ABSTRACT
Chlorogenic acid is a well-known nutraceutical, but it is extensively metabolized by the body. More valuable information can be obtained from its metabolites. Dihydrocaffeic acid is a metabolite of chlorogenic acid and has shown antioxidant, cardioprotective, and neuroprotective effects; however, information about its anticancer activity is very scarce. Therefore, the main objective of this study was to determine the anticancer potential of dihydrocaffeic acid. The cancer cell lines used were MCF-7, Hep-G2, PC-3, and HCT-116, while HDFa was used as healthy cells. The cytotoxic concentrations to kill 50%, 75%, and 90% of the cells (CC50, CC75, CC90) were determined using the MTS assay. Dihydrocaffeic acid was significantly more cytotoxic for most cancer cell lines, including MCF-7, PC-3, and HCT-116, compared with HDFa; however, Hep-G2 was significantly more resistant than HDFa. Dihydrocaffeic acid is a potential candidate for cancer prevention and treatment. The mechanism of action remains to be elucidated.
RESUMEN
El ácido clorogénico es un nutracéutico bien conocido, pero es metabolizado extensamente por el cuerpo. Se puede obtener información más valiosa de sus metabolitos. El ácido dihidrocafeico es un metabolito del ácido clorogénico y ha mostrado efectos antioxidantes, cardioprotectores y neuroprotectores; sin embargo, la información sobre su actividad anticancerígena es muy escasa. Por lo tanto, el objetivo principal de este estudio fue determinar el potencial anticancerígeno del ácido dihidrocafeico. Las líneas celulares cancerígenas empleadas fueron la MCF-7, Hep-G2, PC-3 y HCT-116, mientras que la HDFa se utilizó como células sanas. Se determinaron las concentraciones citotóxicas para matar el 50%, 75% y 90% de las células (CC50, CC75, CC90) utilizando el ensayo MTS. El ácido dihidrocafeico fue significativamente más citotóxico para la mayoría de las líneas celulares cancerígenas, incluyendo MCF-7, PC-3 y HCT-116, comparado con la HDFa; no obstante, la Hep-G2 fue significativamente más resistente que la HDFa. El ácido dihidrocafeico es un candidato potencial para la prevención y tratamiento del cáncer. El mecanismo de acción queda pendiente de elucidarse.
1. Introduction
Chlorogenic acid is a well-known nutraceutical widely spread in plant foods. This phenolic compound, from the family of hydroxycinnamic acids, possesses many beneficial properties for health, including antioxidant, antidiabetic, antihypertensive, and antiobesity activities (Santana-Gálvez et al., Citation2017). Few studies, however, have been performed to assess the anticancer activity of chlorogenic acid. Some in vitro studies have reported antiproliferative and cytotoxic activity of chlorogenic acid against human breast (Deka et al., Citation2017), lung (Yamagata et al., Citation2018), colon (Hou et al., Citation2017), bone (Zhang et al., Citation2019), and kidney cancer cells (X. Wang et al., Citation2019b), while an in vivo study found antitumoral activity against colon cancer in rats (Matsunaga et al., Citation2002). Nonetheless, chlorogenic acid is extensively metabolized by the human body (Olthof et al., Citation2003), which may diminish or nullify its in vitro anticancer activity, while the benefits found in vivo may be caused not by the compound itself but by its metabolites. Therefore, more valuable information about the anticancer potential of chlorogenic acid can be obtained from its metabolites.
Dihydrocaffeic acid (3,4-dihydroxyhydrocinnamic acid, 3-(3,4-dihydroxyphenyl)propionic acid, hydrocaffeic acid) is one of the main metabolites of chlorogenic acid as evidenced by both in vitro gastrointestinal digestion (Ekbatan et al., Citation2016; Ludwig et al., Citation2013) and human studies (Olthof et al., Citation2003). Briefly, these studies showed that chlorogenic acid is degraded to caffeic acid, and then further transformed into dihydrocaffeic acid and other catabolites. The chemical structure of dihydrocaffeic acid is almost identical to caffeic acid; the only difference is that caffeic acid possesses a double bond, while dihydrocaffeic acid has this bond saturated with two hydrogen atoms (hence, the prefix dihydro) ().
Figure 1. Chemical structures of caffeic and dihydrocaffeic acids.
Figura 1. Las estructuras químicas de los ácidos cafeico y dihidrocafeico
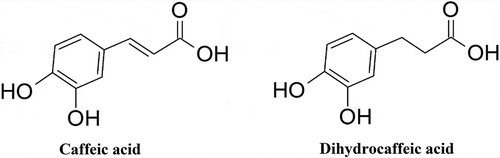
Dihydrocaffeic acid has shown several beneficial activities for health, including antioxidant (S. Wang et al., Citation2019a), cardioprotective (Tang et al., Citation2011), and neuroprotective effects (Lee et al., Citation2015). However, information about its anticancer activity is very scarce. Therefore, the main objective of this short communication was to determine the anticancer potential of dihydrocaffeic acid by evaluating its effect on the viability of several cancer cell lines and compare it with healthy cells.
2. Materials and methods
2.1. Materials
Healthy human cell line HDFa (primary dermal fibroblasts, PCS-201-012), human cancer cell lines MCF-7 (breast, HTB-22), Hep-G2 (liver, HB-8065), PC-3 (prostate, CRL-1435), and HCT-116 (colon, CCL-247), and Dulbecco’s modified Eagle medium-F12 (DMEM-F12) with 3.15 g/L of D-glucose and 0.37 g/L of L-glutamine, were obtained from American Type Culture Collection (ATCC, Manassas, VA, USA). Fetal bovine serum (FBS) was purchased from Gibco Invitrogen (Carlsbad, CA, USA). CellTiter 96® AQueous One Solution Proliferation Assay kit was acquired from Promega (Madison, WI, USA). Antibiotic-antimycotic (Anti-Anti) solution (10,000 U/mL penicillin, 10,000 µg/mL streptomycin, 25 µg/mL amphotericin B) was obtained from Lonza Group AG (Basel, Switzerland). Dihydrocaffeic acid and dimethyl sulfoxide (DMSO) were purchased from Merck (Kenilworth, NJ, USA).
2.2. Cell culture
All cells were grown in DMEM-F12 supplemented with 10% FBS and adjusted to pH 7.4. Cells were maintained at 80 – 90% confluency in tissue culture Petri dishes and incubated in a humidified incubator at 37°C and 5% CO2. Culture medium was changed every 3 days.
2.3. Cell viability assay
Cell viability was assessed using the MTS assay based on Pacheco-Ordaz et al. (Citation2018) with some modifications. Cells were seeded in tissue culture 96-well plates at a density of 15,000 cells/well in 100 µL of the same culture medium (section 2.2) supplemented with 1% Anti-Anti, and incubated for 5 h in a humidified incubator at 37°C and 5% CO2. A stock solution of dihydrocaffeic acid was prepared in DMSO (240 mM) and serially diluted in culture medium. A volume of 100 µL of each dihydrocaffeic acid dilution was added to the cells to a final concentration of 50–1200 μM. All wells had 0.5% DMSO and 1% Anti-Anti. Cells were incubated for 24 h at the same culture conditions. After treatment, 20 µL of CellTiter 96® were added and cells were incubated for 1 h. Absorbance was read at 490 nm in a microplate reader (Synergy MX, BioTek Instruments, Winooski, VT, USA). Cells with medium and 0.5% DMSO were used as controls and considered as 100% viable. A second control with only medium was used to verify that 0.5% DMSO was not cytotoxic to the cells, and indeed no cytotoxicity was observed (data not shown). The 50%, 75%, and 90% cytotoxic concentrations (CC50, CC75, and CC90) were defined as the compound’s concentration required for reduction of cell viability by 50%, 75%, and 90%, respectively. For each cell line, dose-effect data were collected and fit to the 4-parameter logistic (4PL) regression model with at least 6 points using the free online software Quest Graph™ (AAT Bioquest, Inc., Sunnyvale, CA, USA, Citation2019). The resulting dose-effect curves were used to determine CC50, CC75, and CC90. Three independent experiments were done on different days, each experiment with three replicates.
2.4. Statistical analysis
Results were expressed as means ± standard errors. Significant statistical differences were determined by One-Way Analysis of Variance (ANOVA), followed by Tukey’s test (p < .05) using Minitab 18 software (State College, PA, USA).
3. Results and discussion
Dihydrocaffeic acid affected differently each cell line in a dose-dependent manner (). Based on the cytotoxic dose ranges (CC50 – CC90), the order from the least to the most affected cell line was Hep-G2 (529.47–1127.63 μM) < HDFa (501.35–884.45 μM) < HCT-116 (230.05–732.94 μM) < MCF-7 (306.52–540.29 μM) < PC-3 (162.15–389.55 μM). Dihydrocaffeic acid was significantly more cytotoxic for most cancer cell lines, including MCF-7, PC-3, and HCT-116, compared with HDFa healthy cells. However, Hep-G2 was significantly more resistant than HDFa.
Table 1. Cytotoxic doses of dihydrocaffeic acid on cancer and healthy cell lines.
Tabla 1. Las dosis citotóxicas del ácido dihidrocafeico en líneas celulares cancerígenas y sanas
The different sensitivities of the cell lines towards dihydrocaffeic acid could be due to the different genetic profiles of each cell line, which can lead to variations in the rate at which the compound is metabolized, expression of detoxifying enzymes, and the efficacy of the efflux systems (Papi et al., Citation2013). The mechanism of action through which dihydrocaffeic acid exerts its toxicity against cancer cells remains to be elucidated. It can be hypothesized, however, that the mechanism may be similar to caffeic acid, since their chemical structures are almost identical (). Caffeic acid has shown anticancer activity against liver, breast, and colon cancers. In Hep-G2 liver cancer cells, caffeic acid caused a decrease in viability by 40% with a 200 μg/mL dose (Chung et al., Citation2004). Furthermore, caffeic acid caused a 30% decrease in proliferation and 13% cell death with a 50 μM dose in the hormone dependent MCF-7 breast cancer cells (Rosendahl et al., Citation2015). The authors associated these effects with a reduction in estrogen receptor (ER) and insulin-like growth factor-I receptor (IGFIR) levels, as well as a decrease in Akt phosphorylation. Murad et al. (Citation2015) found a moderate decrease in viability (35%) of HT-29 colon cancer cells with 80 μM of caffeic acid. These observations were related to an increase in the uptake of caffeic acid by the HT-29 cells, which lead to apoptosis and cell cycle arrest in the G0/G1 phase. Jaganathan (Citation2012) also reported a cytotoxic effect on HCT-15 colon cancer cells (CC50 = 800 μM, CC75 and CC90 between 1000 and 2500 μM). An increase in the sub-G1 arrest was observed in a dose-dependent manner and this was associated with an increase in reactive oxygen species (ROS) and mitochondrial dysfunction.
4. Conclusions
In this study, it was proven that dihydrocaffeic acid is more cytotoxic to several cancer cell lines than healthy cells; therefore, it is a potential candidate for cancer prevention and treatment. More research will be needed to determine the specific mechanisms by which dihydrocaffeic acid exerts its anticancer activity.
Declaration of Interest Statement
The authors declare no conflict of interest.
Acknowledgments
This work was supported by Bioprocess & Synthetic Biology and NutriOmics research groups of Tecnologico de Monterrey. Author J.S.G. acknowledges CONACYT’s scholarship #291137.
References
- AAT Bioquest, Inc. (2019, December 17). Quest Graph™ four parameter logistic (4PL) curve calculator. https://www.aatbio.com/tools/four-parameter-logistic-4pl-curve-regression-online-calculator
- Chung, T.-W., Moon, S.-K., Chang, Y.-C., Ko, J.-H., Lee, Y.-C., Cho, G., … Kim, C.-H. (2004). Novel and therapeutic effect of caffeic acid and caffeic acid phenyl ester on hepatocarcinoma cells: Complete regression of hepatoma growth and metastasis by dual mechanism. The FASEB Journal, 18(14), 1670–1681. https://doi.org/10.1096/fsb2.v18.14
- Deka, S. J., Gorai, S., Manna, D., & Trivedi, V. (2017). Evidence of PKC binding and translocation to explain the anticancer mechanism of chlorogenic acid in breast cancer cells. Current Molecular Medicine, 17(1), 79–89. https://doi.org/10.2174/1566524017666170209160619
- Ekbatan, S. S., Sleno, L., Sabally, K., Khairallah, J., Azadi, B., Rodes, L., Prakash, S., Donnelly, D. J., & Kubow, S. (2016). Biotransformation of polyphenols in a dynamic multistage gastrointestinal model. Food Chemistry, 204, 453–462. https://doi.org/10.1016/j.foodchem.2016.02.140
- Hou, N., Liu, N., Han, J., Yan, Y., & Li, J. (2017). Chlorogenic acid induces reactive oxygen species generation and inhibits the viability of human colon cancer cells. Anti-cancer Drugs, 28(1), 59–65. https://doi.org/10.1097/CAD.0000000000000430
- Jaganathan, S. K. (2012). Growth inhibition by caffeic acid, one of the phenolic constituents of honey, in HCT 15 colon cancer cells. The Scientific World Journal, 2012, 372345. https://doi.org/10.1100/2012/372345
- Lee, K., Lee, B.-J., & Bu, Y. (2015). Protective effects of dihydrocaffeic acid, a coffee component metabolite, on a focal cerebral ischemia rat model. Molecules, 20(7), 11930–11940. https://doi.org/10.3390/molecules200711930
- Ludwig, I. A., de Peña, M. P., Cid, C., & Crozier, A. (2013). Catabolism of coffee chlorogenic acids by human colonic microbiota. Biofactors, 39(6), 623–632. https://doi.org/10.1002/biof.1124
- Matsunaga, K., Katayama, M., Sakata, K., Kuno, T., Yoshida, K., Yamada, Y., … Mori, H. (2002). Inhibitory effects of chlorogenic acid on azoxymethane-induced colon carcinogenesis in male F344 rats. Asian Pacific Journal of Cancer Prevention, 3(2), 163–166. http://journal.waocp.org/?sid=Entrez:PubMed&id=pmid:12718596&key=2002.3.2.163
- Murad, L. D., Soares, N. C. P., Brand, C., Monteiro, M. C., & Teodoro, A. J. (2015). Effects of caffeic and 5-caffeoylquinic acids on cell viability and cellular uptake in human colon adenocarcinoma cells. Nutrition and Cancer, 67(3), 532–542. https://doi.org/10.1080/01635581.2015.1004736
- Olthof, M. R., Hollman, P. C. H., Buijsman, M. N. C. P., van Amelsvoort, J. M. M., & Katan, M. B. (2003). Chlorogenic acid, quercetin-3-rutinoside and black tea phenols are extensively metabolized in humans. The Journal of Nutrition, 133(6), 1806–1814. htt ps://doi.org/10.1093/jn/133.6.1806
- Pacheco-Ordaz, R., Antunes-Ricardo, M., Gutiérrez-Uribe, J. A., & González-Aguilar, G. A. (2018). Intestinal permeability and cellular antioxidant activity of phenolic compounds from mango (Mangifera indica cv. Ataulfo) peels. International Journal of Molecular Sciences, 19(2), 514. https://doi.org/10.3390/ijms19020514
- Papi, A., Farabegoli, F., Iori, R., Orlandi, M., De Nicola, G. R., Bagatta, M., … Ninfali, P. (2013). Vitexin-2-O-xyloside, raphasatin and (-)-epigallocatechin-3-gallate synergistically affect cell growth and apoptosis of colon cancer cells. Food Chemistry, 138(2–3), 1521–1530. https://doi.org/10.1016/j.foodchem.2012.11.112
- Rosendahl, A. H., Perks, C. M., Zeng, L., Markkula, A., Simonsson, M., Rose, C., … Jernström, H. (2015). Caffeine and caffeic acid inhibit growth and modify estrogen receptor and insulin-like growth factor I receptor levels in human breast cancer. Clinical Cancer Research, 21(8), 1877–1887. https://doi.org/10.1158/1078-0432.CCR-14-1748
- Santana-Gálvez, J., Cisneros-Zevallos, L., & Jacobo-Velázquez, D. A. (2017). Chlorogenic acid: Recent advances on its dual role as a food additive and a nutraceutical against metabolic syndrome. Molecules, 22(3), 358. https://doi.org/10.3390/molecules22030358
- Tang, Y., Wang, M., Le, X., Meng, J., Huang, L., Yu, P., … Wu, P. (2011). Antioxidant and cardioprotective effects of Danshensu (3-(3,4-dihydroxyphenyl)-2-hydroxy-propanoic acid from Salvia miltiorrhiza) on isoproterenol-induced myocardial hypertrophy in rats. Phytomedicine, 18(12), 1024–1030. https://doi.org/10.1016/j.phymed.2011.05.007
- Wang, S., Sarriá, B., Mateos, R., Goya, L., & Bravo-Clemente, L. (2019a). TNF-α-induced oxidative stress and endothelial dysfunction in EA.hy926 cells is prevented by mate and green coffee extracts, 5-caffeoylquinic acid and its microbial metabolite, dihydrocaffeic acid. International Journal of Food Sciences and Nutrition, 70(3), 267–284. https://doi.org/10.1080/09637486.2018.1505834
- Wang, X., Liu, J., Xie, Z., Rao, J., Xu, G., Huang, K., … Yin, Z. (2019b). Chlorogenic acid inhibits proliferation and induces apoptosis in A498 human kidney cancer cells via inactivating PI3K/Akt/mTOR signalling pathway. Journal of Pharmacy and Pharmacology, 71(7), 1100–1109. https://doi.org/10.1111/jphp.13095
- Yamagata, K., Izawa, Y., Onodera, D., & Tagami, M. (2018). Chlorogenic acid regulates apoptosis and stem cell marker-related gene expression in A549 human lung cancer cells. Molecular and Cellular Biochemistry, 441(1–2), 9–19. https://doi.org/10.1007/s11010-017-3171-1
- Zhang, F., Yin, G., Han, X., Jiang, X., & Bao, Z. (2019). Chlorogenic acid inhibits osteosarcoma carcinogenesis via suppressing the STAT3/Snail pathway. Journal of Cellular Biochemistry, 120(6), 10342–10350. https://doi.org/10.1002/jcb.v120.6
