ABSTRACT
The aim of this study was to assess the possibility of using selected bacterial strains (lactic acid bacteria – LAB) isolated from traditional fermented plant-origin food, to produce potentially functional (probiotic and/or synbiotic) cereal-based vegan desserts. The scope of the study included: optimization of the recipe and fermentation conditions of cereal raw materials, taking into account the an adequate concentration of the number of probiotic bacteria in the product at a minimum level of 6 log CFU g−1), an accepted sensory quality of the final product after the fermentation process and estimation of microbiological and sensory quality of developed products after 21 days refrigerated storage. The level of vitamin B12 in fresh products and after refrigerated storage was also determined. The results showed that it is possible to develop safe, potentially functional cereal-based vegan desserts with an appropriate number of LAB bacteria, accepted sensory quality and content of vitamin B12.
RESUMEN
El presente estudio se propuso evaluar la posibilidad de utilizar cepas bacterianas seleccionadas (cultivos lácticos - LAB) aisladas de alimentos tradicionales fermentados de origen vegetal, para producir postres veganos potencialmente funcionales (probióticos y/o simbióticos) a base de cereales. El alcance del estudio incluyó: la optimización de la receta y de las condiciones de fermentación de las materias primas de cereales, teniendo en cuenta la concentración adecuada del número de bacterias probióticas en el producto a un nivel mínimo de 6 log UFC g−1, el logro de una calidad sensorial aceptada del producto final después del proceso de fermentación y la estimación de la calidad microbiológica y sensorial de los productos elaborados después de 21 días de almacenamiento refrigerado. Asimismo, se determinó el nivel de vitamina B12 en los productos frescos y tras el almacenamiento refrigerado. Los resultados dan cuenta de que es posible elaborar postres veganos a base de cereales, seguros y potencialmente funcionales, con un número adecuado de bacterias LAB, una calidad sensorial que reciba aceptación y cierto contenido de vitamina B12.
1. Introduction
Probiotic microorganisms are defined as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host”. This definition is inclusive of a broad range of microbes and applications, whilst capturing the essence of probiotics (microbial, viable and beneficial to health) (Hill et al., Citation2014). In the past few years, several studies indicated that cereals represent a good substrate for the growth and survival of probiotic bacteria (Altamirano-Fortoul et al., Citation2012; Coda et al., Citation2011; Mridula & Sharma, Citation2015; Muyanja et al., Citation2012; Omotade et al., Citation2015; Salmeron et al., Citation2015; Zhang et al., Citation2018).
The development of functional fermented cereal products through application of defined LAB meets the current demand for healthier and more diversified foods. Because of the fermentation process and the production of lactic acid by LAB, the consequent pH decrease, improves the bioavailability of minerals. Moreover, the production of organic acids is responsible for a lower degree of starch gelatinization, which would probably explain the diminished starch digestibility leading to low glycaemic responses. Therefore, the fermentation process can be considered as the most suitable method for improvement of sensory attributes and nutritional value, and the shelf life of final products (Nadeem et al., Citation2010; Sharma et al., Citation2017).
The full prediction of the final sensory attributes, however, is still a very complex task due to dynamic interactions between starter cultures, substrate and fermentation conditions. The cultures used in food fermentation can also contribute by secondary reactions to the formation of flavour and texture. The LAB fermentation can be recognized as a suitable tool for improving poor flavours of raw cereals. Carboxylic acids and aldehydes are examples of the most prominent taste and flavour compounds (Peyer et al., Citation2016).
Cereals are considered one of the most important sources of nutrients and beneficial compounds for human nutrition. Since antinutritional factors such as phytate, trypsin inhibitor, and polyphenols are present, the bioavailability of cereal minerals is typically poor. As it is present in most cereals and has a strong binding affinity for complex multi-charged metal ions, particularly Zn, Ca, and Fe, phytic acid is the most important antinutrient, inhibiting their absorption in the human body. Many technologies are used for the processing of cereals into food matrix. With the exception of vegetables, fruits and similar products, cereals are the most commonly fermented plant-origin materials (Nadeem et al., Citation2010; Trząskowska, Citation2013).
Furthermore, cereal grains are a good source of non-digestible carbohydrates, which can function as prebiotics by selectively stimulating the growth of Lactobacilli and Bifidobacteria in the colon, in addition to promoting many beneficial physiological effects (Reid, Citation2008).
Vegans, in particular, are searching for alternatives to supplement their diet, which is really difficult to maintain, as they choose to eliminate animal-derived foods in their daily meals. Vegan consumers eat less fat overall, including saturated fats, cholesterol, vitamin B12, vitamin D, Fe, Ca, and Zn. Whereas a lower supply of ingredients such as fat or cholesterol is positive in the aspect of health, but on the other hand deficiencies of Fe, Ca, vitamin B12 or vitamin D can be dangerous for them. Fermented cereal products are a good example of bioactive ingredients found in raw cereal content as well as microorganisms that can help in the proper balance of a non-animal-origin product diet (Dewell et al., Citation2008; Waldmann et al., Citation2003).
Human body cannot produce vitamin B12, but has nutritional requirements for it and they obtain vitamin B12 mainly from foods derived from animals, such as milk, meat and eggs. Therefore, vegetarians and especially vegans, can be deficient in this vitamin. Alternative sources of vitamin B12 for people who only consume products of plant origin are therefore sought. According to LeBlanc et al. (Citation2011) the use of vitamin-producing LAB could be a cost-effective alternative to current vitamin fortification programmes and be useful in the elaboration of novel vitamin-enriched products.
It has recently appeared that there is a modern tendency in development of vegan probiotic products. However, since most strains currently available are not isolated from plant-origin matrices, the source of the strain can compromise the vegan food status (Colombo Pimentel et al., Citation2021). Zielińska and Kołożyn-Krajewska (Citation2018) report that, extending the term “probiotic” to include bacteria isolated from traditionally, spontaneously fermented foods seems justified. Microbiota isolated from fermented products constitute the microbiota of an environment in which the products were produced. If they are tested, particularly in terms of their probiotic properties and safety, they may constitute an interesting alternative to gut bacteria.
Therefore, the aim of this study was the development of potentially probiotic and/or synbiotic cereal-based vegan desserts using the selected bacterial strains (LAB) isolated from Polish traditional plant-origin spontaneously fermented food and the determination of the quality of novel products during refrigerated storage.
2. Materials and methods
In , the main stages and experimental tasks of the study with performed analyses were shown. The studies were carried out in two main stages. The purpose of the preliminary studies was to select the strain of bacteria with the best technological properties, such as the best growth in the cereal food matrix to obtain a product with the best sensory acceptability, choice of fermentation process conditions (time and temperature) and optimization of recipes for manufactured desserts. In appropriate tests, three batches of products with different levels of prebiotic additions were produced. Storage tests were also conducted.
Table 1. Main stages and experimental tasks of study.
Tabla 1. Principales etapas y tareas experimentales del estudio
2.1. Probiotic bacterial strains and growth conditions
In this study were used five potentially probiotic strains isolated from traditional fermented cucumber pickles: L. casei O14 (GenBank accession KM 186154); L. plantarum O20 (GenBank accession KM 186158); L. plantarum O21 (GenBank accession KM 186159); L. plantarum O23 (GenBank accession KM 186161); L. brevis O24 (GenBank accession KM 186162) and one from cabbage pickles L. johnsonii K4 (GenBank accession KM 186165). The selected probiotic properties of these strains were described by Zielińska et al. (Citation2015).
The well-studied commercial probiotic strains: L. plantarum 299 V (DSM 9843) isolated from the Sanprobi IBS dietary supplement (Sanprobi Co., Szczecin, Poland) and L. rhamnosus GG (ATCC 53103) isolated from the Dicoflor 6 dietary supplement (Bayer AG Co., Leverkusen, Germany) were also used as the reference strains.
The bacterial strains were stored at −80°C in 20% (m/w) glycerol and were cultivated by two-fold passage in Vegitone MRS broth modified (Sigma-Aldrich Co., Darmstadt, Germany), using 1% v/v of inoculum. In our study, the aim of using Vegitone MRS broth modified was to completely eliminate animal compounds. Thanks to that, the final product could be intended for vegan diet. Incubation was provided at 37°C for 24 h. For comparison of the LAB bacteria count in different growth medium, MRS broth was also used (Biokar Diagnostics Co., Allonne, France). The cultures were centrifuged at 10.000 × g for 5 min. The cell pellets were obtained, washed in distilled water and then resuspended in 10 mL of sterile 0.85% saline solution to its original volume.
The probiotic bacteria count in the preparation starter cultures was approximately 9 log CFU (colony forming units) ml−1. The bacterial strain was added in the amount of 1% v/v, 4% v/v and 6% v/v of the cereal-based dessert (stage 1 of the study; ) according to data from Angelov et al. (Citation2006) and Gupta et al. (Citation2010). Finally, the level of inoculum addition was determined to 6% v/v (stage 2 of the study, ).
2.2. Preparation of cereal-based dessert recipes
The materials used for the fermentation process were: millet flour (Radix-bis Co., Rotmanka, Poland), oat flour (Melvit Co., Ostrołęka, Poland), short-chain inulin (Symbio Co., Lublin, Poland), sucrose (Diamant Co., Gostyń, Poland) and distilled water. The cereal-based potentially probiotic (sample P1) and synbiotic (samples P2; P3) vegan desserts were obtained from a mixture of selected ingredients submitted to a gelatinization process (90°C for 10 min.). All mixtures contained oat flour (4% w/v), millet flour (4% w/v), sucrose (4% w/v) and distilled water depending on the type of sample, 78% v/v (P1), 77% v/v (P2) and 75% v/v (P3), respectively. The mixtures of the presented compositions were prepared with a final volume 300 mL. The gelatinization process was included to achieve a proper creamy texture, which is desired in similar vegetable products, as well as to prevent syneresis (water phase separation) during processing and storage. Next, the products were cooled down to 37°C. Afterwards the prebiotic – inulin was added: P2 (1% w/v) P3 (3% w/v), respectively. Then the startup LAB bacteria culture, which was prepared earlier, was grafted and put to fermentation in 37°C for 20 hours (Marko et al., Citation2014), simultaneously in experimentally determined conditions. The process was conducted in sterile, glass vessels of 500 mL capacity. After the fermentation, some flavouring was added – peach jam (Symbio Co.) in the proportion of 10% w/v of the fermented product.
At stage 1 () related to the selection of the strain needed for the fermentation, some preliminary conditions were applied to the process: 37°C for 20 hours (according to Marko et al., Citation2014), 7% w/v cereal raw material addition (Angelov et al., Citation2006), 6% v/v inoculum addition (Gupta et al., Citation2010), 4% w/v sucrose addition, 10% w/v fruit jam addition. At stage 1 of the study (), there was no prebiotic addition included in the composition. Each batch of cereal-based desserts was produced in triplicates for each time period (0, 7, 14, 21 days) of refrigerated storage at 4°C .
2.3. Microbiological analyses
The analyses were carried out using the TEMPO ® System, automated quality in-dicator solution (BioMérieux, Mercy Etoile, France). The calculation of bacteria number in the study samples (log CFU g−1), according to the TEMPO ® System, is based on the most probable number (MPN) method. The dilution of the samples was 1/400 in a single vial. The inoculated medium was moved into the Tempo card by Tempo Filler. After filling, the cards were incubated at 25°C for 22 h for the counting of Enterobacteriaceae (EB), at 25°C for 72 h for the counting of Total yeast and mold count (YM), at 37°C for 24 h for the counting of Escherichia coli (EC), at 30°C for 48 h for the counting of Aerobic count (AC), at 35°C for 24 h for the counting of Coliform count (CC) (Association of Official Analytical Chemists [AOAC], Citation2020). The Tempo LAB test was able to achieve performance levels that were comparable to the NF ISO15214 (NF ISO 15214, Citation1998) standard, the cards were incubated at 37°C for 48 h. The software system that decides which of the wells tested positive automatically analyzed the data. The number of positive wells obtained in relation to the volume of wells and sample dilution allowed for automatic enumeration of the results in CFU g−1.
2.4. pH measurement
The changes in pH values of products were measured in fresh products and after 7, 14, 21 days of storage at 4°C in triplicate using a pH meter (Elmetron, CP551, Zabrze, Poland).
2.5. Sensory analysis
The sensory evaluations were conducted with the participation of 10 staff members of the Department of Food Gastronomy and Food Hygiene, WULS. The panellists were trained with regard to basic sensory evaluation method (ISO Citation8586–2, 1994). All evaluations were made in triplicate. The basis of obtaining the average results was 30 individual performances.
To evaluate the overall acceptance of cereal desserts, at stage 1 of the study (), the ranking method according to International Standard EN ISO Citation8587(2006) was used. A higher average of mean rank meant a more desirable sample.
The sensory quality of cereal vegan products was assessed as fresh products and after 7, 14, 21 days of storage (stage 2; ) at 4°C using QDA (Quantitative Descriptive Analysis; International Standard EN ISO 13299, Citation2016). Finally, 16 sensory attributes were measured to quantify the quality of the tested products: appearance and texture: thickness, smoothness, delamination; odour: fruity, yoghurt, cereal, other, flavour: fruity, yoghurt, cereal, sweet, storage, other and bitter taste, acidic taste as well as overall quality. The panelists’ aim was to determine the intensity of each of the quality attributes mentioned and to conduct their assessment on an appropriate scale (unstructured graphical scale, 0–10 c.u.). On the basis of the evaluation of the features mentioned, the overall sensory quality of the products was additionally determined on a separate scale.
2.6. Determination of vitamin B12 content
Vitamin B12 content in investigated cereal-based desserts was evaluated according to (Campos-Giménez et al., Citation2008). Vitamin B12 was extracted in the sodium acetate buffer in the presence of sodium cyanide (100°C for 30 minutes) and was determined by liquid chromatography (conditions: flow rate, 0.4 mL/min; injection volume, 50 µL) with UV detection (361 nm).
2.7. Statistical analysis
Results were elaborated with the use of Statistica 13.3 program (StatSoft, Kraków, Poland). All experiments in this study were performed in triplicate.
A one-way analysis of variance (Anova), Friedman test, Tukey test were used for the statistical analysis. The significance level p = .01 was assumed only for sensory evaluation. In the case of other data, the significance level p = .05 was used.
The Principal Component Analysis (PCA) was used to interpret the sensory evaluation results of developed products during 21 days refrigerated storage. This statistical method is commonly used in high-dimensional data analysis to compact multivariate datasets and expose their hidden structure (Granato et al., Citation2018). In this study, 3 principal components were identified, of which the total of the components explains 52.4% of the total variance of variables, and the total of the first two principal components – 42.3% of variance of variables.
3. Results and discussion
3.1. Preliminary research
3.1.1. Selection of bacterial strain for fermentation of cereal based dessert mixture
While the bacterial strain that was after all supposed to be used in the manufacturing of fermented cereal products was being selected, the following factors were taken into account: ability to grow on a non-animal-origin substrate, capability of multiplication in food matrix during the fermentation, sensory acceptability of the fermented products. Also the immunity of bacterial strains to the conditions of a model digestive system, based on literature data (Zielińska et al., Citation2015) was very important.
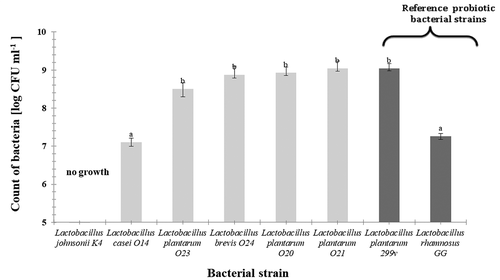
Growth of selected LAB bacterial strains in the plant-origin substrate-Vegitone (count of bacteria in 24-hours bacterial culture) is shown in . Among the eight tested strains of bacteria, one (L. johnsonii K4) did not grow in used substrate. Other four of the investigated strains that had been isolated from food and one reference probiotic bacterial strain showed growth higher than 8.5 log CFU ml−1 on the plant substrate, what allowed their application as starter cultures in fermentation process. Those were: L. plantarum O23, L. brevis O24, L. plantarum O20, L. plantarum O21 and L. plantarum 299 V ().
Figure 1. Growth of selected LAB bacterial strains in the plant-origin substrate-Vegitone (count of bacteria in 24-hours bacterial culture). Explanatory notes: The results are expressed as the mean ± standard deviation (n = 3). Mean values denoted by the same letters aren’t significantly different (p > .05).
Figura 1. Crecimiento de las cepas bacterianas LAB seleccionadas en el sustrato de origen vegetal-Vegitone (recuento de bacterias en cultivo bacteriano de 24 horas). Notas explicativas: Los resultados indican la media ± desviación estándar (n = 3). Los valores medios indicados con las mismas letras no son significativamente diferentes (p > .05) para las muestras en un tipo de medio utilizado
At the next stage of the study, the cereal pilot products were derived and their microbiological and sensory quality was assessed. Considering significant differences in the number of bacteria between the LAB strains in 48-hour culture, using different growth medium: MRS Broth, Vegitone MRS Broth Modified (), the high resistance to conditions in the gastrointestinal tract of used bacterial strains (low pH, presence of bile salts) (Zielińska et al., Citation2015), the high count of bacteria in fresh pilot products (higher than 8.0 log CFU ml−1, data not shown), the high sensory acceptance of fermented products () and, L. plantarum O21 was qualified for further research.
Figure 2. Count of LAB in 48-hour culture, depending on the growth medium used (MRS Broth, vegitone MRS broth modified). Explanatory notes: The results are expressed as the mean ± standard deviation (n = 3). Mean values denoted by the same letters are not significantly different (p > .05) for samples in one type of used medium.
Figura 2. Recuento de LAB en un cultivo de 48 horas, en función del medio de cultivo utilizado (Caldo MRS, Caldo MRS modificado de Vegitone). Notas explicativas: Los resultados indican la media ± desviación estándar (n = 3). Los valores medios indicados con las mismas letras no son significativamente diferentes (p > .05)
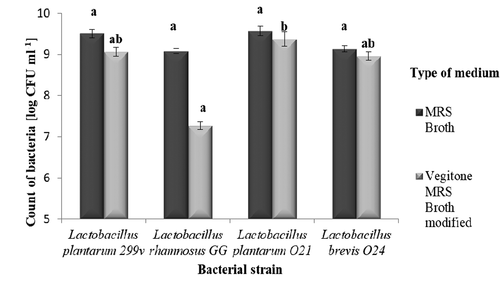
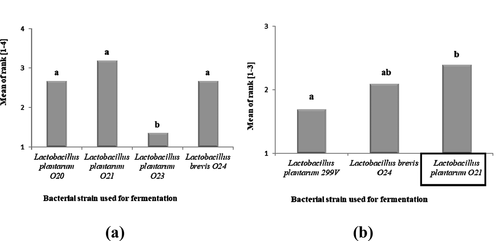
Figure 3. (a) Overall acceptance of cereal-based desserts fermented at temp. 37°C for 16 h, using L. plantarum O20, L. plantarum O21, L. plantarum O23, L. brevis O24 (a ranking method, n = 3). (b) Overall acceptance of cereal-based desserts fermented at temp. 37°C for 16 h, using L. plantarum 299 V, L. brevis O24, L. plantarum O21 (a ranking method, n = 3). Explanatory notes: Mean values denoted by the same letters are not significantly different (p > .01).
Figura 3. (a) Aceptación general de los postres a base de cereales fermentados a una temperatura de 37°C durante 16 h, utilizando L. plantarum O20, L. plantarum O21, L. plantarum O23, L. brevis O24 (método de clasificación, n = 3). (b) Aceptación general de los postres a base de cereales fermentados a una temperatura de 37°C durante 16 h, utilizando L. plantarum 299 V, L. brevis O24, L. plantarum O21 (método de clasificación, n = 3). Notas explicativas: Los valores medios indicados con las mismas letras no difieren de forma estadísticamente significativa (p > .01)
3.2. Choice of fermentation process conditions
After selecting a bacterial strain for fermentation of cereal raw materials, the major process conditions such as time and temperature of fermentation were also determined.
Conditions of fermentation were chosen according to (Angelov et al., Citation2006; Russo et al., Citation2016; Marko et al., Citation2014). The products were fermented at 37°C, in three variants of time (16 h; 20 h; 24 h) (Appendix, ). Considering the number of probiotic bacteria and the highest notes of overall acceptance of dessert fermented at 37°C for 20 h (Figure A1), these technological conditions were adopted for further research.
3.2.1. Optimization of the fermented cereal dessert recipe
Optimization of the fermented cereal dessert recipe included two main aspects: (1) the content of cereal raw materials in terms of obtaining the desired consistency of products and (2) determining the level of starter culture (inoculum), in respect of tenor fermentation process and obtaining repetitive quality of products.
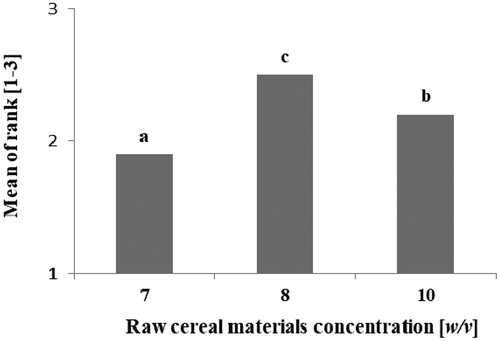
Based on the results achieved by other researchers (Angelov et al., Citation2006; Ghanbari et al., Citation2017) a preliminary experiment was carried out on 3 different levels of raw cereal materials (oat and millet flour, each component 50% w/v): 7% w/v; 8% w/v and 10% w/v (Appendix, ). The selection criterion was to obtain the consistency of the product very similar to thick yogurt. According to the sensory evaluation, the best results were obtained with the option of 8% w/v of total cereal raw materials (Appendix, ). This level was finally adopted in the dessert formula.
When using 1% v/v and 4% v/v inoculum level of strain, it was not possible to obtain reproducible quality of fermented products – some of the samples were spoiled during incubation (changes in colour, unpleasant odour) (data not shown). The application of inoculum at the level of 6% v/v allowed the proper course of fermentation.
3.3. Main research
3.3.1. Control of the harmful microbiota of a fresh cereal-based dessert
Due to the observed signs of spoilage in products at the stage of recipe optimization (1% and 4% inoculum addition) and during storage for 7 days at a cooling temperature (data not shown), the presence of harmful microflora in cereal puddings fermented with the share of the ultimately selected potentially probiotic LAB strain – L. plantarum O21, added according to the recipe at the level of 6% (), was also investigated.
Table 2. Microbiological quality of developed, fresh cereal-based desserts with selected bacterial strain L. plantarum O21.
Tabla 2. Calidad microbiológica de postres frescos a base de cereales elaborados con la cepa bacteriana L. plantarum O21 seleccionada
Fermented foods can be defined as foods in which microbial activity plays an essential role in conferring the required stability, safety and sensory properties to the product. Fermented foods have also a longer shelf life and are safe because of the presence of bacteriocins and a low pH, which hinder the growth of pathogenic bacteria (Prakash, Citation2017).
As shown in , there were no Enterobacteriaceae bacteria, yeast and mould detected at levels that threaten the health safety of the cereal desserts developed. All samples were of good quality and edible due to the fixation of the recipe raw materials as a result of the lactic fermentation process with the participation of the selected strain of potentially probiotic bacteria L. plantarum 021 under established conditions. Russo et al. (Citation2017), demonstrated suitability of 88 L. plantarum strains for the fermentation and antifungal biopreservation of oat-based products.
3.3.2. Evaluation of microbiological, sensory and nutritional quality of products during storage
In the present study, raw cereal materials were fermented with the potentially probiotic L. plantarum O21 strain to produce new potentially functional foods with improved nutritional and technological features. The viability of the selected bacterial strain, physicochemical and sensorial parameters were monitored in fresh products and after 7, 14, 21 days of refrigerated storage.
During the fermentation in selected timeframes, as a result of metabolic activity of L. plantarum O21, a decrease of pH values in each sample was observed. The pH of fermented mass dropped from the initial 6.59–6.61 to the 4.01–4.22, respectively (data not shown). The addition of inulin statistically did not (p > .05) affect the number of LAB bacteria in fermented mass directly after the fermentation process(data not shown) significantly. According to Angelov et al. (Citation2006) different fermentation rates could be attributed both to the bacterial strain used and the differences in cereal media. After 10 h of lactic acid fermentation at 37°C, Kocková et al. (Citation2013) measured the following final pH value in cereal substrates (millet grain) was 5.06, while Martensson et al. (Citation2002) reported pH 3.9–4.5 after longer fermentation (16 h) of an oat base with commercial mixed dairy cultures.
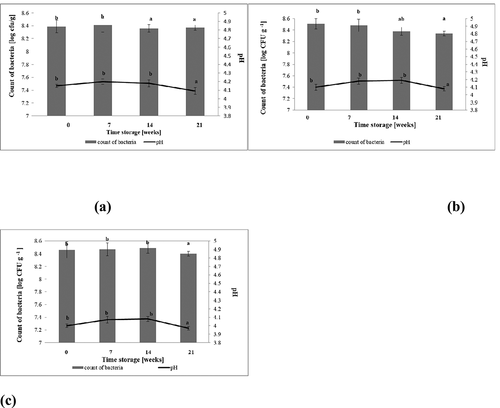
The survival and viability of probiotics has to be determined during the shelf-life of the functional product. The count of viable probiotic bacteria determines this product as an ideal vehicle for probiotic bacteria. As shown in –c), in the case of all samples of cereal desserts, a significant influence of the time of storage on the count of L. plantarum 021 bacteria was observed (p < .05). There was no significant influence (p >.05) of inulin addition on the survivability of probiotic bacteria during 21 days of refrigerated storage. According to Farinha et al. (Citation2015), to improve probiotic survival, prebiotic ingredients can be added to food preparations containing probiotics.
Figure 4. Changes of pH value and survability of L. plantarum 021 in cereal-based desserts during 21 days storage at 4°C (n = 3). (a) The obtained results for P1 cereal based desserts. (b). The obtained results for P2 cereal based desserts. (c). The obtained results for P3 cereal based desserts. Explanatory notes: The results are expressed as the mean ± standard deviation (n = 3). Mean values denoted by the same letters don’t differ statistically significantly (p > .05).
Figura 4. Cambios del valor del pH y de la capacidad de supervivencia de L. plantarum 021 en postres a base de cereales durante 21 días de almacenamiento a 4°C (n = 3). (a) Los resultados obtenidos para los postres a base de cereales P1. (b). Los resultados obtenidos para los postres a base de cereales P2. (c). Los resultados obtenidos para los postres a base de cereales P3. Notas explicativas: Los resultados se expresan como media ± desviación estándar (n = 3). Los valores medios indicados con las mismas letras no difieren de forma estadísticamente significativa (p > .05)
On the other hand, Molin (Citation2001) reports that L. plantarum metabolizes prebiotic-related components such as xylo-oligosaccharides in oatmeal, which could be of critical importance for its growth and for potential health benefits caused by the corresponding production of short chain fatty acids in the intestinal mucosa.
After 14 days of refrigerated storage, the count of the potentially probiotic bacterial strain L. plantarum 021 in all of the analysed samples decreased statistically significantly (p < .05). Over the storage period, products contained the lactic acid bacteria count below 8.34– 8.4 log CFU g−1 (–c)). So even after 21 days of storage, cereal-based desserts contained an appropriate probiotic bacterial number, therefore could be considered as potentially probiotic and /or synbiotic products.
The growth of LAB in the cereal-based desserts in own study was comparable with the results obtained from previous research. Gupta et al. (Citation2010) reported high survival of L. plantarum ATCC (9.5 CFU ml−1) in oat-based drinks after 21 days of storage at the temperature of 4°C. According to Russo et al. (Citation2016) the survival of L. plantarum Lp90 in fermented oat-based foods was higher than 8 log CFU g−1 at the end of the shelf life of products, after 21 days of cold storage.
High viable counts of LAB bacterial strain are necessary to get the desired acid production and reduction in the pH value which affects the shelf-life of the final product. On the basis of our research conducted on storage of cereal-based vegan desserts (–c)), a statistically significant impact (p < .05) of the time of storage on the pH value was found in the case of all samples: potentially probiotic (P1) and potentially synbiotic products with 1% w/w (P2) and 3% w/w (P3) of inulin addition. The pH value of investigated products underwent a slight although statistically significant (p < .05) reduction during 21 days of refrigerated storage, to level 3.97– 4.09, respectively. However, the pH values of samples with addition of inulin (P2; P3) were slightly lower compared to sample P1, without the addition of a prebiotic. Helland et al. (Citation2004) reported that the pH levels in milk cereal-based desserts after 21 days of refrigerated storage at 4–6°C, were reduced to 3.4–4.4. On the other hand, Dharmasena et al. (Citation2015) observed that the pH value of fermented oatmeal coconut water with the addition of inulin, was at the level of 5.07, respectively after 21 days of refrigerated storage.
3.3.3. Evaluation of sensory quality
The results of the sensory evaluation of potentially functional, fermented cereal-based desserts were elaborated by PCA method. The PCA graph of the selected sensory attributes in investigated samples of potentially functional cereal-based vegan desserts is shown in . Samples were grouped into three clusters with their sensory profile and selected time of refrigerated storage (0, 7 and 14 days). The first group constituted samples of three kinds of fresh cereal-based desserts. It had the highest intensity of overall acceptance, sweet taste, fruity flavour, and the highest notes of texture attribute-thickness (). The second indicated group were products after 7-day storage. This group was characterized by lower intensity of sweet taste, overall quality similar as in group I and a higher level of delamination. The third group were samples after 14-day storage. The third group was distinguished by the highest intensity of cereal flavour, acid taste and other odour, which was also determined as acid. These samples had lower notes by overall quality next to the second group of products ().
Figure 5. Principal Component Analysis (PCA) graph of the selected sensory attributes in studied samples of potentially functional cereal-based vegan desserts. Explanatory notes: Cereal-based desserts fermented with L. plantarum 021: 10-fresh, 17-after 7 days, 114-after 14 days, 121-after 21 days storage. Cereal-based desserts fermented with L. plantarum 021, with 1% w/v of inulin addition: 20-fresh, 27-after 7 days, 214-after 14 days, 221-after 21 days storage. Cereal-based desserts fermented with L. plantarum 021, with 3% w/v of inulin addition: 30-fresh, 37-after 7 days, 314-after 14 days, 321-after 21 days storage.
Figura 5. Gráfico de análisis de componentes principales (PCA) de los atributos sensoriales seleccionados en las muestras estudiadas de postres veganos a base de cereales potencialmente funcionales. Notas explicativas: Postres a base de cereales fermentados con L. plantarum 021: 10-fresco, 17-después de 7 días, 114-después de 14 días, 121-después de 21 días de almacenamiento. Postres a base de cereales fermentados con L. plantarum 021, con adición de 1% p/v de inulina: 20-frescos, 27-después de 7 días, 214-después de 14 días, 221-después de 21 días de almacenamiento. Postres a base de cereales fermentados con L. plantarum 021, con 3% p/v de adición de inulina: 30-fresco, 37-después de 7 días, 314-después de 14 días, 321-después de 21 días de almacenamiento.
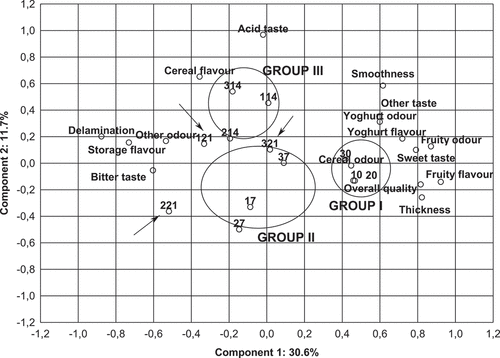
After 21 days of storage, the products did not constitute a separate group in terms of sensory quality. After 21-day refrigerate storage, the sensory assessment indicated among all products the highest overall quality in sample P3, with 3% w/v of inulin addition (). This sample was characterized among of all desserts after 21-day storage, by the highest notes of smoothness, thickness, sweet taste and the lowest notes of delamination as well as an intensity of the bitter taste. Therefore the results of sensory assessment in our study suggest that 3% w/v of inulin addition had a positive impact on the texture of the product during storage and on the flavour by reducing the intensity of the notes (such as a storage flavour and bitter taste), which lowered the overall quality.
In the food technology, inulin is used for its technological properties as a low-calorie sweetener, fat substitute, and also as a texture modifier (Canbulat & Ozcan, Citation2015). A study done by Williams and Hekmat (Citation2017) reported that a fermented probiotic rice pudding sample fortified with 4% w/w short-chain inulin had the highest hedonic score for falvour, sweetness, texture, and overall acceptability. Our study also used short-chain inulin, at the level of 1% w/v (P2) and 3% w/v (P3), respectively. According to Canbulat and Ozcan (Citation2015) short-chain and long-chain inulin in probiotic products revealed that short-chain inulin received higher scores for odour, taste, and overall acceptance than the samples with long-chain inulin. On the other hand, Ghanbari et al. (Citation2017) reported that producers should emphasize on the descriptors which negatively influence the hedonic dimension of cereal-based dairy desserts, because these are possibly the most critical factors for consumer acceptability and as a result the main reason for purchase. These results revealed that liquid and not creamy texture makes desserts with inulin less pleasant for consumers.
3.3.4. Determination of vitamin B12 content
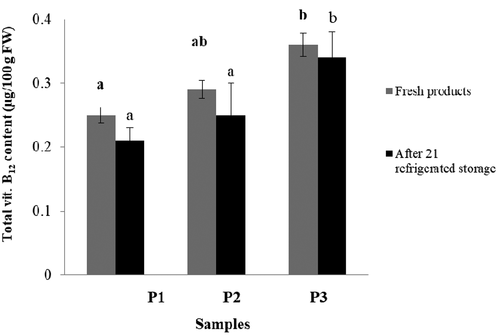
The comparison of vitamin B12 content in fresh products and after 21 days of refrigerated storage was assessed in this research. The obtained results are shown in . The highest significant vitamin B12 content (p < .05) among all fresh samples was observed in the case of a potentially synbiotic dessert with 3% w/v inulin (P3) at 0.36 μg/100 g FW, respectively. There were no statistically significant differences observed between the P1 and P2 samples (p > .05). The refrigerated storage process did not significantly (p > .05) change the vitamin B12 content in the investigated products ().
Figure 6. Comparison of vitamin B12 content in fresh products and after 21 days of refrigerated storage. Explanatory notes: Refer to for identification of test samples. The results are expressed as the mean ± standard deviation (n = 3). Mean values denoted by the same letters aren’t significantly different (p > .05).
Figura 6. Comparación del contenido de vitamina B12 en productos frescos y tras 21 días de almacenamiento refrigerado. Notas explicativas: Consulte la para la identificación de las muestras de prueba. Los resultados se expresan como media ± desviación estándar (n = 3). Los valores medios indicados con las mismas letras no son significativamente diferentes (p > .05)
Cobalamin (vit. B12), a complex corrin compound, was found to be produced by strains of L. reuteri, a well-recognized probiotic species (Taranto et al., Citation2003). On the other hand, genomic studies of the bacterial strain L. corynoformis CRL 1001, isolated from silage, revealed the presence of the complete machinery of the anaerobic biosynthesis pathway of coenzyme B12 (Torres et al., Citation2016). In turn, Saulnier et al. (Citation2011) reported that genome sequencing has shown that L. reuteri strains ATCC PTA 6475 and ATCC 55730 have biosynthetic pathways for vit. B12 and folate synthesis.
The Recommended Dietary Allowances (RDA) for vitamin B12 for adults is 2.4 μg/day (Jarosz et al., Citation2017). The results of own study suggested that about 200 g of developed cereal desserts intake would cover 20–30% of the requirement for active vitamin B12.
4. Conclusions
In conclusion, it is possible to use selected bacterial strain L. plantarum O21 isolated from Polish traditional spontaneously fermented food to produce cereal-based, potentially probiotic and/or synbiotic fermented vegan products containing an appropriate probiotic bacterial number which are also safe and of acceptable sensory quality during 21 days of refrigerated storage. On the premise that the developed cereal-based potentially functional desserts are intended for vegan consumers, it is also very important that such products are fortified in vit. B12 because of the lactic fermentation process with the selected bacterial strain isolated from the plant – origin food matrix. The results of the study provided useful and practical information that could be effective in the development of novel potentially functional cereal-based vegan desserts.
Consent to participate
Informed consent was obtained from each subject prior to their participation in the sensory evaluations carried out in the study.
Data Availability Statement
All data generated or analyzed during this study are included in this article.
Disclosure statement
No potential conflict of interest was reported by the authors.
Additional information
Funding
References
- Altamirano-Fortoul, R., Moreno-Terrazas, R., Quezada-Gallo, A., & Rosell, C. M. (2012). Viability of some probiotic coatings in bread and its effect on the crust mechanical properties. Food Hydrocolloids, 29(1), 166–174. https://doi.org/https://doi.org/10.1016/j.foodhyd.2012.02.015.
- Angelov, A., Gotcheva, V., Kuncheva, R., & Hristozova, T. (2006). Development of a new oat-based probiotic drink. International Journal of Food Microbiology, 112(1), 75–80. https://doi.org/https://doi.org/10.1016/j.ijfoodmicro.2006.05.015.
- Association of Official Analytical Chemists – AOAC. Research institute, 2020. Certification No 080603, TEMPO EC. Certification No 041001, TEMPO YM. Certification No 050801, TEMPO EB. Certification No 060702, TEMPO CC. Certification No 121204, TEMPO AC. BioMérieux S.A. AOAC. Retrieved December 20, 2020, from https://members.aoac.org/AOAC/PTM_Validated_Methods.aspx
- Campos-Giménez, E., Fontannaz, P., Trisconi, M. J., Kilinc, T., Gimenez, C., & Andrieux, P. (2008). Determination of vitamin B12 in food products by liquid chromatography/UV detection with immunoaffinity extraction: Single-laboratory validation. Journal of AOAC International, 91(4), 786–793. https://doi.org/https://doi.org/10.1093/jaoac/91.4.786
- Canbulat, Z., & Ozcan, T. (2015). Effects of short-chain and long-chain inulin on the quality of probiotic yogurt containing Lactobacillus rhamnosus. Journal of Food Processing and Preservation, 39(6), 1251–1260. https://doi.org/https://doi.org/10.1111/jfpp.12343
- Coda, R., Rizzello, C. G., Trani, A., & Gobbetti, M. (2011). Manufacture and characterization of functional emmer beverages fermented by selected lactic acid bacteria. Food Microbiology, 28(3), 526–536. https://doi.org/http://dx.doi.org/10.1016/j.fm.2010.11.001
- Colombo Pimentel, T., Almeida da Costa, W. K., Barão, C. E., Rosset, M., & Magnani, M. (2021). Vegan probiotic products: A modern tendency or the newest challenge in functional foods. Food Research International, 140(110033), 0963–9969. https://doi.org/https://doi.org/10.1016/j.foodres.2020.110033
- Dewell, A., Weidner, G., Summer, M. D., Chi, C. S., & Ornish, D. (2008). A very-low-fat vegan diet increases intake of protective dietary factors and decreases intake of pathogenic dietary factors. Journal of American Dietetic Association, 108(2), 347–356. https://doi.org/https://doi.org/10.1016/j.jada.2007.10.044
- Dharmasena, M., Barron, F., Fraser, A., & Jiang, X. (2015). Refrigerated shelf life of a coconut water-oatmeal mix and the viability of Lactobacillus Plantarum Lp 115-400B. Foods, 10(4(3)), 328–337. https://doi.org/https://doi.org/10.3390/foods4030328
- Farinha, L. R. L., Sabo, S. S., Porto, M. C., Souza, E. C., Oliveira, M. N., & Oliveira, R. P. S. (2015). Influence of prebiotic ingredients on the growth kinetics and bacteriocin production of. Lactococcus Lactis. Chemical Engineering Journal, 43(5), 313–318. https://doi.org/https://doi.org/10.3303/CET1543053
- Ghanbari, M., Mortazavian, A. M., Ghasemi, J. B., Mohammadi, A., Hosseini, H., & Neyestani, T. R. (2017). Formulation and development of a new prebiotic cereal-based dairy dessert: Rheological, sensory and physical attributes. Food Science and Technology Research, 23(5), 637–649. https://doi.org/https://doi.org/10.3136/fstr.23.637
- Granato, D., Putnik, P., Kovacevic, D. B., Santos, J. S., Calado, V., Rocha, R. S., Da Cruz, A. G., Jarvis, B., Rodionova, O. Y., & Pomerantsev, A. (2018). Trends in chemometrics: Food authentication, microbiology, and effects of processing. Comprehensive Reviews in Food Science and Food Safety, 17(466), 663–677. https://doi.org/https://doi.org/10.1111/1541-4337.12341
- Gupta, S., Cox, S., & Abu-Ghannam, N. (2010). Process optimization for the development of a functional beverage based on lactic acid fermentation of oats. Biochemical Engineering Journal, 52(2–3), 199–204. https://doi.org/https://doi.org/10.1016/j.bej.2010.08.008
- Helland, M. H., Wicklund, T., & Narvhus, J. A. (2004). Growth and metabolism of selected strains of probiotic bacteria in milk- and water-based cereal puddings. International Dairy Journal, 14(11), 957–965. https://doi.org/https://doi.org/10.1016/j.idairyj.2004.03.008
- Hill, C., Guarner, F., Reid, G., Gibson, G. R., Merenstein, D. J., Pot, B., Morelli, L., Canani, R. B., Flint, H. J., Salminen, S., Calder, P. C., & Sanders, M. E. (2014). The international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nature Reviews Gastroenterology & Hepatology, 11(8), 506–514. https://doi.org/https://doi.org/10.1038/nrgastro.2014.662491238624912386
- International Standard EN ISO 13299. (2016). Sensory Analysis. Methodology. General guidance for establishing a sensory profile.
- International Standard EN ISO 8587. (2006). Sensory analysis-Methodology – Ranking.
- ISO 8586–2. (1994). Sensory analysis. General guidance for the selection, training and monitoring of assessors – Part 2: Experts.
- Jarosz, M., Stoś, K., Przygoda, B., Matczuk, E., Stolińska-Fiedorowicz, H., & Kłys, W. (2017). Vitamins. In M. Jarosz (Ed.), Nutrition Standards for the polish population (pp. 172–173). Food and Nutrition Institute.
- Kocková, M., Dilongová, M., Hybenová, E., & Valík, L. (2013). Evaluation of cereals and pseudocereals suitability for the development of new probiotic foods. Journal of Chemistry, 2013, 1–8. https://doi.org/https://doi.org/10.1155/2013/414303
- LeBlanc, J. G., Laiño, J. E., del Valle, M. J., Vannini, V., van Sinderen, D., Taranto, M. P., De Valdez, G. F., De Giori, G. S., & Sesma, F. (2011, December). B-group vitamin production by lactic acid bacteria–current knowledge and potential applications. Journal of Applied Microbiology, 111(6), 1297–1309. Epub 2011 Oct 10. PMID: 21933312. https://doi.org/https://doi.org/10.1111/j.1365-2672.2011.05157.x.
- Marko, A., Rakická, M., Mikušová, L., Valík, Ľ., & Šturdík, E. (2014). Lactic acid fermentation of cereal substrates in nutritional perspective. International Journal of Research in Chemistry and Environment, 4(4), 80–92.
- Martensson, O., Öste, R., & Holst, O. (2002). The effect of yoghurt culture on the survival of probiotic bacteria in oat-based, non-dairy products. Food Research International, 35(8), 775–784. https://doi.org/https://doi.org/10.1016/S0963-9969(02)00074-1
- Molin, G. (2001). Probiotics in foods not containing milk or milk constituents, with special reference to Lactobacillus plantarum 299v. . The American Journal of Clinical Nutrition, 73(Suppl. S2), 380–385. https://doi.org/https://doi.org/10.1093/ajcn/73.2.380s
- Mridula, D., & Sharma, M. (2015). Development of non-dairy probiotic drink uti-lizing sprouted cereals, legume and soymilk. LWT - Food Science and Technology, 62(1), 482–487. https://doi.org/https://doi.org/10.1016/j.lwt.2014.07.011
- Muyanja, C. M. B. K., Narvhus, J. A., & Langsrud, T. (2012). Organic acids and volatileorganic compounds produced during traditional and starter culture fer-menta tion of Bushera, a Ugandan fermented cereal beverage, a Ugandan Fermented Cereal Beverage. Food Biotechnology, 26(1), 1e28. https://doi.org/https://doi.org/10.1080/08905436.2011.617252.
- Nadeem, M., Anjum, F. M., Amir, R. M., Khan, M. R., & Hussein, S. (2010). An overview of anti-nutritional factors in cereal grains with special reference to wheat-A review. Pakistan Journal of Food Sciences, 20(1–4), 54–61.
- NF ISO 15214. (1998). Microbiology of food and animal feeding stuffs. Horizontal method for the enumeration of mesophilic lactic acid bacteria. Colony count technique at 30°C.
- Ogunremi, O. R., Agrawal, R., & Sanni, A. I. (2015). Development of cereal-based functional food using cereal-mix substrate fermented with probiotic strain – Pichia kudriavzevii OG32. Food Science & Nutrition, 3, 486–494. https://doi.org/https://doi.org/10.1002/fsn3.239
- Peyer, L., Zannini, E., & Arendt, E. K. (2016). Lactic acid bacteria as sensory biomodulators for fermented cereal-based beverages. Trends in Food Science & Technology, 54, 17–25. https://doi.org/https://doi.org/10.1016/j.tifs.2016.05.009.
- Prakash, J. (2017). Chapter 14 - safety of fermented cereals and legumes. In Prakash, V., Martín-Belloso, O., Keener, L., Astley, S., Braun, S., McMahon, H., & Lelieveld, H., (Eds.), Regulating safety of traditional and ethnic foods (pp. 283–310). Academic Press. https://doi.org/https://doi.org/10.1016/B978-0-12-800605-4.00014-1.
- Reid, G. (2008). Probiotics and prebiotics-progress and challenges. International Dairy Journal, 18(10–11), 969–975. https://doi.org/https://doi.org/10.1016/j.idairyj.2007.11.025
- Russo, P., Arena, M. P., Fiocco, D., Capozzi, V., Drider, D., & Spano, G. (2017). Lactobacillus plantarum with broad antifungal activity: A promising approach to increase safety and shelf-life of cereal-based products. International Journal of Food Microbiology, 247, 48–54. https://doi.org/https://doi.org/10.1016/j.ijfoodmicro.2016.04.027.
- Russo, P., Valeria de Chiara, M. L., Capozzi, V., Pia Arena, M., Amodio, M. L., Rascón, A., Dueñas, M. T., López, P., & Spano, G. (2016). Lactobacillus plantarum strains for multifunctional oat-based foods. LWT - Food Science and Technology, 68, 288–294. https://doi.org/https://doi.org/10.1016/j.lwt.2015.12.040
- Salmeron, I., Thomas, K., & Pandiella, S. S. (2015). Effect of potentially probiotic acticacid bacteria on the physicochemical composition and acceptance of fermented cereal beverages. Journal of Functional Foods, 15, 106e115. https://doi.org/https://doi.org/10.1016/j.jff.2015.03.012
- Saulnier, D. M., Santos, F., Roos, S., Mistretta, T. A., Spinler, J. K., Molenaar, D., Teusink, B., & Versalovic, J. (2011). Exploring metabolic pathway reconstruction and genome-wide expression profiling in Lactobacillus reuteri to define functional probiotic features. PLoS One, 6(4), e18783. https://doi.org/https://doi.org/10.1371/journal.pone.0018783.
- Sharma, P., Trivedi, N., & Gat, Y. (2017). Development of functional fermented whey-oat based product using probiotic bacteria. 3 Biotech, 7(4), 272. https://doi.org/https://doi.org/10.1007/s13205-017-0906-3
- Taranto, M. P., Vera, J. L., Hugenholtz, J., De Valdez, G. F., & Sesma, F. (2003). Lactobacillus reuteri CRL1098 produces cobalamin. Journal of Bacteriology, 185(18), 5643–5647. https://doi.org/https://doi.org/10.1128/JB.185.18.5643-5647.2003
- Torres, A. C., Vannini, V., Bonacina, J., Font, G., Saavedra, L., & Taranto, M. P. (2016). Cobalamin production by Lactobacillus coryniformis: Biochemical identification of the synthetized corrinoid and genomic analysis of the biosynthetic cluster. BMC Microbiology, 16(1), 240. https://doi.org/https://doi.org/10.1186/s12866-016-0854-9
- Trząskowska, M. (2013). Probiotics in products of plant origin. Żywność. Nauka. Technologia. Jakość//Food. Science Technology. Quality, 4(89), 5–20.
- Waldmann, A., Koschizke, J. W., Leitzman, C. (2003). Dietary intakes and life factors of a vegan population in Germany. Results from German Vegan study. European Journal of Clinal Nutrition, 57(8), 947–955. https://doi.org/https://doi.org/10.1038/sj.ejcn.1601629
- Williams, M., & Hekmat, S. (2017). Lactobacillus rhamnosus GR-1 in fermented rice pudding supplemented with short chain inulin, long chain inulin, and oat as a novel functional food. Fermentation, 3(4), 55. https://doi.org/https://doi.org/10.3390/fermentation3040055.
- Zhang, L., Lou, Y., & Schutyser, M. A. I. (2018). 3D printing of cereal-based food structures containing probiotics. Food Structure, 18, 14–22. 2213–3291. https://doi.org/https://doi.org/10.1016/j.foostr.2018.10.002
- Zielińska, D., & Kołożyn-Krajewska, D. (2018). Food-origin lactic acid bacteria may exhibit probiotic properties: Review. Biomed Research International, 2018, 5063185. https://doi.org/https://doi.org/10.1155/2018/5063185
- Zielińska, D., Rzepkowska, A., Radawska, A., & Zieliński, K. (2015). In vitro screening of selected probiotic properties of Lactobacillus strains isolated from traditional fermented cabbage and cucumber. Current Microbiology, 70(2), 183–194. https://doi.org/https://doi.org/10.1007/s00284-014-0699-0
Appendix A
Figure A1. Determination of fermentation process conditions with L. plantarum O21 of cereal-based vegan desserts. Explanatory notes: Mean values denoted by the same letters don’t differ statistically significantly (p > .05 for microbilogical analysis; p > .01 for sensory estimation), (n = 3).
Figura A1. Determinación de las condiciones del proceso de fermentación con L. plantarum O21 de postres veganos a base de cereales. Notas explicativas: Los valores medios indicados con las mismas letras no difieren de forma estadísticamente significativa (p > .05 para el análisis microbiológico; p > .01 para la estimación sensorial), (n = 3)
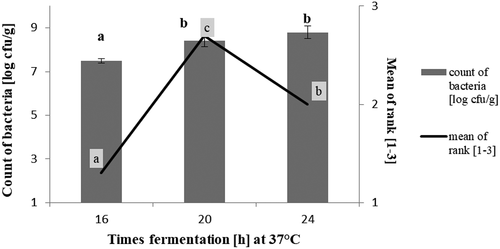
Figure A2. Optimalization recipes of cereal-based vegan dessertss fermented with L. plantarum O21 at 37°C for 24 h. Explanatory notes: The results are expressed as the mean ± standard deviation (n = 3). Mean values denoted by the same letters aren’t significantly different (p > .01).
Figura A2. Recetas de optimización de postres veganos a base de cereales fermentados con L. plantarum O21 a 37°C durante 24 horas. Notas explicativas: Los resultados se expresan como la media ± desviación estándar (n = 3). Los valores medios indicados con las mismas letras no son significativamente diferentes (p > .01)