Abstract
Aim: A series of isocoumarin–chalcone hybrids were prepared and assays for the inhibition of four isoforms of human carbonic anhydrase (hCA; EC 4.2.1.1), hCA I, II, IX and XII. Materials & methods: Isocoumarin–chalcone hybrids were synthesized by condensing acetyl–isocoumarin with aromatic aldehydes. They did not significantly inhibit off-target cytosolic isoforms hCA I and II (KI >100 μM) but acted as low micromolar or submicromolar inhibitors for the tumor-associated isoforms hCA IX and XII. Results & conclusion: Our work provides insights into a new and scarcely investigated chemotype which provides interesting tumor-associated CA inhibitors, considering that some such derivatives like sulfonamide SLC-0111 are in advanced clinical trials for the management of metastatic advanced solid tumors.
Plain language summary
A series of isocoumarin–chalcone hybrids was prepared and assays for the inhibition of four isoforms of the metalloenzyme carbonic anhydrase (CA; EC 4.2.1.1), i.e., human (h) isoforms hCA I, II, IX and XII. Isocoumarins were less investigated as inhibitors of this enzyme. Here we show that the isocoumarin–chalcone hybrids do not significantly inhibit the off-target cytosolic isoforms hCA I and II (KIs >100 μM) but act as low micromolar inhibitors for the tumor-associated isoforms hCA IX and XII. Our work thus provides insights into a new and scarcely investigated chemotype which may provide interesting tumor-associated CA inhibitors, because some such compounds, e.g., the sulfonamide SLC-0111, are presently in advanced clinical trials for the management of metastatic advanced solid tumors.
GRAPHICAL ABSTRACT
Carbonic anhydrases are enzymes involved in many physiologic and pathologic processes connected with pH regulation, CO2/bicarbonate homeostasis and metabolism.
Carbonic anhydrase inhibitors have clinical applications as diuretics, antiglaucoma, antiepileptic, antiobesity and anticancer agents.
Isocoumarins were scarcely investigated as carbonic anhydrase inhibitors.
As the isostructural coumarins, isocoumarins may act as isoform-selective carbonic anhydrase inhibitors.
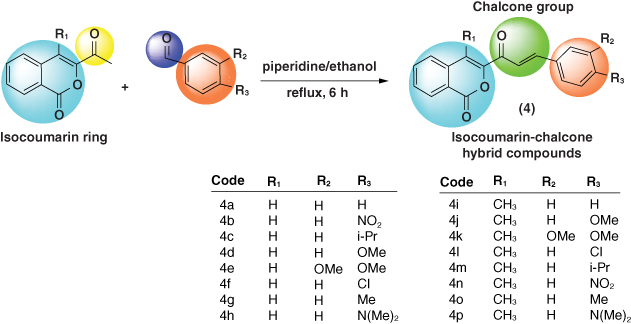
We report a series of isocoumarins incorporating also chalcone moieties in their molecules.
The synthesis was done from 3-acetyl-isocoumarin by condensation reactions with substituted benzaldehydes, leading to the formation of the chalcone fragment.
The isocoumarin–chalcone hybrids were assayed for the inhibition of four physiologically relevant human carbonic anhydrase isoforms, hCA I, II, IX and XII, the last two being tumor-associated enzymes and validated anticancer targets.
The hybrids did not inhibit the offtarget cytosolic isocforms hCA I and II.
Many of the new hybrids effectively inhibited the tumor associated isoforms hCA IX and XII, in the low micromolar or submicromolar range.
This underexplored new chemotype with carbonic anhydrase inhibitory activity, the isocoumarins, may provide isoform-selective inhibitors for the investigation and management of diseases in which such enzymes are overexpressed.
Acknowledgments
The authors would like to thank Harran University Scientific Research Projects Coordination Department for financial support (HÜBAP, Project number: 21089).
Financial disclosure
The authors would like to thank Harran University Scientific Research Projects Coordination Department for financial support (HÜBAP, Project number: 21089). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Competing interests disclosure
The authors have no competing interests or relevant affiliations with any organization or entity with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Writing disclosure
No writing assistance was utilized in the production of this manuscript.
