.; Corke, H. Analysis of Genotypic Diversity in the Starch Physicochemical Properties of Nonwaxy Rice: Apparent Amylose Content, Pasting Viscosity and Gel Texture. Starch‐Stärke 2006, 58, 259–267.
- You, S. Y.; Oh, S. K.; Kim, H. S.; Chung, H. J. Influence of Molecular Structure on Physicochemical Properties and Digestibility of Normal Rice Starches. International Journal of Biological Macromolecules 2015, 77, 375–382.
- Menzel, C.; Olsson, E.; Plivelic, T. S.; Andersson, R.; Johansson, C.; Kuktaite, R.; Järnström, L.; Koch, K. Molecular Structure of Citric Acid Cross-Linked Starch Films. Carbohydrate Polymers 2013, 96, 270–276.
- Gani, A.; Ashwar, B. A.; Akhter, G.; Shah, A.; Wani, I. A.; Masoodi, F. A. Physico-Chemical, Structural, Pasting and Thermal Properties of Starches of Fourteen Himalayan Rice Cultivars. International Journal of Biological Macromolecules 2017, 95, 1101–1107.
- Kim, M. J.; Choi, S. J.; Shin, S. I.; Sohn, M. R.; Lee, C. J.; Kim, Y.; Cho, W. I.; Moon, T. W. Resistant Glutarate Starch from Adlay: Preparation and Properties. Carbohydrate Polymers 2008, 74, 787–796.
- El Halal, S. L. M.; Colussi, R.; Pinto, V. Z.; Bartz, J.; Radunz, M.; Carreño, N. L. V.; Dias, A. R. G.; Da Rosa Zavareze, E. Structure, Morphology and Functionality of Acetylated and Oxidised Barley Starches. Food Chemistry 2015, 168, 247–256.
- Dastidar, T. G.; Netravali, A. N. Green’ Crosslinking of Native Starches with Malonic Acid and Their Properties. Carbohydrate Polymers 2012, 90, 1620–1628.
- Biswas, A.; Shogren, R.; Selling, G.; Salch, J.; Willett, J.; Buchanan, C. M. Rapid and Environmentally Friendly Preparation of Starch Esters. Carbohydrate Polymers 2008, 74, 137–141.
- Shi, R.; Zhang, Z.; Liu, Q.; Han, Y.; Zhang, L.; Chen, D.; Tian, W. Characterization of Citric Acid/Glycerol Co-Plasticized Thermoplastic Starch Prepared by Melt Blending. Carbohydrate Polymers 2007, 69, 748–755.
- Zhou, J.; Tong, J.; Su, X.; Ren, L. Hydrophobic Starch Nanocrystals Preparations through Crosslinking Modification Using Citric Acid. International Journal of Biological Macromolecules 2016, 91, 1186–1193.
- Karwasra, B. L.; Gill, B. S.; Kaur, M. Rheological and Structural Properties of Starches from Different Indian Wheat Cultivars and Their Relationships. International Journal of Food Properties 2017 doi:10.1080/10942912.2017.1328439.
- Komulainen, S.; Verlackt, C.; Pursiainen, J.; Lajunen, M. Oxidation and Degradation of Native Wheat Starch by Acidic Bromate in Water at Room Temperature. Carbohydrate Polymers 2013, 93, 73–80.
- Chi, H.; Xu, K.; Wu, X.; Chen, Q.; Xue, D.; Song, C.; Zhang, W.; Wang, P. Effect of Acetylation on the Properties of Corn Starch. Food Chemistry 2008, 106, 923–928.
- Dong, C.; Li, C.; Xiao, H.; He, B.; Qian, L. β-Cyclodextrin Grafted Cellulose and Cationic Starch for Antibacterial Paper Products: A Comparative Study. BioResources 2014, 9, 3580–3590.
- Mohamad, K. A. W.; Ismail, H.; Othman, N. Characterization of Citric Acid-Modified Tapioca Starch and Its Influence on Thermal Behavior and Water Absorption of High Density Polyethylene/Natural Rubber/Thermoplastic Tapioca Starch Blends. Polymer-Plastics Technology and Engineering 2011, 50, 748–753.
- Kartha, K.; Srivastava, H. Reaction of Epichlorhydrin with Carbohydrate Polymers. Part I. Starch Reaction Kinetics. Starch‐Stärke 1985, 37, 270–276.
- Włodarczyk-Stasiak, M.; Mazurek, A.; Kowalski, R.; Pankiewicz, U.; Jamroz, J. Physicochemical Properties of Waxy Corn Starch after Three-Stage Modification. Food Hydrocolloids 2017, 62, 182–190.
- Yussof, N. S.; Utra, U.; Alias, A. K. Hydrolysis of Native and Cross‐Linked Corn, Tapioca, and Sweet Potato Starches at Sub‐Gelatinization Temperature Using a Mixture of Amylolytic Enzymes. Starch‐Stärke 2013, 65, 285–295.
- Hoover, R.; Zhou, Y. In Vitro and in Vivo Hydrolysis of Legume Starches by α-amylase and Resistant Starch Formation in Legumes—A Review. Carbohydrate Polymers 2003, 54, 401–417.
- Chung, H.; Shin, D.; Lim, S. In Vitro Starch Digestibility and Estimated Glycemic Index of Chemically Modified Corn Starches. Food Research International 2008, 41, 579–585.