Figures & data
Figure 1. Degree of hydrolysis of SFMPH hydrolysed with papain, trypsin, and alcalase at various times. Within the same enzymatic action, values (mean ± SD of triplicates) with different letters (a–c, h–j, w–y) are significantly different (Duncan, P < 0.05). A control test (without proteases addition) for each enzymatic treatment was performed according to normalization protocol and its DH value was in the range of 11.8–12.0%.
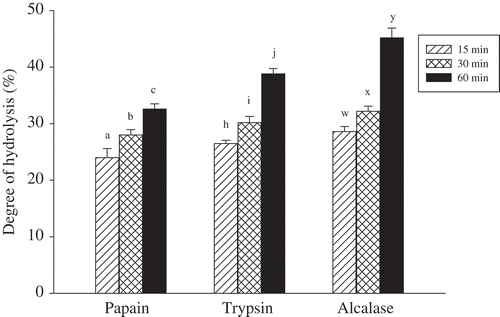
Table 1. The distribution of molecular size fractions in the SFMPH hydrolysed with papain, trypsin, and alcalase.
Table 2. Effect of SFMPH after treatment with papain, trypsin, and alcalase for 60 min on functional propertiesa.
Table 3. Effect of SFMPH after treatment with papain, trypsin, and alcalase for 60 min on disintegration of cholesterol micellesa.
Table 4. ACE inhibitory activity (IC50) of SFMPH hydrolysed with alcalase at various hydrolysis times.
